1. What is the projected Compound Annual Growth Rate (CAGR) of the WFI Production System?
The projected CAGR is approximately XX%.
 WFI Production System
WFI Production SystemWFI Production System by Type (Distillation System, Membrane System, World WFI Production System Production ), by Application (Pharmaceutical, Biotechnology, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
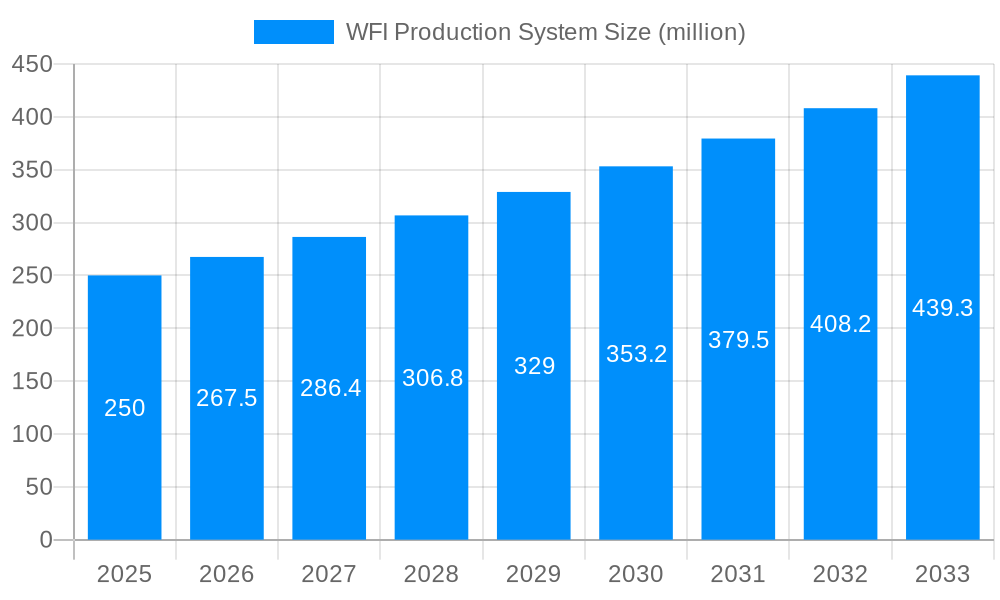
The global Water for Injection (WFI) Production System market is poised for significant expansion, estimated at USD 250 million in 2025, with a projected Compound Annual Growth Rate (CAGR) of approximately 7.5% over the forecast period from 2025 to 2033. This robust growth is primarily driven by the escalating demand for high-purity water in critical applications within the pharmaceutical and biotechnology sectors. The stringent regulatory requirements for sterile and pyrogen-free water in drug manufacturing, coupled with the continuous innovation in drug development and biologics production, are creating a sustained need for advanced WFI systems. Advancements in distillation and membrane technologies are enhancing efficiency, reliability, and cost-effectiveness, further bolstering market adoption. The increasing focus on process validation and quality control in the healthcare industry reinforces the importance of dependable WFI generation and distribution systems, positioning this market for substantial future growth.

Emerging trends such as the adoption of advanced purification techniques, including electro-deionization (EDI) and improved reverse osmosis (RO) systems, are contributing to the market's dynamism. Furthermore, the growing trend towards modular and scalable WFI solutions, designed to meet the evolving needs of both large pharmaceutical corporations and smaller biotech startups, is shaping market strategies. While the market is largely driven by the pharmaceutical and biotechnology applications, the "Other" segment, which may encompass research institutions and specialized medical device manufacturers, is also anticipated to witness steady growth. Geographical expansion in Asia Pacific, particularly in China and India, driven by their burgeoning pharmaceutical industries and increasing healthcare investments, represents a key growth frontier. Restraints, such as the high initial capital investment for sophisticated WFI systems and the complexity of regulatory compliance, are being addressed through technological innovations and the availability of customized solutions.

Here's a unique report description for the WFI Production System, incorporating your specified elements:
The global Water for Injection (WFI) Production System market is poised for significant expansion, driven by the escalating demand for high-purity water across critical industries. Over the study period of 2019-2033, with a base and estimated year of 2025, and a forecast period from 2025-2033, the market has witnessed robust growth, particularly in the historical period of 2019-2024. The Pharmaceutical and Biotechnology sectors are emerging as the primary consumers, their stringent quality requirements necessitating advanced WFI production technologies. Distillation systems, a traditional yet highly reliable method, continue to hold a substantial market share, valued at an estimated $1,250 million in 2025, due to their proven efficacy in producing ultra-pure water. However, membrane systems, including reverse osmosis (RO) and electro-deionization (EDI), are experiencing a surge in adoption, projected to reach a market value of $980 million in 2025. This shift is attributed to their energy efficiency, smaller footprint, and ability to achieve comparable purity levels, aligning with industry trends towards sustainability and operational cost reduction. The "World WFI Production System Production" segment, representing the overall market output, is anticipated to grow from an estimated $2,500 million in 2025 to over $4,000 million by 2033, demonstrating a compound annual growth rate (CAGR) of approximately 6.5%. This growth trajectory is further fueled by the increasing global prevalence of chronic diseases, requiring extensive drug manufacturing and consequently, a greater need for pharmaceutical-grade WFI. Innovations in system design, such as enhanced pre-treatment modules and advanced monitoring capabilities, are also contributing to market dynamism. The "Other" application segment, encompassing research institutions and medical device sterilization, is also exhibiting steady growth, though at a more modest pace. Overall, the WFI Production System market is characterized by a convergence of technological advancement, stringent regulatory frameworks, and expanding end-user industries, creating a fertile ground for sustained market expansion.
The WFI Production System market is propelled by a confluence of potent factors, predominantly stemming from the ever-increasing stringency of global pharmaceutical and biotechnology regulatory standards. Agencies worldwide, including the FDA and EMA, mandate the highest levels of water purity for parenteral drug manufacturing, making reliable and compliant WFI production systems indispensable. This regulatory imperative directly translates into sustained investment in advanced WFI technologies. Furthermore, the burgeoning global pharmaceutical and biotechnology industries, driven by an aging population, rising chronic disease prevalence, and accelerated drug discovery and development, are experiencing unprecedented growth. This surge in biopharmaceutical production inherently escalates the demand for WFI. The estimated market size for WFI production systems in 2025 is projected to be around $2,500 million, and this figure is expected to climb significantly due to these foundational growth drivers. The increasing outsourcing of pharmaceutical manufacturing also plays a crucial role, as contract manufacturing organizations (CMOs) require robust WFI capabilities to serve their diverse client base. Finally, technological advancements are continually improving the efficiency, reliability, and cost-effectiveness of WFI production, making these systems more accessible and attractive to a wider range of stakeholders.
Despite the robust growth prospects, the WFI Production System market faces several significant challenges and restraints. One of the primary hurdles is the substantial initial capital investment required for advanced WFI production systems, particularly for distillation-based technologies which can be energy-intensive. This can be a deterrent for smaller pharmaceutical manufacturers or emerging biotechnology firms with limited financial resources, with the capital expenditure for a high-capacity system often running into several million dollars. Moreover, the complex regulatory landscape and the need for continuous compliance with evolving standards necessitate ongoing investment in validation, monitoring, and maintenance, adding to the operational costs. The stringent validation processes for WFI systems can also lead to extended project timelines, delaying the deployment of new manufacturing facilities. Another restraint is the operational complexity and the requirement for highly skilled personnel to operate and maintain these sophisticated systems, ensuring consistent water quality and preventing contamination. Potential supply chain disruptions for critical components and raw materials can also impact production schedules and increase costs. Finally, the energy consumption associated with some traditional WFI production methods, like multi-effect distillation, presents an ongoing challenge in the face of rising energy prices and increasing pressure for environmental sustainability, despite the adoption of more energy-efficient membrane systems.
The Pharmaceutical and Biotechnology application segments are undeniably set to dominate the WFI Production System market. Their dominance is underpinned by the non-negotiable requirement for exceptionally high-purity water in the manufacturing of injectable drugs, vaccines, and other sterile pharmaceutical products. In 2025, the Pharmaceutical segment is estimated to account for a market share exceeding $1,800 million, while the Biotechnology segment is projected to contribute around $750 million. This is due to the inherent nature of these industries, where water quality directly impacts patient safety and drug efficacy. Any compromise in WFI purity can lead to catastrophic consequences, including patient harm and regulatory sanctions, thus ensuring a perpetual demand for state-of-the-art WFI production solutions.
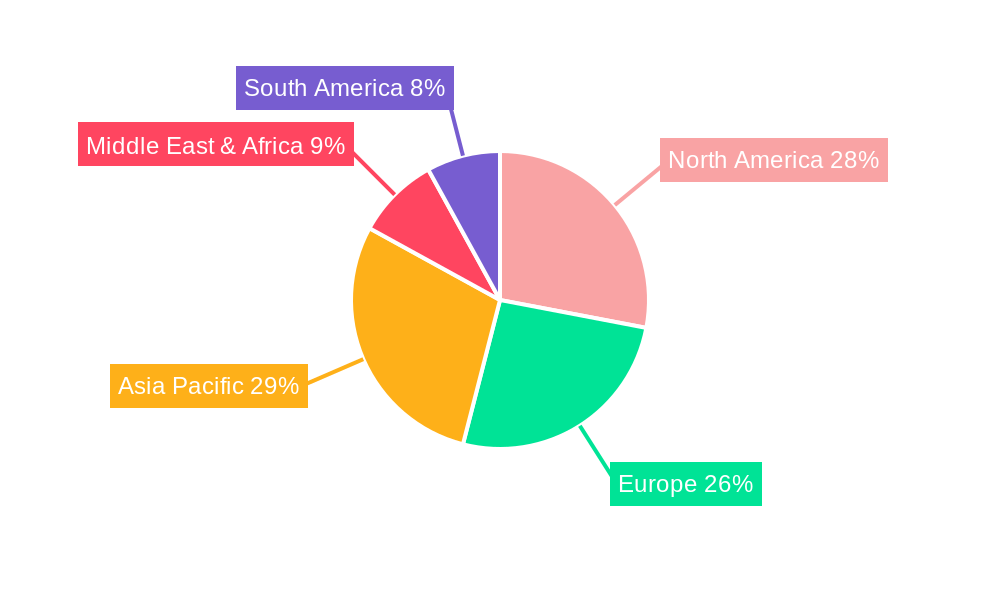
Regions exhibiting a strong presence of pharmaceutical and biotechnology manufacturing hubs are consequently poised to lead the market. North America, particularly the United States, and Europe, including Germany, Switzerland, and the United Kingdom, are at the forefront. The presence of major pharmaceutical giants, a robust research and development ecosystem, and stringent regulatory oversight by bodies like the FDA and EMA collectively drive significant demand. In 2025, North America alone is projected to represent over 35% of the global WFI Production System market value, with Europe following closely.
The Distillation System type segment, though facing competition from membrane technologies, is expected to maintain a substantial market share, valued at approximately $1,250 million in 2025. This is due to its long-standing reputation for reliability and its ability to achieve the highest purity standards, often mandated for certain critical applications where redundancy and absolute assurance are paramount. However, the Membrane System segment is exhibiting the most dynamic growth, with a projected market value of $980 million in 2025, and a higher CAGR forecast. This growth is fueled by their energy efficiency, lower operational costs, and compact design, making them increasingly attractive, especially for newer facilities and expansions where sustainability and space optimization are key considerations. The global WFI Production System Production, encompassing the entire market, is estimated to reach $2,500 million in 2025 and is projected to expand to over $4,000 million by 2033.
The WFI Production System industry is experiencing significant growth catalysts that are shaping its trajectory. A primary driver is the relentless pursuit of enhanced drug safety and efficacy, demanding ever-higher standards of water purity. This pushes innovation in WFI technologies. The expanding global pharmaceutical and biotechnology sectors, fueled by an aging population and rising healthcare expenditure, directly translate into increased demand for WFI. Furthermore, technological advancements leading to more energy-efficient and cost-effective WFI production methods, such as improved membrane technologies and advanced distillation techniques, are broadening market accessibility.
This comprehensive report provides an in-depth analysis of the WFI Production System market, covering the period from 2019 to 2033, with a base year of 2025. It delves into the intricate dynamics of the global market, dissecting trends, drivers, challenges, and opportunities. The report offers detailed insights into the market segmentation by type (Distillation Systems, Membrane Systems), application (Pharmaceutical, Biotechnology, Other), and geographical regions. Valued at an estimated $2,500 million in 2025, the market is projected to witness robust growth, exceeding $4,000 million by 2033. The report further highlights the strategic initiatives and innovations of leading players like Stilmas, BWT, MECO, STERIS, and Veolia Water Technologies, alongside significant market developments that are shaping the industry's future.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Stilmas, BWT, MECO, STERIS, Veolia Water Technologies, BRAM-COR, Syntegon, Aqua-Chem, Evoqua Water Technologies, Truking Technology, Shenzhen Carryclean, Shanghai GenTech, Puretech Process Systems, Fabtech Technologies, Organo, NGK Filtech, TSA Process Equipments, Nihon Rosuiki Kogyo, Nomura Micro Science, Haedong Technology, JUNGHYUN PLANT.
The market segments include Type, Application.
The market size is estimated to be USD 250 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "WFI Production System," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the WFI Production System, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.