1. What is the projected Compound Annual Growth Rate (CAGR) of the Tools for SARS-CoV-2?
The projected CAGR is approximately 6.9%.
 Tools for SARS-CoV-2
Tools for SARS-CoV-2Tools for SARS-CoV-2 by Type (Virus Detection, Antibody Development), by Application (Commercial, Industrial, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for Tools for SARS-CoV-2 is poised for significant expansion, projected to reach an estimated USD 5.67 billion in 2025 and experience a robust Compound Annual Growth Rate (CAGR) of 6.9% through 2033. This growth is fueled by the sustained demand for advanced diagnostic, therapeutic, and research tools essential for managing the ongoing and future threats posed by SARS-CoV-2 variants. Key drivers include the increasing prevalence of COVID-19, the continuous development of new vaccines and antiviral therapies, and the growing emphasis on widespread genomic surveillance to detect emerging strains. Furthermore, the expanding applications of SARS-CoV-2 research tools beyond immediate pandemic response, into areas like fundamental virology and novel drug discovery, are creating new avenues for market penetration. The market segments of Virus Detection and Antibody Development are anticipated to lead this expansion, owing to their critical roles in diagnosis and therapeutic intervention.
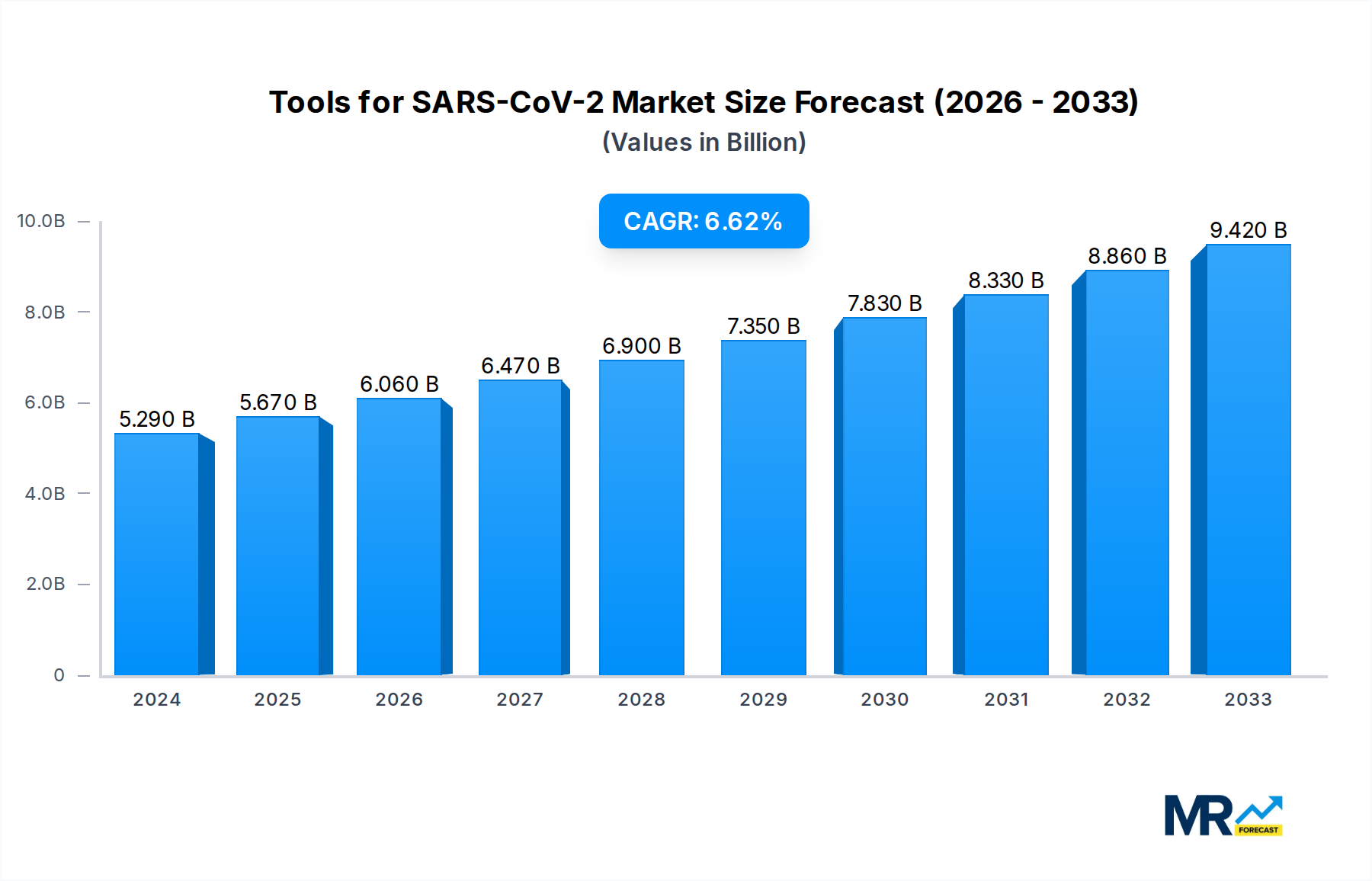

The market dynamics are further shaped by ongoing technological advancements and strategic collaborations among leading biotechnology and pharmaceutical firms. Companies like Emergent BioSolutions, Catalent, The Serum Institute of India, Fujifilm Diosynth Biotechnologies, Lonza, and Thermo Fisher Scientific are at the forefront, investing heavily in research and development to enhance the efficacy and accessibility of SARS-CoV-2 related tools. Regional analysis indicates North America and Europe as dominant markets, driven by substantial investments in healthcare infrastructure and strong research capabilities. However, the Asia Pacific region is expected to witness the highest growth rate, propelled by government initiatives to strengthen public health systems and a growing manufacturing base. While the market is robust, potential restraints may include the high cost of certain advanced tools, regulatory hurdles, and the challenge of ensuring equitable global access to these essential resources.

This report provides an in-depth analysis of the global market for Tools for SARS-CoV-2, covering the period from 2019 to 2033. With a base year of 2025 and a forecast period extending to 2033, the study offers critical insights into market trends, driving forces, challenges, and key growth opportunities. The market, valued in the billions, is meticulously segmented by type, application, and industry developments, with an extensive examination of leading companies.
The global market for SARS-CoV-2 tools has witnessed an unprecedented surge, transitioning from a nascent stage during the initial outbreak in the historical period of 2019-2024 to a robust and indispensable sector in the estimated year of 2025 and beyond. This evolution is largely attributed to the sustained global health imperative to detect, monitor, and combat the virus and its evolving variants. The market has been characterized by rapid innovation, a dramatic increase in production capacity, and a widening array of applications. During the historical period, the primary focus was on diagnostic tools, particularly PCR-based and antigen tests, to enable widespread testing and containment efforts. This led to substantial investments in raw materials, manufacturing infrastructure, and the scaling up of production by key players. As the pandemic progressed, the demand diversified, encompassing tools for antibody development and characterization, crucial for understanding immunity and for the development of vaccines and therapeutics. This shift spurred advancements in assay development, high-throughput screening platforms, and specialized reagents. The market’s trajectory is projected to remain strong throughout the forecast period of 2025-2033, driven by the ongoing need for surveillance, the development of next-generation diagnostics capable of detecting emerging variants, and the increasing integration of these tools into routine healthcare and public health surveillance systems. The value proposition of these tools has expanded beyond immediate pandemic response to encompass long-term preparedness, research into long COVID, and the broader field of respiratory pathogen monitoring. Emerging trends include the development of point-of-care diagnostics with enhanced speed and accessibility, the utilization of AI and machine learning for data analysis and interpretation of test results, and the increasing importance of multiplex assays capable of detecting multiple respiratory pathogens simultaneously. The substantial market value, measured in the billions, underscores the critical role these tools play in global health security and the ongoing economic impact of the pandemic and its aftermath.
The remarkable growth of the Tools for SARS-CoV-2 market is propelled by a confluence of potent factors that have reshaped global health priorities and scientific endeavors. Paramount among these is the persistent threat posed by SARS-CoV-2 and its continuous evolution into new variants. This ongoing challenge necessitates continuous and sophisticated diagnostic capabilities to track the spread of the virus, inform public health policies, and enable timely clinical interventions. The demand for highly sensitive and specific virus detection tools remains robust, driving innovation in areas such as molecular diagnostics and rapid antigen testing. Furthermore, the global imperative for vaccine and therapeutic development has been a significant catalyst. The intricate process of discovering, testing, and manufacturing vaccines and antiviral treatments requires a vast array of specialized tools, including reagents for cell culture, assay kits for efficacy testing, and platforms for protein expression and purification. Companies like Catalent and Lonza have been instrumental in scaling up the production of these critical components. Public health investments, both governmental and private, have also played a crucial role. Governments worldwide have allocated substantial budgets towards pandemic preparedness and response, funding the procurement of diagnostic kits, research initiatives, and the development of robust surveillance systems. This financial backing has directly fueled market expansion and encouraged technological advancements. The increasing recognition of the economic and societal impact of infectious diseases has also bolstered the market. Businesses and industries are increasingly investing in tools that facilitate safe operations, such as employee testing programs and workplace monitoring solutions, further expanding the application spectrum of SARS-CoV-2 tools.
Despite its robust growth, the Tools for SARS-CoV-2 market is not without its significant challenges and restraints that could temper its trajectory. A primary concern revolves around the cost and accessibility of advanced diagnostic and research tools. While the market has scaled significantly, ensuring equitable access to these tools across all socioeconomic strata and geographical regions remains a persistent hurdle. High-throughput diagnostic platforms and specialized reagents can be prohibitively expensive for low-resource settings, potentially exacerbating health disparities. Furthermore, the rapid pace of technological evolution presents a dual-edged sword. While it drives innovation, it also leads to quick obsolescence of existing technologies, requiring continuous investment in research and development and potentially creating a market for frequent upgrades. This rapid evolution can also lead to regulatory hurdles and complex approval processes. Developing and validating new diagnostic tests or research tools that meet stringent regulatory standards can be time-consuming and resource-intensive, potentially delaying market entry for promising innovations. The supply chain vulnerabilities, exposed during the initial phases of the pandemic, continue to pose a risk. Disruptions in the availability of key raw materials, such as specialized enzymes, nucleotides, and assay components, can significantly impact production capacity and lead times, affecting the affordability and availability of critical tools. Finally, market saturation and declining demand in specific segments as the immediate crisis subsides could also act as a restraint. As the acute phase of the pandemic wanes, the demand for mass testing may decrease, requiring market players to pivot towards more specialized applications or long-term surveillance solutions.
The global market for Tools for SARS-CoV-2 is a complex ecosystem with several regions and segments vying for dominance, driven by distinct economic, demographic, and public health landscapes. Among the segments, Virus Detection stands out as a consistently dominant force, projected to maintain its leading position throughout the forecast period of 2025-2033. This dominance is fueled by the ongoing need for accurate and rapid identification of SARS-CoV-2 infections, essential for public health surveillance, clinical management, and preventing further transmission. The segment encompasses a wide array of technologies, from highly sensitive PCR-based tests to more accessible antigen-based rapid diagnostic tests.
Within the Virus Detection segment, the sub-segment of Molecular Diagnostics, particularly RT-PCR, has historically been, and is expected to remain, a cornerstone due to its unparalleled accuracy and sensitivity. However, there is a significant and growing trend towards Rapid Antigen Tests, driven by their speed, lower cost per test, and suitability for point-of-care settings. This evolution is particularly pronounced in commercial and industrial applications where frequent, on-site testing is crucial for maintaining operational continuity.
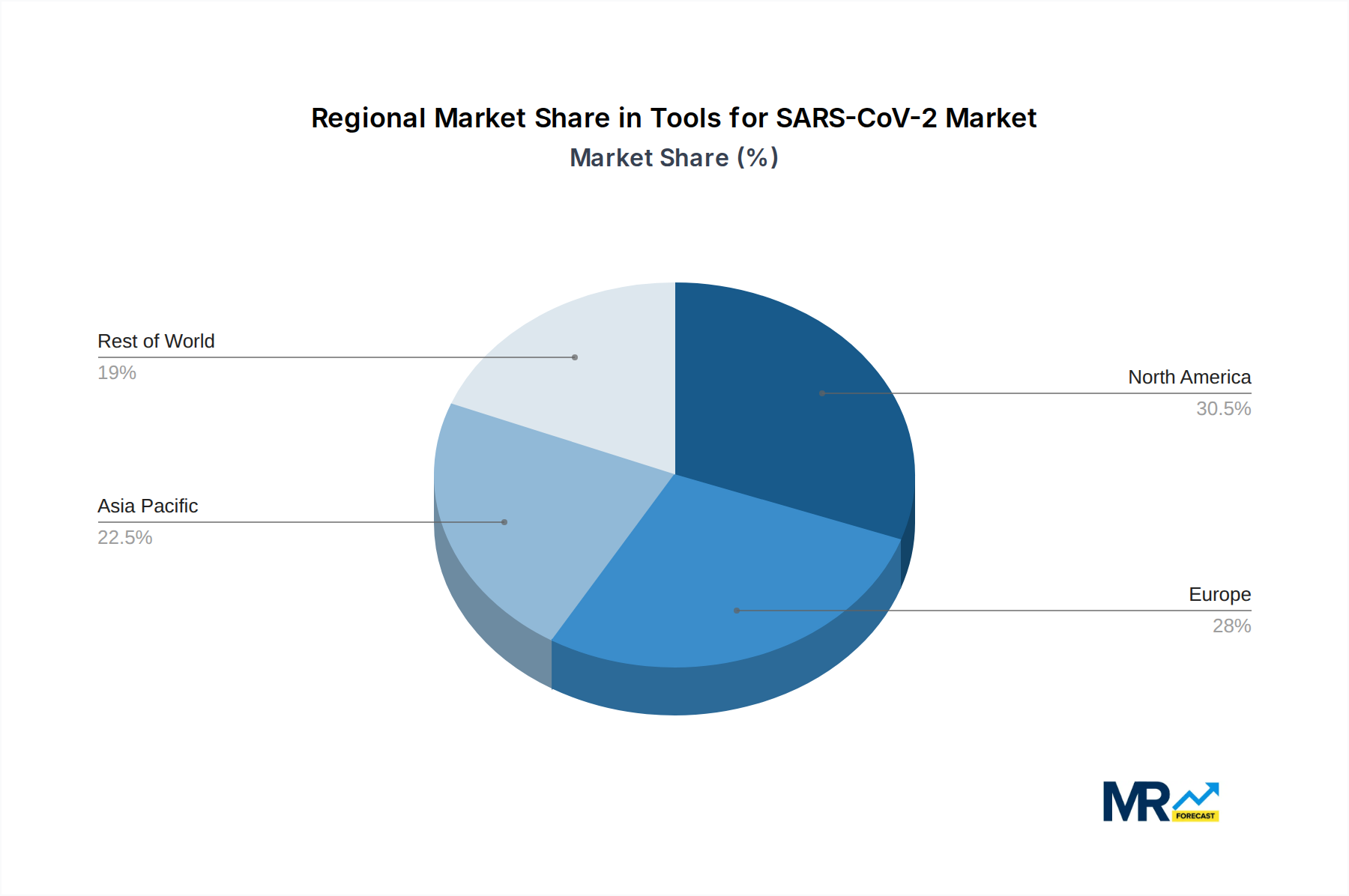
Geographically, North America is poised to retain its significant market share, driven by substantial government investment in public health infrastructure, a high level of technological innovation, and a proactive approach to pandemic preparedness. The presence of leading research institutions and biopharmaceutical companies in the United States and Canada fosters a robust ecosystem for the development and adoption of advanced SARS-CoV-2 tools. Furthermore, the well-established healthcare systems and a high per capita expenditure on healthcare contribute to strong market penetration across all segments.
Following closely, Europe presents another major market, characterized by a fragmented yet strong demand across various countries, each with its own public health strategies and regulatory frameworks. The region benefits from a strong research base, particularly in Germany, the United Kingdom, and France, and significant investments from the European Union in pandemic response and future preparedness. The emphasis on public health initiatives and the presence of prominent life science companies contribute to the region's market leadership.
The Asia-Pacific region is expected to witness the most significant growth over the forecast period, driven by a large and burgeoning population, increasing healthcare expenditure, and a growing awareness of infectious disease threats. Countries like China and India, with their vast populations and significant manufacturing capabilities, are not only major consumers but also increasingly important producers of SARS-CoV-2 tools. The Serum Institute of India, for instance, plays a crucial role in the global supply chain. The increasing adoption of advanced diagnostic technologies and the development of localized manufacturing capabilities are key drivers for this region's ascendance.
The Application segment of Commercial is also expected to demonstrate substantial growth, especially in the context of workforce health and safety. Businesses across various sectors are implementing routine testing protocols to minimize disruptions, protect employees, and maintain operational continuity. This has spurred demand for accessible and scalable testing solutions. The Industrial application, encompassing sectors like manufacturing, logistics, and pharmaceuticals, will continue to rely heavily on these tools for ensuring workforce health and product integrity.
Several key factors will catalyze continued growth within the Tools for SARS-CoV-2 industry. The ongoing need for surveillance of emerging variants necessitates the continuous development and deployment of advanced detection tools. Furthermore, advancements in point-of-care diagnostics are expanding accessibility and speed of testing, particularly in resource-limited settings and decentralized healthcare environments. The increasing integration of artificial intelligence and machine learning in diagnostic platforms is enhancing accuracy, efficiency, and data analysis, paving the way for predictive modeling of outbreaks. Finally, growing investments in long-term pandemic preparedness by governments and international organizations will ensure sustained demand for a broad spectrum of tools, fostering ongoing innovation and market expansion.
This report offers a comprehensive and exhaustive analysis of the Tools for SARS-CoV-2 market, providing invaluable insights for stakeholders seeking to understand its current landscape and future potential. The study delves deeply into market dynamics, analyzing key trends, driving forces, and prevailing challenges that shape the industry. It meticulously segments the market by type (Virus Detection, Antibody Development, etc.), application (Commercial, Industrial, Others), and industry developments, offering a granular view of growth opportunities. Leading players are identified and profiled, alongside a detailed examination of their strategic initiatives and market positioning. The report’s extensive historical analysis (2019-2024) and forward-looking projections (2025-2033), anchored by a base year of 2025, equip readers with a robust understanding of market value, growth trajectories, and critical investment considerations. This in-depth coverage ensures that all facets of this vital market are explored, providing a definitive resource for strategic decision-making.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.9% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6.9%.
Key companies in the market include Emergent BioSolutions, Catalent, The Serum Institute of India, Fujifilm Diosynth Biotechnologies, Lonza, Phlow Corporation, CSL, Oxford Biomedica, Rovi, Thermo Fisher Scientific.
The market segments include Type, Application.
The market size is estimated to be USD 5.67 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion.
Yes, the market keyword associated with the report is "Tools for SARS-CoV-2," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Tools for SARS-CoV-2, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.