1. What is the projected Compound Annual Growth Rate (CAGR) of the Traumatic Brain Injury Assessment Device?
The projected CAGR is approximately 14.99%.
 Traumatic Brain Injury Assessment Device
Traumatic Brain Injury Assessment DeviceTraumatic Brain Injury Assessment Device by Type (Imaging Device, Monitoring Equipment, World Traumatic Brain Injury Assessment Device Production ), by Application (Hospital, Diagnostic Center, Others, World Traumatic Brain Injury Assessment Device Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
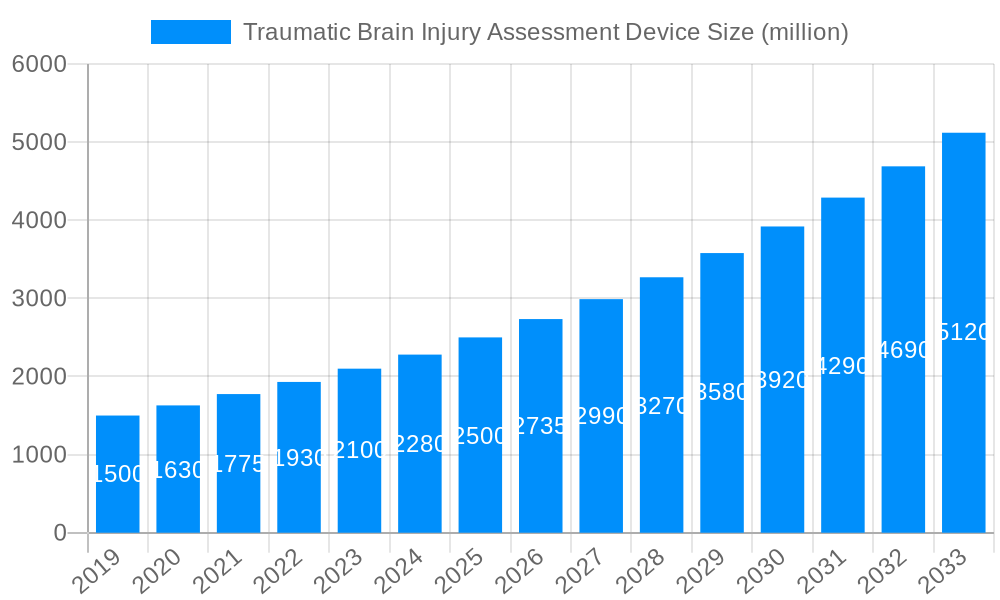
The global Traumatic Brain Injury (TBI) Assessment Device market is poised for significant growth, projected to reach approximately $2,500 million by 2025, with an estimated Compound Annual Growth Rate (CAGR) of 11.5% from 2019-2033. This robust expansion is fueled by a confluence of factors, including the increasing incidence of TBIs due to rising sports-related injuries, road accidents, and military conflicts. The growing awareness among healthcare professionals and the public regarding the critical need for early and accurate TBI diagnosis further propels market demand. Advancements in imaging technologies and the development of portable, non-invasive diagnostic devices are also key drivers, offering improved patient outcomes and reducing the burden on traditional diagnostic methods.
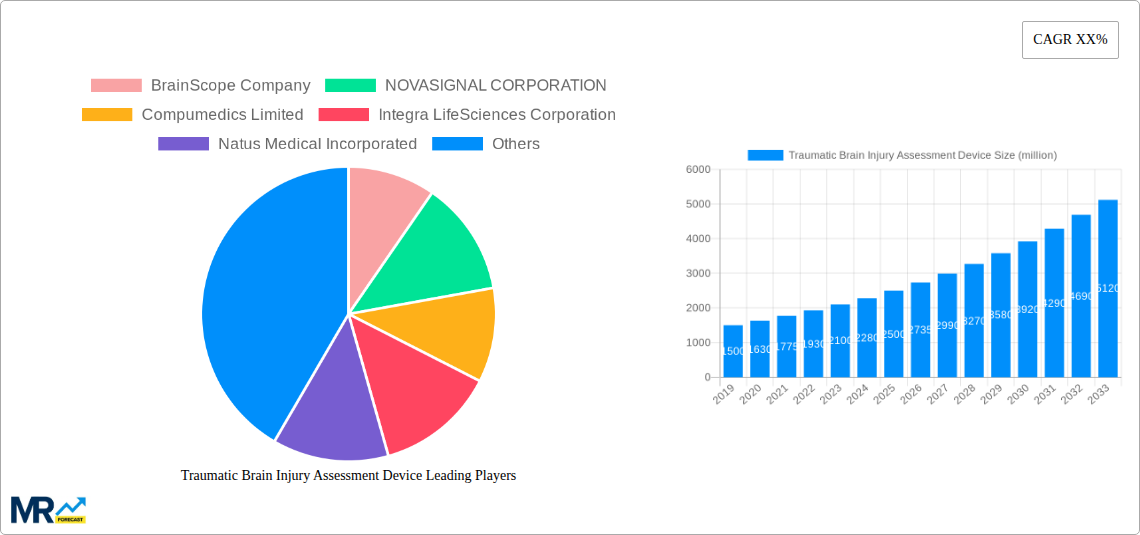

The market is segmented into imaging devices and monitoring equipment, with both segments showing promising growth trajectories. Hospitals and diagnostic centers represent the primary application areas, leveraging these advanced devices for timely and precise assessment of TBI severity. Key players such as BrainScope Company, NOVASIGNAL CORPORATION, Compumedics Limited, and Koninklijke Philips N.V. are actively investing in research and development, introducing innovative solutions that cater to the evolving needs of healthcare providers. While the market exhibits strong growth potential, challenges such as the high cost of some advanced devices and the need for specialized training for their operation could pose moderate restraints. However, the overarching trend of improved diagnostics and patient care in TBI management is expected to overshadow these limitations, leading to sustained market expansion.

Here's a unique report description for a Traumatic Brain Injury Assessment Device market analysis, incorporating your specified elements:
The global Traumatic Brain Injury (TBI) Assessment Device market is poised for substantial growth, projected to reach an estimated value of over $2,500 million by 2025. This dynamic landscape is being sculpted by an increasing global incidence of TBI, driven by factors such as rising sports-related concussions, vehicular accidents, and combat injuries. The historical period from 2019-2024 has witnessed a burgeoning awareness and a consequent demand for more accurate and timely diagnostic tools, laying a strong foundation for the market's expansion. The base year of 2025 stands as a critical pivot point, with the forecast period of 2025-2033 anticipating sustained, robust growth. A key trend shaping this market is the shift towards non-invasive and portable assessment devices. Traditionally, TBI diagnosis relied heavily on subjective clinical assessments and resource-intensive imaging techniques like CT scans and MRIs. However, advancements in electroencephalography (EEG), near-infrared spectroscopy (NIRS), and advanced signal processing algorithms are enabling the development of devices that can provide rapid, objective, and point-of-care TBI assessments. This democratizes access to crucial diagnostic capabilities, extending beyond specialized medical centers to emergency rooms, athletic sidelines, and even remote field settings. Furthermore, the integration of artificial intelligence (AI) and machine learning (ML) into TBI assessment devices is a significant emerging trend. These sophisticated algorithms can analyze complex neurological data with unprecedented speed and accuracy, aiding in the differentiation of TBI severity and the identification of subtle neurological changes that might be missed by traditional methods. The market is also seeing a diversification of device types, moving beyond purely imaging solutions to encompass sophisticated monitoring equipment designed for continuous patient surveillance and the tracking of neurological recovery post-injury. This comprehensive approach to TBI assessment is a defining characteristic of the current and future market trajectory.
Several powerful forces are converging to propel the Traumatic Brain Injury Assessment Device market into an era of accelerated expansion. Foremost among these is the escalating global prevalence of TBI. This surge is directly linked to an uptick in high-impact sports participation, an unfortunate rise in road traffic accidents, and the ongoing presence of combat-related head injuries. This growing burden of TBI necessitates more efficient, accurate, and accessible diagnostic solutions, creating a fertile ground for innovation and market demand. Simultaneously, there is a palpable global push for enhanced patient outcomes and reduced healthcare costs. Early and accurate TBI diagnosis is paramount in preventing secondary brain injury, mitigating long-term disability, and optimizing rehabilitation strategies. Devices that facilitate rapid assessment at the point of care, thus reducing the time to diagnosis and treatment, directly contribute to better patient prognoses and potentially lower overall healthcare expenditures. Furthermore, significant advancements in neurotechnology, including miniaturization of sensors, sophisticated signal processing, and the integration of AI/ML, are enabling the development of novel and more effective TBI assessment tools. These technological breakthroughs are not only enhancing diagnostic accuracy but also increasing the portability and usability of these devices, making them more adaptable to diverse clinical and pre-clinical settings. The increasing investment in research and development by both established medical device companies and emerging neurotech startups is further fueling innovation and bringing cutting-edge TBI assessment solutions to market.
Despite the promising growth trajectory, the Traumatic Brain Injury Assessment Device market faces several significant hurdles and restraints that could temper its expansion. A primary challenge lies in the inherent complexity of TBI itself. TBI is not a singular entity but a spectrum of injuries with varying mechanisms, severities, and resultant neurological deficits. Developing a single assessment device that can accurately diagnose and characterize all types of TBI remains a significant scientific and engineering challenge. This variability can lead to difficulties in standardization and validation of assessment devices, potentially impacting their widespread adoption. Another considerable restraint is the high cost associated with the research, development, and manufacturing of advanced TBI assessment technologies. The development of sophisticated neuroimaging and monitoring equipment often involves substantial capital investment, which can translate into high price points for the end-user. This can limit accessibility, particularly in resource-constrained healthcare settings or developing economies. Regulatory hurdles also present a formidable challenge. Obtaining approval from regulatory bodies like the FDA in the United States or the EMA in Europe for novel medical devices, especially those involving complex algorithms and new diagnostic methodologies, can be a lengthy and rigorous process. This can delay market entry and increase development costs. Furthermore, the need for specialized training and expertise to operate and interpret the data from advanced TBI assessment devices can be a barrier to adoption in settings where such specialized personnel may not be readily available. Finally, established diagnostic pathways, while often less sophisticated, can create inertia, with healthcare providers sometimes reluctant to embrace new technologies without extensive evidence of superior clinical utility and cost-effectiveness.
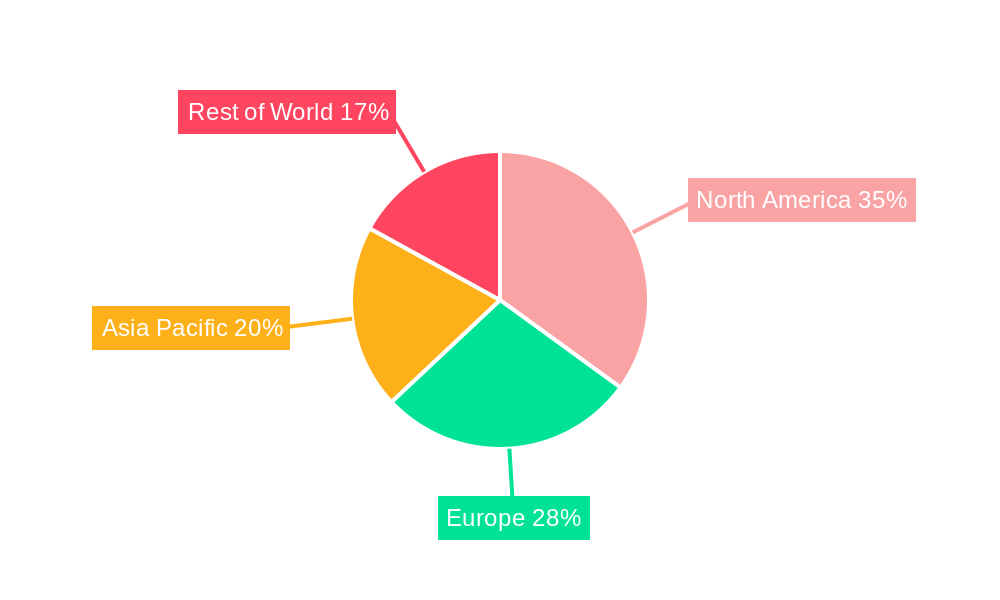
Within the global Traumatic Brain Injury Assessment Device market, the North America region is projected to assert significant dominance, driven by a confluence of compelling factors. This dominance is underpinned by several key indicators and supported by both the Imaging Device and Monitoring Equipment segments, with Hospital applications playing a pivotal role in their adoption.
North America's Market Leadership: The United States, in particular, is a powerhouse in medical device innovation and adoption. The region boasts a robust healthcare infrastructure, significant investment in research and development, and a high level of public and private funding allocated to neurological research. The presence of leading TBI assessment device manufacturers such as BrainScope Company, NOVASIGNAL CORPORATION, Integra LifeSciences Corporation, and General Electric Company, among others, further solidifies North America's position. Furthermore, a high incidence of TBI, stemming from factors like a large population engaged in contact sports, a significant number of vehicular accidents, and a well-established military presence with associated combat injuries, creates a substantial demand for advanced assessment solutions. The regulatory environment, while stringent, also fosters innovation by providing pathways for the approval of novel technologies once they meet rigorous safety and efficacy standards.
Dominance of Imaging Devices: The Imaging Device segment is expected to be a primary growth engine. Advancements in non-invasive imaging techniques are revolutionizing TBI assessment. Devices leveraging electroencephalography (EEG) and near-infrared spectroscopy (NIRS) are gaining traction due to their ability to provide rapid, objective, and quantitative measures of brain function. Companies like Compumedics Limited, NIHON KOHDEN CORPORATION, and Elekta are at the forefront of developing sophisticated EEG-based systems that can detect subtle changes in brain activity indicative of TBI. The trend towards portable and bedside EEG devices is particularly strong, facilitating immediate assessment in emergency settings.
Rise of Monitoring Equipment: The Monitoring Equipment segment is experiencing remarkable growth, driven by the increasing need for continuous patient assessment and tracking of recovery post-TBI. This category includes devices designed for long-term surveillance of neurological function in intensive care units (ICUs) and rehabilitation settings. Companies like Natus Medical Incorporated and Advanced Brain Monitoring are developing advanced neuromonitoring systems that track parameters such as intracranial pressure (ICP), cerebral perfusion pressure (CPP), and brain tissue oxygenation. The integration of AI and ML with monitoring equipment enables predictive analytics, allowing clinicians to anticipate potential complications and optimize treatment strategies. This segment is crucial for understanding the dynamic nature of brain recovery and ensuring personalized rehabilitation plans.
Hospital Applications as the Primary Driver: The Hospital segment, encompassing emergency departments, ICUs, neurosurgery units, and rehabilitation centers, represents the largest and most influential application area for TBI assessment devices. The critical nature of TBI demands immediate and precise diagnostic capabilities, making hospitals the primary adopters of these technologies. The ability of imaging devices to rapidly identify the presence and severity of TBI, coupled with the continuous insights provided by monitoring equipment, directly impacts patient management and outcomes within the hospital setting. As the global healthcare expenditure continues to rise, and with an increasing focus on improving neurological care, the adoption of advanced TBI assessment devices in hospitals worldwide is set to surge, further cementing the dominance of North America in this market.
The Traumatic Brain Injury Assessment Device industry is being catalyzed by a powerful interplay of factors. The persistent and growing global incidence of TBI due to sports, accidents, and military conflicts creates an ever-present demand for reliable diagnostic tools. Concurrent with this is the rapid evolution of neurotechnology, including advancements in non-invasive sensing, signal processing, and AI integration, which are enabling the development of more accurate, portable, and user-friendly devices. Furthermore, increasing government and private sector investments in neurological research and the development of innovative medical technologies are providing the financial impetus for market expansion. The growing awareness among healthcare professionals and the public regarding the long-term consequences of untreated or misdiagnosed TBI is also a significant driver, pushing for earlier and more comprehensive assessments.
This comprehensive report offers an in-depth analysis of the global Traumatic Brain Injury Assessment Device market, providing invaluable insights for stakeholders. It meticulously examines market trends, growth drivers, and key challenges, offering a nuanced understanding of the current landscape. The report features detailed segmentation by device type (Imaging Device, Monitoring Equipment), application (Hospital, Diagnostic Center, Others), and geographic region, enabling targeted strategic planning. With an extensive study period covering 2019-2033, including a base year of 2025 and a forecast period of 2025-2033, the report delivers robust market estimations and projections. It identifies key players and their strategic initiatives, alongside significant technological advancements and regulatory developments, ensuring a holistic view of the industry's trajectory and providing actionable intelligence for navigating this complex and evolving market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 14.99% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 14.99%.
Key companies in the market include BrainScope Company, NOVASIGNAL CORPORATION, Compumedics Limited, Integra LifeSciences Corporation, Natus Medical Incorporated, InfraScan, Dr. Langer Medical GmbH, RAUMEDIC AG, SyncThink, Imeka, General Electric Company, pro2cool Tectrum, NanoDx, NIHON KOHDEN CORPORATION, Elekta, Advanced Brain Monitoring, Boston Scientific Corporation, ESAOTE SPA, Koninklijke Philips N.V, Vivonics.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Traumatic Brain Injury Assessment Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Traumatic Brain Injury Assessment Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.