1. What is the projected Compound Annual Growth Rate (CAGR) of the Clinical Trial Laboratory Testing?
The projected CAGR is approximately 5.09%.
 Clinical Trial Laboratory Testing
Clinical Trial Laboratory TestingClinical Trial Laboratory Testing by Type (Central Laboratory Services, Clinical Bioanalysis Services, Genomics and Biomarkers, Other Services), by Application (Pharmaceuticals, Biotechnology, Medical Devices, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
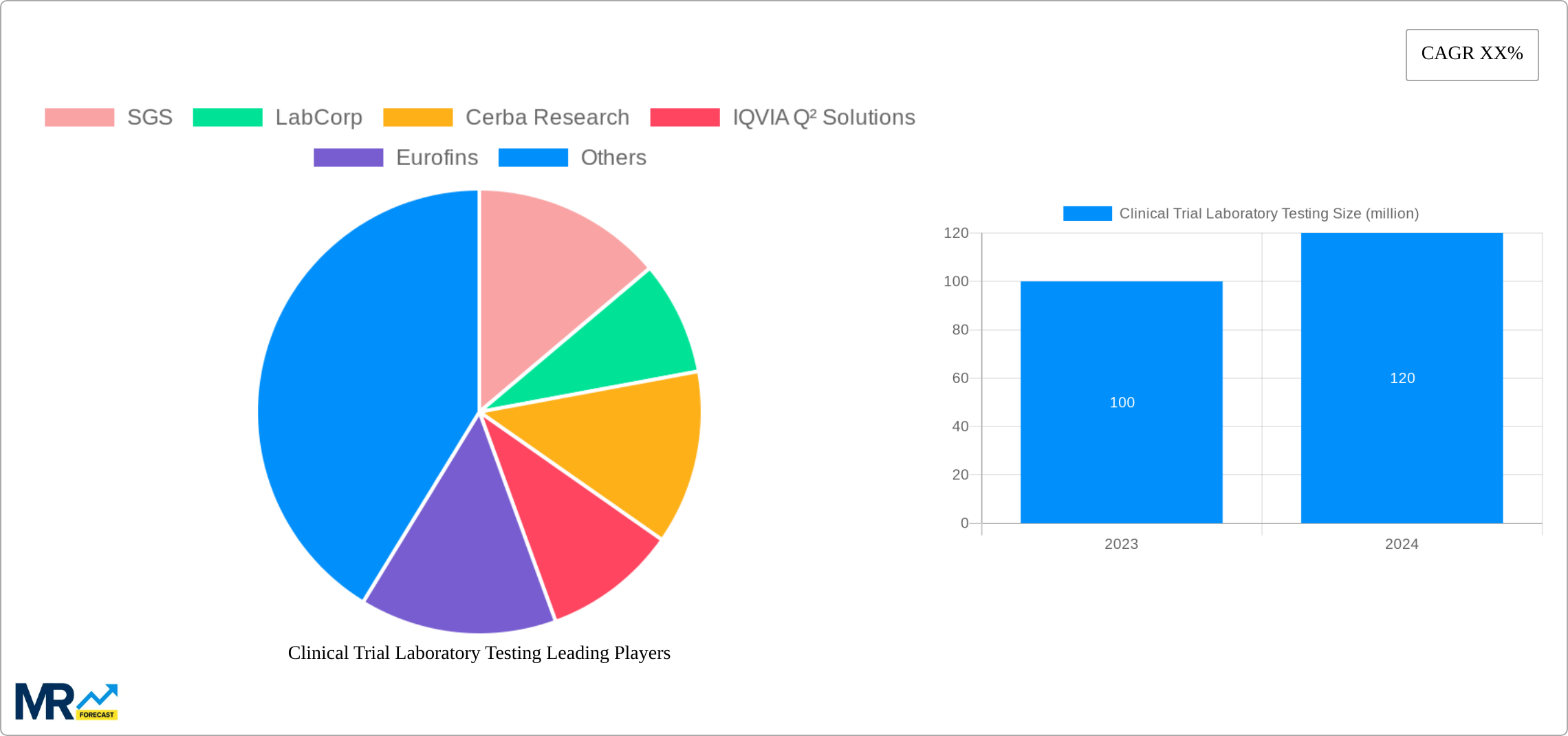
The global clinical trial laboratory testing market is set for substantial expansion, projected to grow from USD 10.35 billion in the base year 2025 to an estimated USD 73.1 billion by 2033. This growth is fueled by a compound annual growth rate (CAGR) of 5.09%. Key drivers include escalating demand for clinical trials, the rising incidence of chronic diseases, and continuous technological innovations in diagnostic capabilities.

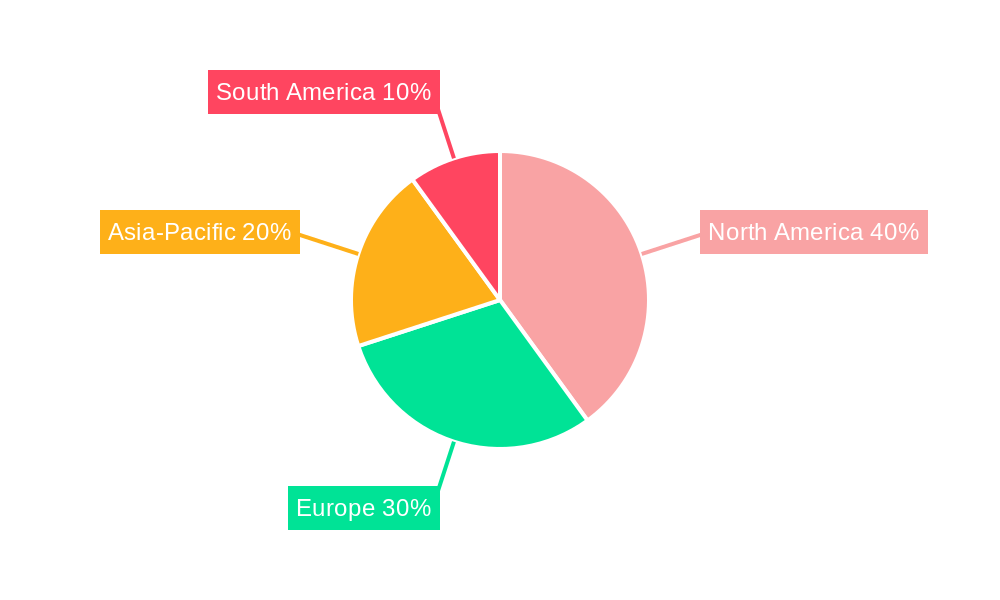
Market segmentation identifies central laboratory services, clinical bioanalysis services, genomics and biomarkers, and other specialized services as key segments. Central laboratory services are anticipated to lead the market, driven by increasing reliance on centralized testing for clinical trial efficiency. In terms of applications, pharmaceuticals, biotechnology, medical devices, and other sectors are recognized. The pharmaceutical sector is expected to dominate, reflecting the high volume of clinical trials within this industry. Geographically, North America is poised to maintain a leading market share, owing to its robust presence of pharmaceutical and biotechnology enterprises.
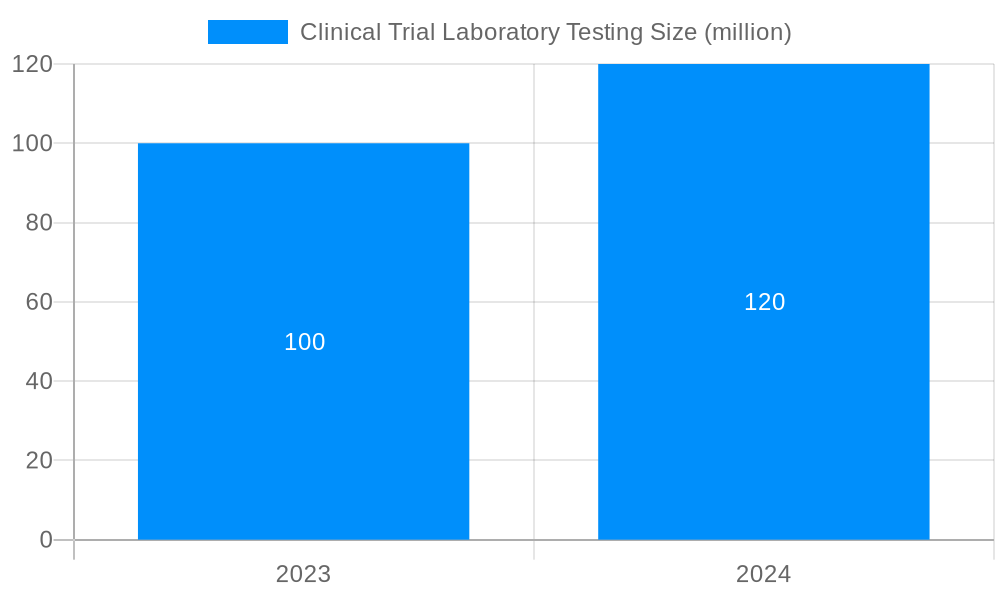

The global clinical trial laboratory testing market is projected to reach $37.6 billion by 2026, growing at a CAGR of 9.0% from 2021 to 2026. The growth of the market is attributed to the increasing number of clinical trials, the rising prevalence of chronic diseases, and the growing adoption of personalized medicine.
The growth of the clinical trial laboratory testing market is also being driven by the increasing demand for outsourcing of clinical trial laboratory services by pharmaceutical and biotechnology companies. Outsourcing clinical trial laboratory services helps companies to reduce costs, improve efficiency, and focus on their core competencies.
The North America region is expected to dominate the clinical trial laboratory testing market, followed by the Europe and Asia-Pacific regions. The growth of the North America market is attributed to the high prevalence of chronic diseases, the strong presence of pharmaceutical and biotechnology companies, and the high adoption of outsourcing of clinical trial laboratory services.
The Central Laboratory Services segment is expected to account for the largest share of the clinical trial laboratory testing market, followed by the Clinical Bioanalysis Services and Genomics and Biomarkers segments. The growth of the Central Laboratory Services segment is attributed to the increasing demand for centralized laboratory services by pharmaceutical and biotechnology companies.
This report provides a comprehensive overview of the clinical trial laboratory testing market. The report includes detailed market insights, forecasts, and analysis of the key trends, drivers, and challenges in the market. The report also provides a competitive analysis of the leading players in the market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.09% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.09%.
Key companies in the market include SGS, LabCorp, Cerba Research, IQVIA Q² Solutions, Eurofins, Charles River, ACM Global Laboratories, Australian Clinical Labs, PPD, Almac Group, Mayo Clinic, LabConnect, Pharmaron, Frontage, WuXi AppTec, CTI, KingMed, CIRS.
The market segments include Type, Application.
The market size is estimated to be USD 10.35 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion.
Yes, the market keyword associated with the report is "Clinical Trial Laboratory Testing," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Clinical Trial Laboratory Testing, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.