1. What is the projected Compound Annual Growth Rate (CAGR) of the Vagus Nerve Stimulation Implantable Device?
The projected CAGR is approximately 8.48%.
 Vagus Nerve Stimulation Implantable Device
Vagus Nerve Stimulation Implantable DeviceVagus Nerve Stimulation Implantable Device by Type (Metallic, Ceramics, Polymeric), by Application (Hospitals, Neurology Clinics, Ambulatory Surgical Centers, Research Centers), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Vagus Nerve Stimulation (VNS) Implantable Device market is experiencing robust growth, projected to reach an estimated \$14.13 billion by 2025, with a Compound Annual Growth Rate (CAGR) of 8.48% from 2019-2033. This significant expansion is driven by a confluence of factors, including the increasing prevalence of neurological disorders such as epilepsy, depression, and anxiety, for which VNS therapy has proven to be a highly effective treatment. The growing awareness and acceptance of VNS as a viable therapeutic option, coupled with advancements in implantable device technology that enhance efficacy and patient comfort, are further fueling market demand. Hospitals and neurology clinics represent the largest application segments, owing to their established infrastructure and expertise in managing complex neurological conditions. The metallic segment, favored for its durability and biocompatibility, is expected to dominate the material types, although innovations in ceramic and polymeric materials are poised to offer more specialized applications and potentially improved patient outcomes.
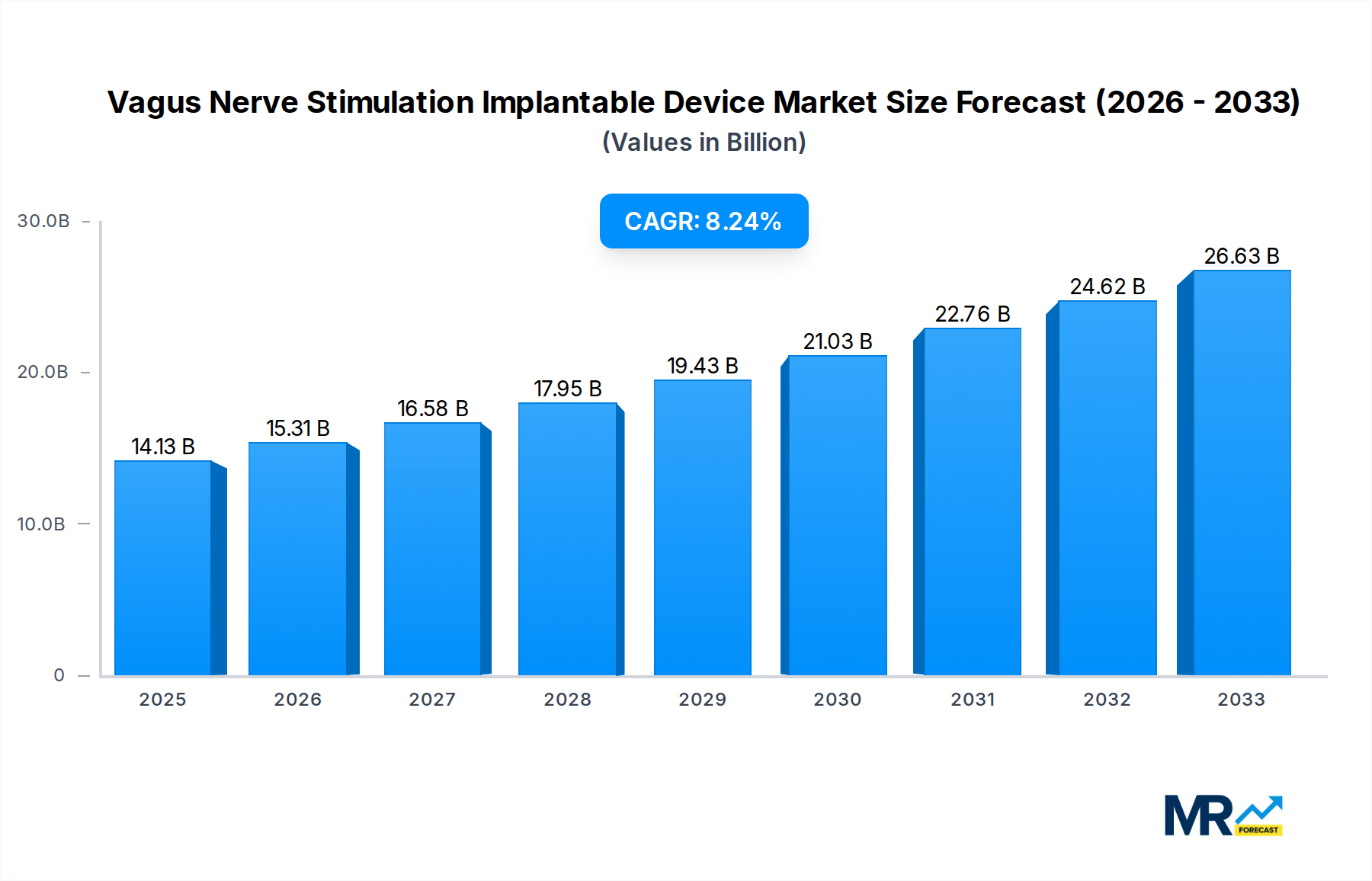

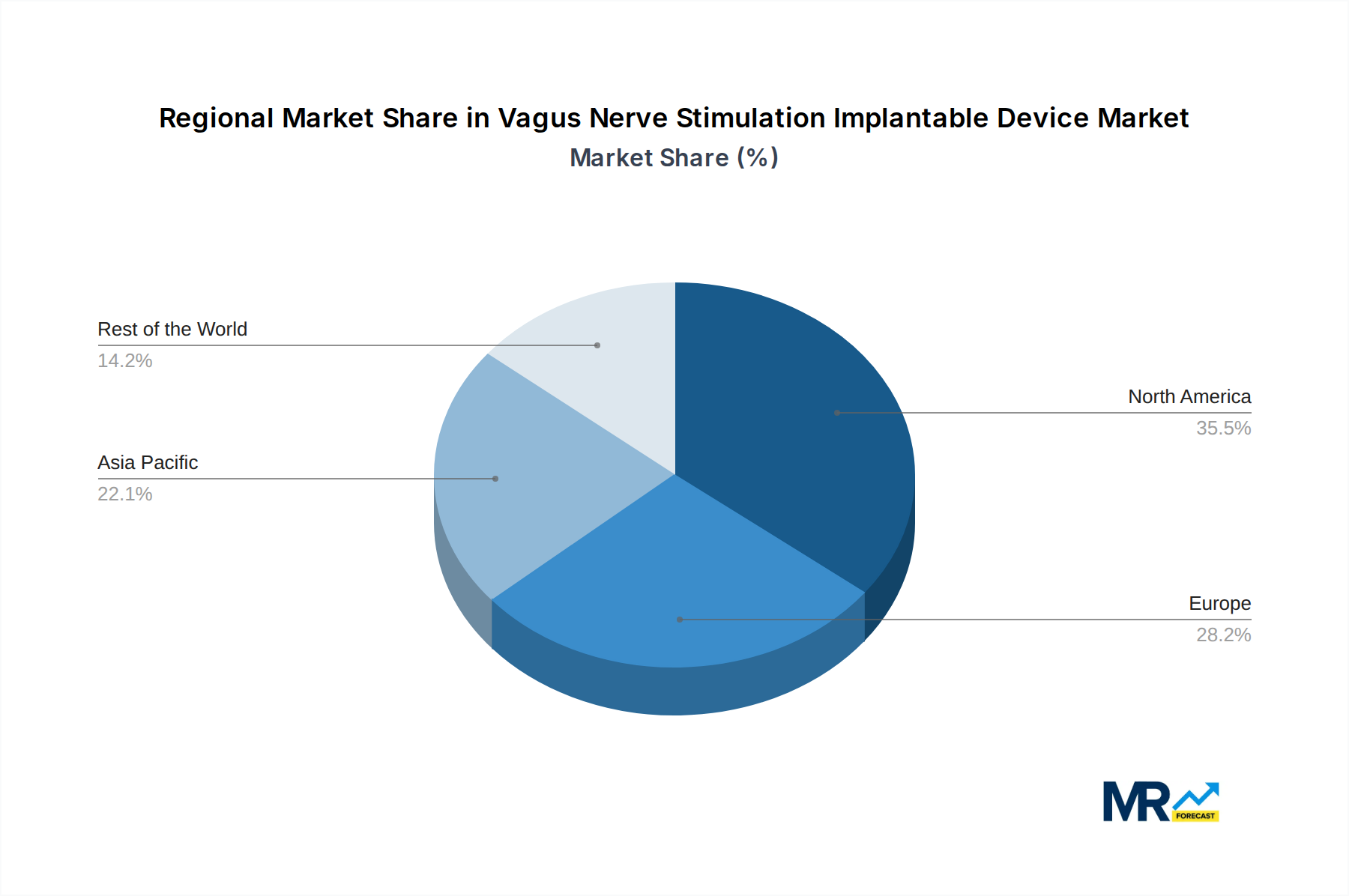
The market's trajectory is also influenced by rising healthcare expenditures and a growing demand for less invasive treatment alternatives to traditional surgical interventions. Leading companies such as Boston Scientific, LivaNova, and NeuroPace are actively investing in research and development, introducing next-generation VNS devices with improved programmability and reduced side effects, thereby expanding the therapeutic potential of this technology. North America currently holds a substantial market share, driven by advanced healthcare infrastructure and a high incidence of neurological disorders. However, the Asia Pacific region is anticipated to witness the fastest growth due to increasing healthcare investments, a burgeoning patient population, and the growing adoption of advanced medical technologies. While the market presents a promising outlook, potential restraints such as the high cost of implantation procedures and a limited number of trained neurosurgeons in certain regions could present challenges. Nevertheless, the overwhelming clinical benefits and continuous technological advancements position the Vagus Nerve Stimulation Implantable Device market for sustained and significant expansion.

The global Vagus Nerve Stimulation (VNS) implantable device market is poised for substantial expansion, projected to reach an estimated $12.5 billion by 2033, a significant leap from its valuation in the historical period. This upward trajectory is underpinned by a confluence of factors, including an increasing prevalence of neurological disorders such as epilepsy, depression, and heart failure, alongside a growing awareness and acceptance of neuromodulation therapies. The study period, spanning from 2019 to 2033, with a base year of 2025, reveals a consistent growth pattern driven by technological advancements and a broadening range of therapeutic applications. The market is moving beyond its traditional role in refractory epilepsy to encompass a wider array of indications, including potential applications in obesity, inflammatory bowel disease, and even stroke rehabilitation. This diversification is a key trend, attracting new entrants and fostering innovation among established players. Furthermore, the ongoing refinement of implantable device technology, focusing on miniaturization, improved battery life, and enhanced programmability, is contributing to better patient outcomes and increased demand. The reimbursement landscape also plays a critical role, with favorable policies in key markets supporting wider adoption. The estimated market size in 2025 is expected to be around $7.2 billion, indicating robust growth in the forecast period of 2025-2033. The historical period from 2019-2024 laid the groundwork, demonstrating initial market penetration and technological development that has paved the way for this projected boom. This evolving market dynamic underscores the increasing importance of VNS therapy as a viable and effective treatment option for a growing patient population.
Several powerful forces are acting as significant propellers for the Vagus Nerve Stimulation (VNS) implantable device market. The most prominent driver is the escalating global burden of neurological and psychiatric disorders. Conditions like drug-resistant epilepsy, major depressive disorder (MDD), and certain cardiovascular ailments are affecting millions, creating a substantial unmet medical need. VNS therapy offers a compelling alternative or adjunct treatment for patients who have not responded adequately to conventional pharmacological interventions, thus expanding the patient pool for these devices. Furthermore, there is a discernible shift in healthcare paradigms towards less invasive and more personalized treatment modalities. Neuromodulation, with VNS at its forefront, aligns perfectly with this trend by offering targeted stimulation that can alleviate symptoms and improve quality of life with fewer systemic side effects compared to many drug-based treatments. Continuous advancements in the underlying technology of VNS devices are also fueling growth. Innovations leading to more sophisticated algorithms, wireless programming capabilities, and longer battery lifespans are making the devices more user-friendly and effective, thereby enhancing patient satisfaction and physician confidence. The growing body of clinical evidence and positive real-world outcomes reported from studies and clinical practices further solidifies the therapeutic value of VNS, encouraging wider adoption by healthcare providers and payers. The projected market value of $12.5 billion by 2033 is a testament to the combined impact of these propelling factors.
Despite the promising growth trajectory, the Vagus Nerve Stimulation (VNS) implantable device market faces several significant challenges and restraints that could temper its expansion. One of the primary hurdles is the relatively high cost associated with VNS implantation and the devices themselves. The initial surgical procedure and the ongoing cost of the implantable generator can be prohibitive for some healthcare systems and patients, particularly in lower-income regions. This cost factor, combined with complex reimbursement policies in certain geographical areas, can limit market penetration. Another significant challenge is the invasive nature of the implantation procedure. While minimally invasive compared to some surgeries, it still requires surgical intervention, which carries inherent risks such as infection, bleeding, and nerve damage. This can be a deterrent for some patients and physicians, especially when less invasive treatment options, albeit potentially less effective, are available. Furthermore, the VNS therapy is not universally effective for all patients, and identifying the ideal candidates for successful treatment can be challenging. The efficacy can vary significantly depending on the underlying condition and individual patient characteristics, leading to patient dissatisfaction if expectations are not met. The learning curve for programming and managing VNS devices can also be steep for some clinicians, requiring specialized training and expertise, which may not be readily available in all healthcare settings. Finally, the ongoing development of alternative and emerging therapies for the conditions treated by VNS, such as deep brain stimulation (DBS) or advanced pharmacological agents, presents a competitive threat that could impact market share.
The Vagus Nerve Stimulation (VNS) implantable device market is anticipated to witness significant dominance from North America, particularly the United States, propelled by a combination of robust healthcare infrastructure, high disposable incomes, and a strong emphasis on adopting advanced medical technologies. The region's well-established reimbursement frameworks for advanced therapies and a proactive approach to innovation contribute significantly to its market leadership. The prevalence of neurological disorders such as epilepsy and depression is also considerably high in North America, further bolstering demand for VNS devices.
Within the segmentation analysis, the Hospitals application segment is expected to emerge as the dominant force in the VNS implantable device market. Hospitals, being comprehensive healthcare institutions, are equipped with the necessary surgical facilities, specialized medical personnel, and diagnostic capabilities required for VNS implantation and ongoing patient management. The complexity of the procedure and the need for multidisciplinary care teams, including neurologists, neurosurgeons, and rehabilitation specialists, naturally steer VNS implantations towards hospital settings. Furthermore, hospitals are often the primary point of contact for patients with severe or refractory neurological conditions, making them ideal hubs for the adoption and utilization of VNS technology. The estimated market share for hospitals is substantial, reflecting their central role in delivering advanced medical interventions.
The Metallic type segment is also projected to hold a dominant position. Metallic components, such as titanium alloys, are crucial for the biocompatibility, durability, and structural integrity of implantable medical devices. These materials are well-tolerated by the human body, reducing the risk of allergic reactions and inflammatory responses, which are critical considerations for long-term implants. The strength and resilience of metallic alloys ensure the longevity and reliable performance of VNS pulse generators and lead wires, minimizing the need for premature device replacement. The well-established manufacturing processes and proven track record of metallic biomaterials in the medical device industry further solidify its dominance.
The Vagus Nerve Stimulation (VNS) implantable device industry is experiencing significant growth catalysts, most notably the expanding indications for VNS therapy beyond traditional epilepsy treatment. The successful application of VNS in managing treatment-resistant depression and heart failure has opened up vast new patient populations and driven increased research into its efficacy for other conditions like obesity, inflammatory bowel disease, and even cognitive disorders. This diversification of applications is a primary growth catalyst, attracting further investment and innovation. Additionally, continuous advancements in device technology, such as miniaturization, improved battery longevity, and enhanced wireless programming capabilities, are making VNS devices more attractive to both patients and clinicians, thereby accelerating market adoption.
The comprehensive coverage of the Vagus Nerve Stimulation (VNS) implantable device report provides an in-depth analysis of the market dynamics, encompassing market size, segmentation, competitive landscape, and future projections. The report meticulously details market trends and forecasts, offering a clear understanding of the industry's growth trajectory from 2019 to 2033, with a strong focus on the forecast period of 2025-2033. It delves into the key driving forces and challenges shaping the market, offering valuable insights for stakeholders. Furthermore, the report thoroughly examines regional dominance and segment-specific growth, providing strategic perspectives for market participants to capitalize on emerging opportunities. The insights presented are crucial for understanding the current state and future potential of VNS therapy.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.48% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 8.48%.
Key companies in the market include LivaNova, ReShape Lifesciences, Boston Scientific, Cyberonics, SenTiva, Inspire Medical, NeuroPace, Stimwave, Cerbomed GmbH, ImThera Medical, EnteroMedics, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Vagus Nerve Stimulation Implantable Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Vagus Nerve Stimulation Implantable Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.