1. What is the projected Compound Annual Growth Rate (CAGR) of the Laboratory Developed Test?
The projected CAGR is approximately 5.09%.
 Laboratory Developed Test
Laboratory Developed TestLaboratory Developed Test by Type (/> Clinical Biochemistry, Hematology, Immunology, Molecular Diagnostics, Other), by Application (/> Hospitals Laboratory, Clinical Research Organizations, Academic Institutes, Specialty Diagnostic Centers, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Laboratory Developed Test (LDT) market, projected to reach $10.35 billion by 2025, demonstrates robust growth potential. While specific CAGR data requires further analysis, anticipated market expansion, driven by advancements in molecular diagnostics, personalized medicine, and the increasing incidence of chronic diseases, suggests a conservative estimate of a 5.09% CAGR over the forecast period (2025-2033). Key growth drivers include escalating demand for expedited, accurate, and cost-efficient diagnostic solutions, alongside technological breakthroughs such as next-generation sequencing (NGS) and sophisticated bioinformatics. Emerging trends favor personalized medicine, with LDTs central to tailoring treatments to individual genetic profiles. The proliferation of point-of-care testing and telemedicine further enhances diagnostic accessibility. However, regulatory complexities, particularly regarding LDT standardization and validation, represent significant market restraints. The market is segmented by test type (e.g., genetic, infectious disease, oncology), technology platform, and end-user (hospitals, clinics, reference laboratories). Leading entities like Quest Diagnostics, Thermo Fisher, and Roche are instrumental in fostering innovation and market reach through strategic alliances and acquisitions, with specialized LDT providers contributing to niche market development.
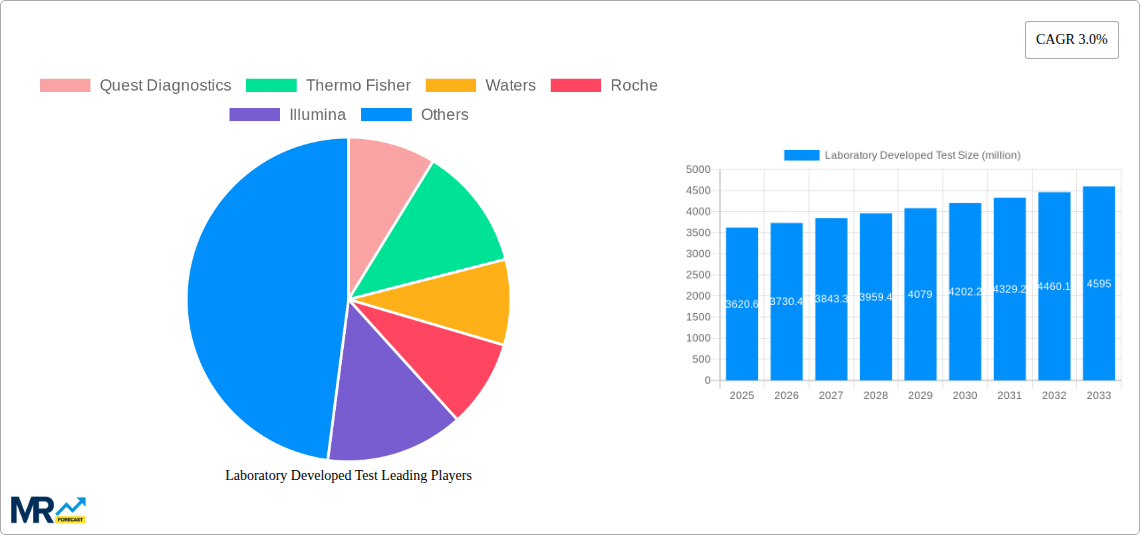

Future LDT market trajectory depends on adeptly navigating regulatory landscapes and integrating cutting-edge technologies. Enhanced R&D investment and strategic collaborations between diagnostic firms and healthcare providers are critical for accelerating market expansion. Sustainable market success necessitates a synergy between innovation, regulatory adherence, and accessibility to optimize healthcare outcomes. This includes addressing data security and privacy concerns associated with sensitive patient data in LDT development and deployment. Expansion into nascent markets and the creation of cost-effective, accessible LDTs for underserved populations will also be pivotal for future market proliferation.
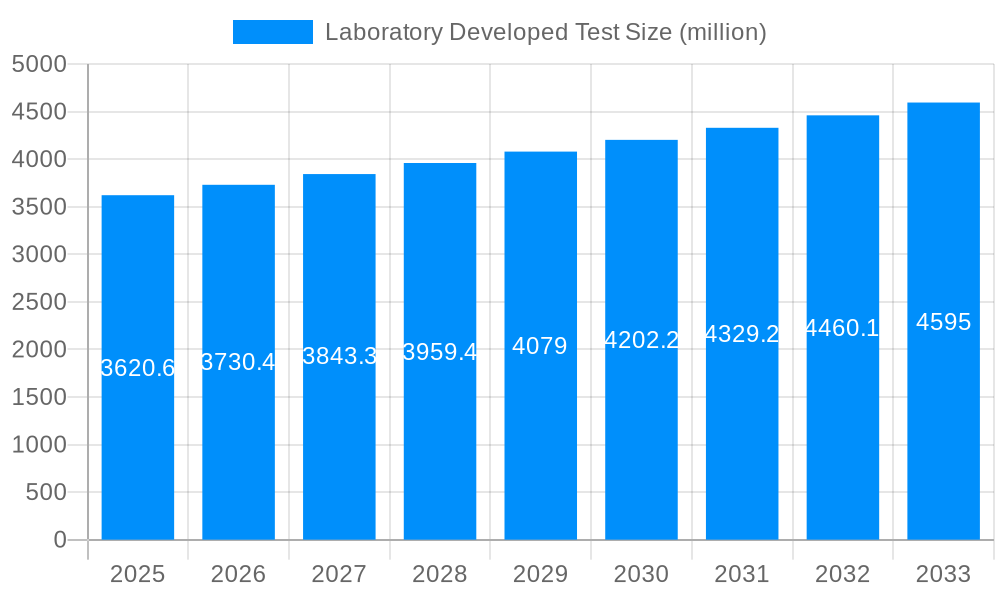

The Laboratory Developed Test (LDT) market exhibited robust growth during the historical period (2019-2024), exceeding USD 100 million in estimated revenue for 2025. This surge is primarily attributed to the increasing prevalence of chronic diseases, the rising demand for personalized medicine, and advancements in molecular diagnostic technologies. The market is characterized by a dynamic interplay of established players like Quest Diagnostics and Roche, alongside emerging companies specializing in niche areas like genomic testing (23andMe, Illumina). The forecast period (2025-2033) projects continued expansion, driven by factors such as the growing adoption of next-generation sequencing (NGS) technologies and the increasing investment in research and development (R&D) by both large corporations and smaller biotech firms. The market's growth trajectory is significantly influenced by regulatory changes and reimbursement policies, creating both opportunities and challenges for LDT providers. The increasing demand for faster, more accurate, and cost-effective diagnostic solutions is fostering innovation within the sector, leading to the development of sophisticated assays for a broader range of diseases. This trend towards personalized medicine, where LDTs play a crucial role in tailoring treatment strategies to individual patients, is a significant driver of market growth. The competitive landscape is intense, with companies constantly striving to differentiate their offerings through technological advancements and strategic partnerships. Data from the study period (2019-2033) reveals a consistent upward trend, with projected annual growth rates exceeding 5% throughout the forecast period, signifying a significant and sustained market opportunity.
Several factors are propelling the growth of the LDT market. Firstly, the increasing prevalence of chronic diseases globally, such as cancer, cardiovascular diseases, and diabetes, necessitates improved diagnostic capabilities. LDTs offer a tailored approach to diagnosis, enabling early detection and personalized treatment strategies. Secondly, the rise of personalized medicine is a major catalyst. LDTs enable physicians to select the most effective therapies based on an individual patient's genetic makeup and disease characteristics, leading to improved treatment outcomes and reduced healthcare costs in the long run. Advancements in molecular diagnostic technologies, including NGS and microarrays, have significantly enhanced the accuracy, speed, and efficiency of LDTs. This technological progress enables the development of more sophisticated assays for a wider range of diseases, driving market expansion. Furthermore, the increasing demand for point-of-care diagnostics, enabling rapid diagnosis in diverse settings, contributes to market growth. The growing adoption of telehealth and remote patient monitoring further fuels the demand for convenient and accessible diagnostic solutions, propelling the growth of LDTs. Finally, increasing investments in R&D by both large corporations and smaller biotech companies fuel innovation and the development of new and improved LDTs.
Despite the promising growth trajectory, the LDT market faces several challenges. Regulatory complexities and variations in reimbursement policies across different regions create significant hurdles for LDT providers. The lengthy and expensive regulatory approval processes, coupled with uncertainty regarding reimbursement rates, can impede market entry and expansion. The high cost of developing and validating new LDTs poses a significant financial barrier, particularly for smaller companies. The need for specialized equipment and expertise to perform LDTs can limit accessibility for smaller labs and clinics. Furthermore, ensuring the quality and accuracy of LDTs is crucial to maintain patient safety and confidence. Stringent quality control measures and rigorous validation processes are essential but add to the overall cost and complexity. The potential for variability in LDT performance across different laboratories is another concern. Establishing standardized protocols and best practices is critical to ensure consistent results and minimize inter-laboratory variations. Finally, the increasing competition among LDT providers intensifies the pressure on pricing and profitability, demanding continuous innovation and efficiency improvements.
The combined factors of technological advancements, increased prevalence of chronic diseases, and the rising demand for personalized medicine positions the LDT market for considerable expansion. The ongoing development and improvement of LDTs, alongside increasing investments in research, will continue to shape the growth of this dynamic market segment.
The LDT industry is experiencing rapid growth propelled by several key factors. The increasing prevalence of chronic diseases necessitates more accurate and personalized diagnostics. Simultaneously, the growing adoption of personalized medicine requires tailored diagnostic solutions like LDTs. Advancements in technologies such as NGS and microarrays have improved the accuracy, speed, and affordability of these tests, making them more accessible. Government initiatives and increased funding for research and development further accelerate the innovation and growth within this crucial sector.
This report provides a comprehensive overview of the LDT market, covering trends, driving forces, challenges, key players, and significant developments. The detailed analysis provides valuable insights into the market's dynamics and future prospects, enabling informed decision-making for stakeholders in the healthcare industry. The forecast period projections are based on rigorous data analysis and expert assessments, offering a reliable outlook for the growth and evolution of the LDT market.
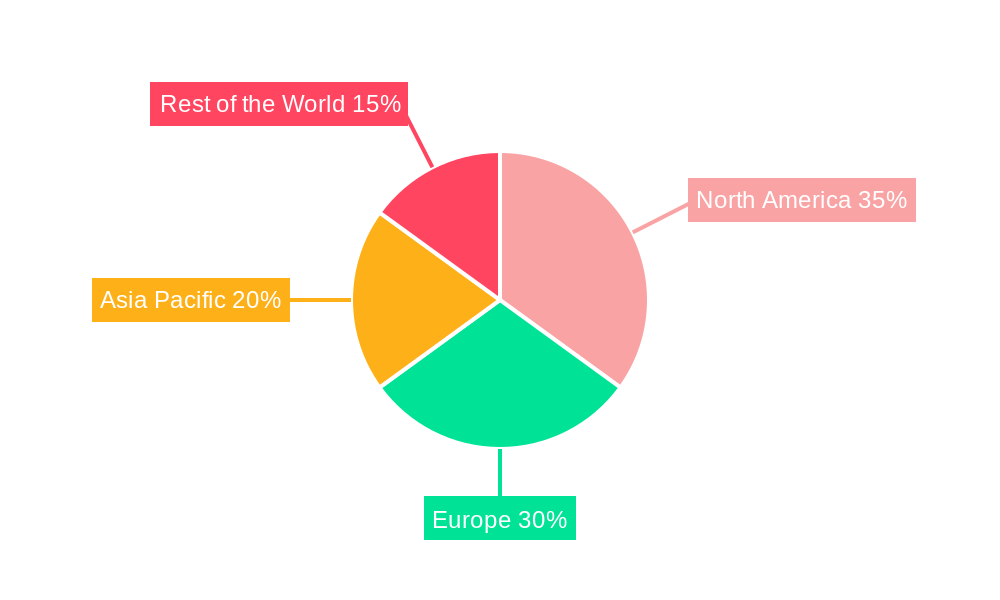

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.09% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.09%.
Key companies in the market include Quest Diagnostics, Thermo Fisher, Waters, Roche, Illumina, Qiagen, 23andMe, Eurofins, Guardant Health, Biotheranostics, Adaptive Biotechnologies, Rosetta Genomics, Biodesix, Helix.
The market segments include Type, Application.
The market size is estimated to be USD 10.35 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in billion.
Yes, the market keyword associated with the report is "Laboratory Developed Test," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Laboratory Developed Test, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.