1. What is the projected Compound Annual Growth Rate (CAGR) of the Viral Clearance Service?
The projected CAGR is approximately 21.4%.
 Viral Clearance Service
Viral Clearance ServiceViral Clearance Service by Type (Basic Service, Enhanced Service, Full Service, Turnkey Service), by Application (Research Institution, Pharmaceutical Industry, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
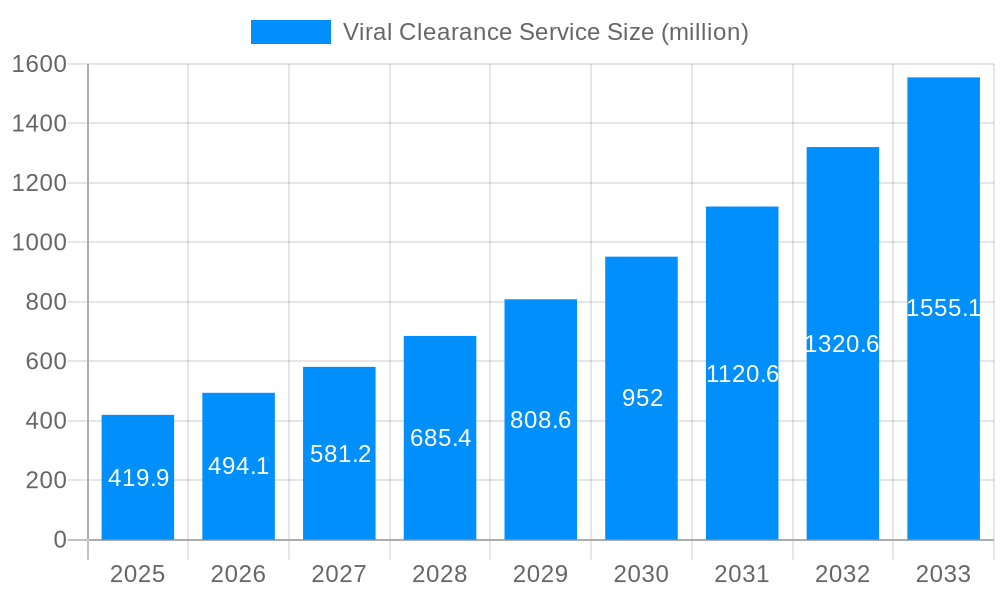
The global viral clearance service market is experiencing robust growth, projected to reach an estimated USD 419.9 million by 2025, expanding at an impressive Compound Annual Growth Rate (CAGR) of 17.7%. This significant expansion is primarily fueled by the escalating demand for biopharmaceuticals, the increasing stringency of regulatory requirements for drug safety, and the continuous rise in investments in research and development activities within the pharmaceutical and biotechnology sectors. As novel therapies, including gene and cell therapies, gain traction, the need for rigorous viral clearance processes becomes paramount to ensure patient safety and product efficacy. The market is segmented into various service types, including Basic Service, Enhanced Service, Full Service, and Turnkey Service, catering to diverse client needs across research institutions and the pharmaceutical industry. These services are critical for mitigating viral contamination risks throughout the drug development and manufacturing lifecycle, thereby underpinning the market's upward trajectory.
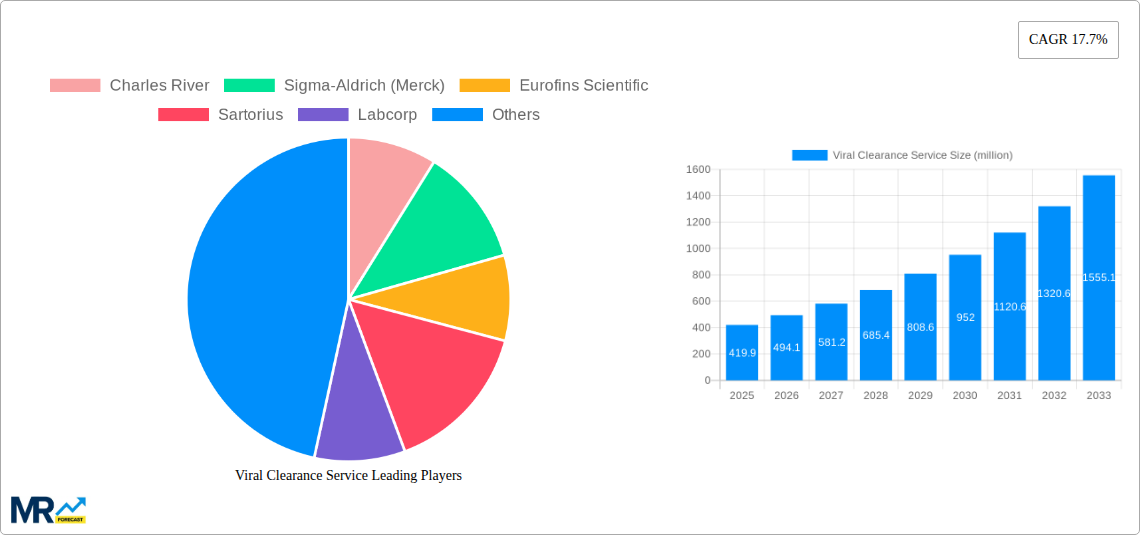

The market dynamics are further shaped by emerging trends such as the integration of advanced analytical techniques and automation in viral clearance studies, leading to increased efficiency and accuracy. The growing outsourcing of these specialized services by smaller biotech firms and research institutions to contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) is also a significant driver. Key players like Charles River, Sigma-Aldrich (Merck), Eurofins Scientific, and Labcorp are actively investing in expanding their service portfolios and geographical reach to capitalize on this burgeoning market. Despite the optimistic outlook, potential restraints include the high cost associated with specialized equipment and expertise, and the time-intensive nature of validation processes for novel clearance methods. Nevertheless, the overarching imperative for drug safety and the continuous innovation in biotherapeutics are expected to sustain the strong growth momentum of the viral clearance service market.

Here's a report description on Viral Clearance Services, incorporating your specifications:
This report provides an in-depth analysis of the global viral clearance service market, offering critical insights into its historical performance, current trends, and future projections. Spanning a study period from 2019 to 2033, with a base year of 2025 and a forecast period of 2025-2033, this research delves into the intricate dynamics shaping this vital sector. The historical period (2019-2024) has laid the groundwork for understanding the evolutionary path of viral clearance services, highlighting key milestones and initial market responses. The estimated year of 2025 serves as a pivotal point for current market evaluation, from which future growth trajectories are meticulously mapped. With a projected market valuation reaching into the millions of USD, this report aims to equip stakeholders with actionable intelligence for strategic decision-making.
The viral clearance service market is experiencing a profound transformation, driven by an escalating demand for biopharmaceutical safety and an increasingly stringent regulatory landscape. The global market for viral clearance services has witnessed a consistent upward trajectory, fueled by the burgeoning biopharmaceutical industry and the growing recognition of its critical role in ensuring the safety and efficacy of therapeutic products. The historical period (2019-2024) saw significant growth, as companies increasingly outsourced these specialized services to experts, leveraging their advanced technologies and regulatory expertise. The base year of 2025 marks a point of continued expansion, with expectations of sustained momentum through the forecast period (2025-2033). Key market insights reveal a growing emphasis on advanced, multi-orthogonal clearance methods, moving beyond traditional single-step approaches. The adoption of innovative technologies, such as single-use systems and continuous manufacturing, is also influencing the demand for tailored viral clearance strategies. Furthermore, the increasing complexity of biologics and gene therapies necessitates highly specialized clearance solutions, driving the market towards niche and customized service offerings. The rise of personalized medicine and the development of novel therapeutic modalities are further amplifying the need for robust and adaptable viral clearance protocols. Regulatory bodies worldwide are continuously refining guidelines, demanding higher levels of assurance and transparency, which in turn propels the market towards more sophisticated and validated clearance techniques. The strategic collaborations between service providers and biopharmaceutical companies are also becoming more prevalent, fostering innovation and accelerating the development of next-generation viral clearance technologies. As the biopharmaceutical pipeline expands with complex modalities, the market for viral clearance services is poised for significant and sustained growth, underpinning the safety and accessibility of life-saving treatments.
The viral clearance service market is propelled by a confluence of powerful drivers, paramount among them being the unwavering commitment to patient safety. The increasing complexity and genetic engineering involved in modern biotherapeutics, including monoclonal antibodies, gene therapies, and cell therapies, inherently introduce potential risks of viral contamination. Consequently, robust viral clearance strategies are no longer an option but a critical necessity to mitigate these risks and ensure the production of safe and effective treatments. This imperative is amplified by stringent global regulatory requirements. Agencies like the FDA, EMA, and others mandate comprehensive viral safety assessments and validated clearance processes for all biologics entering the market. Non-compliance can lead to significant delays, costly recalls, and reputational damage, thereby incentivizing robust viral clearance efforts. The exponential growth of the biopharmaceutical industry itself is another major catalyst. As more novel biologics progress through clinical trials and towards commercialization, the demand for expert viral clearance services escalates. This growth is particularly pronounced in the areas of personalized medicine and orphan drugs, where even small-scale production requires meticulous viral safety measures. Furthermore, the increasing trend of outsourcing by pharmaceutical and biotechnology companies, seeking to leverage specialized expertise and cost-efficiencies, fuels the demand for dedicated viral clearance service providers. The continuous innovation in bioprocessing technologies, such as advanced filtration methods and inactivation techniques, also necessitates expert validation and application, driving the market forward.
Despite its robust growth, the viral clearance service market faces several significant challenges and restraints. One of the primary hurdles is the inherent complexity and variability of biological products. Each therapeutic molecule and manufacturing process presents unique viral clearance challenges, requiring highly customized and validated approaches. Developing and validating these specific clearance strategies can be time-consuming and resource-intensive. Furthermore, the evolving regulatory landscape, while a driver, can also be a restraint. Rapidly changing guidelines and interpretations from regulatory bodies can necessitate significant revalidation efforts and investments, adding to the cost and timeline of product development. The high cost associated with advanced viral clearance studies and the specialized equipment required can also be a barrier, particularly for smaller biotechnology companies and research institutions with limited budgets. The availability of skilled personnel is another critical factor. Operating sophisticated viral clearance studies requires highly trained scientists with specialized knowledge in virology, molecular biology, and regulatory affairs. A shortage of such expertise can limit the capacity and scalability of service providers. The potential for false positives or negatives in viral detection and clearance assays, although minimized through rigorous validation, remains a concern that necessitates careful interpretation of results. Finally, the increasing demand for faster turnaround times in the competitive biopharmaceutical market can clash with the often lengthy validation and execution phases of viral clearance studies.
The Pharmaceutical Industry segment is poised to dominate the viral clearance service market, driven by its intrinsic need for rigorous product safety and regulatory compliance. Within this segment, the development and manufacturing of biologics, including monoclonal antibodies, vaccines, gene therapies, and cell therapies, are the primary consumers of viral clearance services. The sheer volume of these complex biotherapeutics entering the global pipeline, coupled with the stringent regulatory hurdles they must overcome before reaching patients, makes the pharmaceutical industry the largest and most critical market for these specialized services. The increasing investment in R&D for novel therapies, particularly in the oncology, immunology, and infectious disease areas, further amplifies this demand. Pharmaceutical companies recognize that investing in comprehensive viral clearance studies is paramount to de-risking their product development pipelines and ensuring market access. They often collaborate with specialized service providers to conduct these studies, leveraging external expertise and state-of-the-art facilities.
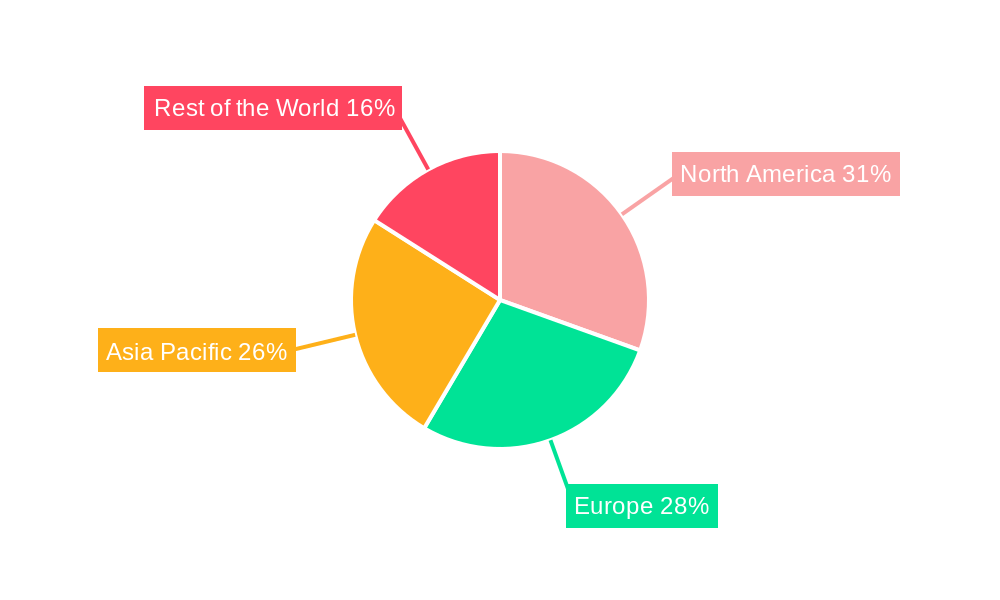
North America is projected to emerge as a dominant region in the viral clearance service market. This dominance is attributed to several synergistic factors. Firstly, North America, particularly the United States, is a global hub for biopharmaceutical innovation and manufacturing. The presence of a vast number of leading pharmaceutical and biotechnology companies, coupled with a robust ecosystem of academic research institutions and venture capital funding, fuels a continuous stream of novel drug development, all of which require stringent viral clearance protocols. The region boasts a highly developed regulatory framework and a proactive stance by agencies like the FDA, which consistently sets high standards for viral safety, compelling companies to invest heavily in these services. Secondly, North America has a well-established and advanced network of specialized viral clearance service providers, equipped with cutting-edge technologies and highly skilled personnel. This concentration of expertise and infrastructure facilitates efficient and comprehensive service delivery.
The Full Service segment is expected to experience substantial growth and command a significant market share within the broader viral clearance service landscape. This segment encompasses a comprehensive suite of services, ranging from initial risk assessment and process development to validation studies, spiked and unspiked sample testing, and the generation of comprehensive regulatory documentation. Pharmaceutical and biotechnology companies often prefer Full Service providers due to the convenience and efficiency of a single-source solution for their complex viral clearance needs. This integrated approach minimizes the need for managing multiple vendors, streamlines communication, and ensures a cohesive strategy throughout the product development lifecycle. The ability of Full Service providers to offer end-to-end support, from early-stage research to late-stage manufacturing validation, is particularly attractive for companies navigating the intricate and time-consuming process of bringing biotherapeutics to market. Furthermore, as regulatory expectations continue to rise, the demand for robust, well-documented, and expertly executed viral clearance studies provided by Full Service entities is expected to surge, solidifying its leading position in the market.
The viral clearance service industry is experiencing robust growth, propelled by several key catalysts. The increasing complexity of biopharmaceutical products, including advanced gene and cell therapies, inherently raises the stakes for viral safety, demanding sophisticated clearance solutions. Furthermore, evolving and increasingly stringent regulatory guidelines from global health authorities necessitate comprehensive and validated viral clearance studies, driving demand. The burgeoning biotechnology sector and the expanding biologics pipeline, particularly in emerging economies, are also significant growth engines. Moreover, the growing trend of outsourcing by pharmaceutical companies to specialized service providers, seeking cost-efficiency and access to advanced expertise, further fuels market expansion.
This comprehensive report delves deep into the viral clearance service market, providing an exhaustive analysis of its landscape from 2019 to 2033. It meticulously dissects market trends, identifies key driving forces such as escalating patient safety demands and stringent regulatory oversight, and examines the challenges and restraints like the complexity of biological products and the high cost of studies. The report highlights the dominance of the Pharmaceutical Industry segment and North America as a leading region, with a particular focus on the growing influence of the Full Service model. Leading players are identified, and significant developments within the sector are chronicled. This in-depth research offers invaluable insights for stakeholders, empowering them to navigate the evolving market and capitalize on future opportunities within this critical segment of biopharmaceutical development, projected to reach millions in valuation.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 21.4% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 21.4%.
Key companies in the market include Charles River, Sigma-Aldrich (Merck), Eurofins Scientific, Sartorius, Labcorp, ViruSure, Texcell, Bioscience Labs, Vironova Biosafety, Mérieux NutriSciences, WuXi AppTec, Syngene, Labor Dr. Merk (Boehringer Ingelheim), Sino Biological.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Viral Clearance Service," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Viral Clearance Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.