1. What is the projected Compound Annual Growth Rate (CAGR) of the Sterilized Medical Packaging?
The projected CAGR is approximately 5.22%.
 Sterilized Medical Packaging
Sterilized Medical PackagingSterilized Medical Packaging by Type (Thermoform Trays, Bottles, Vials, Ampoules, World Sterilized Medical Packaging Production ), by Application (Surgical Instruments, Pharmaceutical, World Sterilized Medical Packaging Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global sterilized medical packaging market is poised for significant expansion, projected to reach approximately $35.86 billion in 2025 and demonstrating a robust Compound Annual Growth Rate (CAGR) of 5.22% throughout the forecast period of 2019-2033. This sustained growth is primarily fueled by the escalating demand for sterile and safe packaging solutions across the pharmaceutical and healthcare industries. The increasing prevalence of chronic diseases, a growing aging population, and advancements in medical device technology are all contributing to a higher volume of surgical procedures and the need for meticulously sterilized medical supplies. Key drivers include stringent regulatory requirements for medical device and pharmaceutical packaging, emphasizing patient safety and product integrity, which necessitates the use of advanced sterilized packaging materials and technologies. Furthermore, the expanding global healthcare infrastructure, particularly in emerging economies, is creating new avenues for market growth. The market segments, including thermoform trays, bottles, vials, and ampoules, cater to diverse applications such as surgical instruments and pharmaceuticals, each contributing to the overall market dynamism.
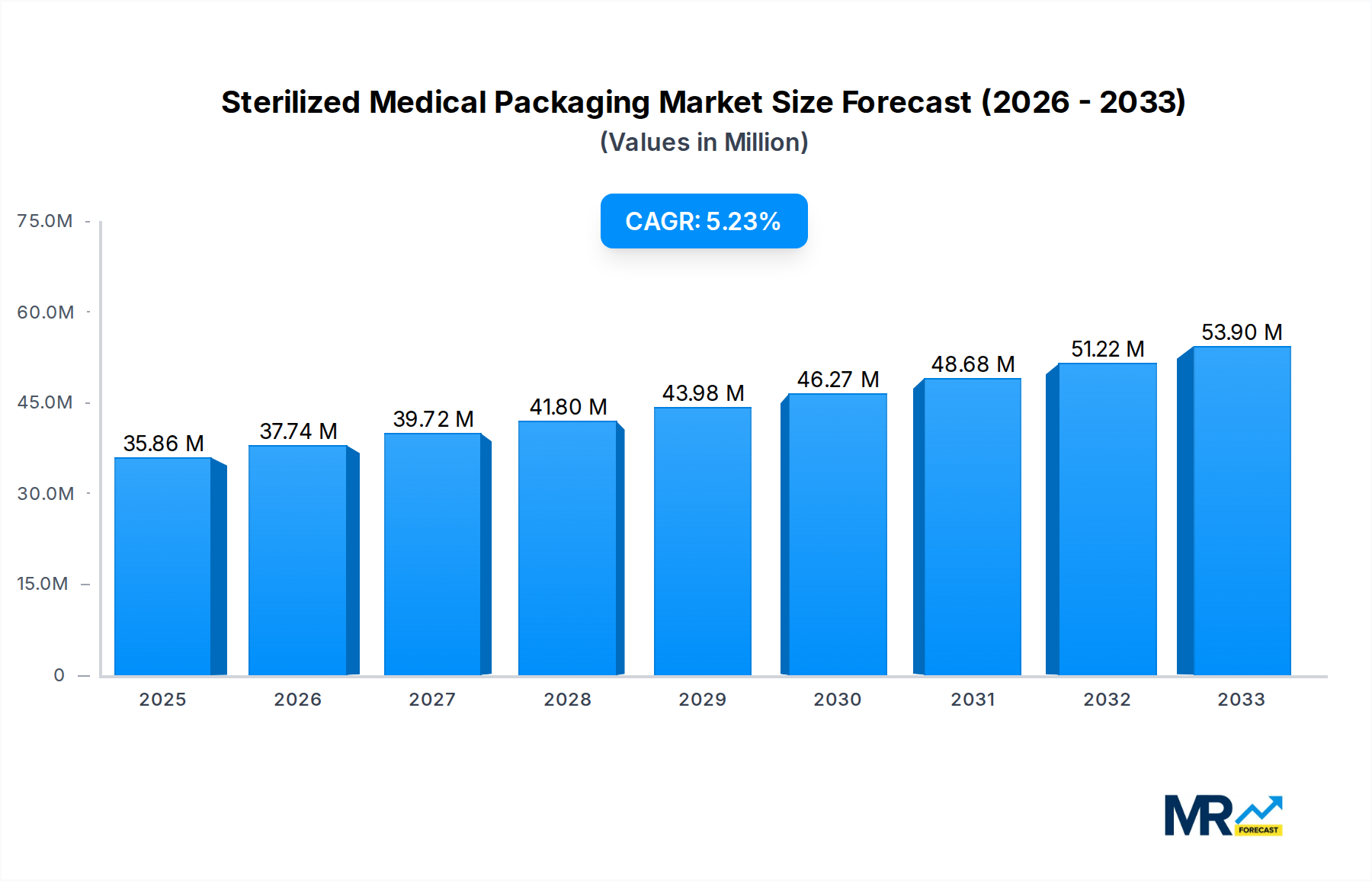

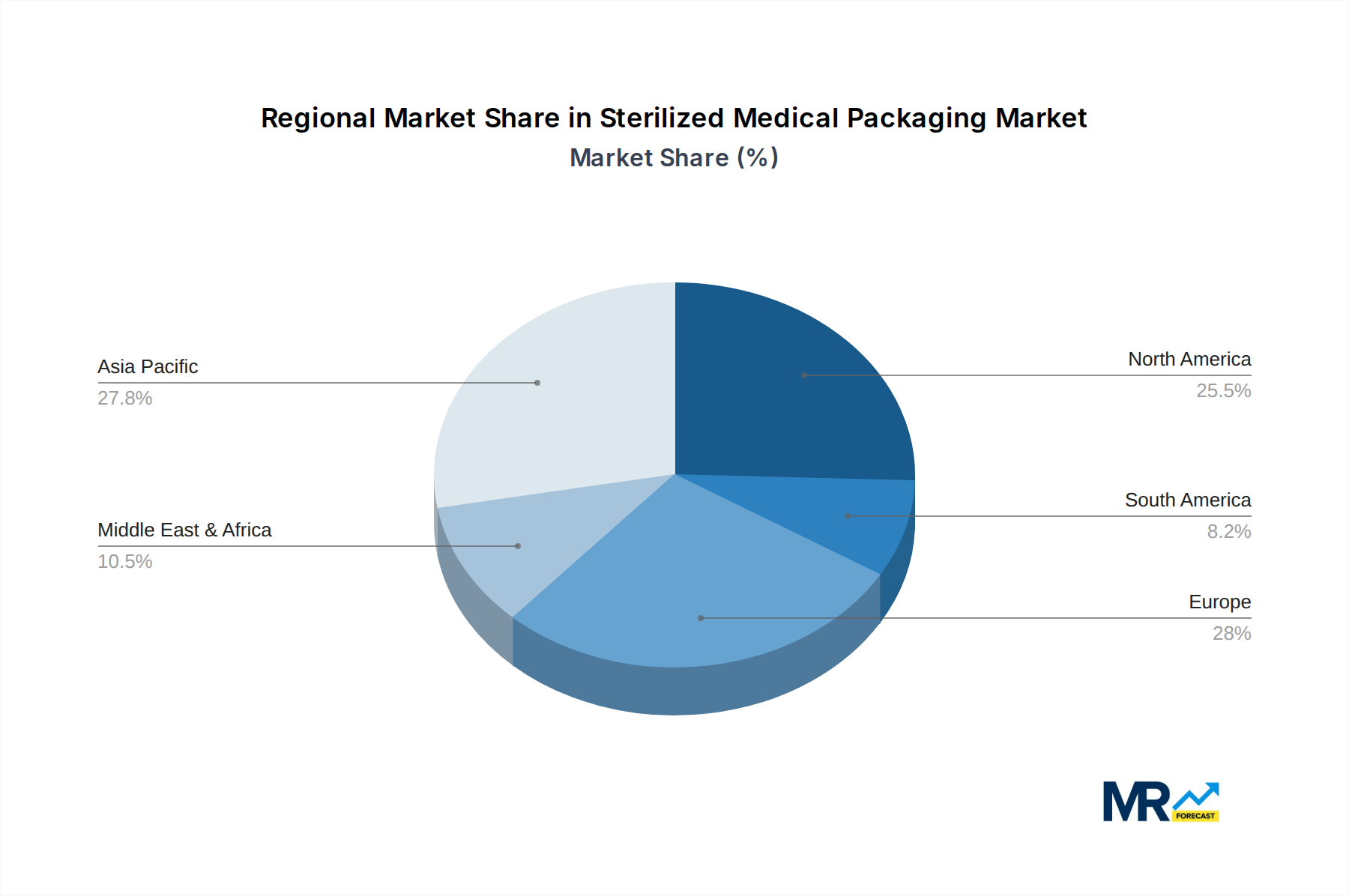
The market's upward trajectory is also influenced by emerging trends like the development of innovative barrier properties to enhance product shelf-life and protection, and the increasing adoption of sustainable packaging materials without compromising sterilization efficacy. Innovations in sterilization methods and materials, such as advanced polymers and sterilization-compatible films, are crucial for maintaining product integrity and preventing contamination. However, certain restraints, including the high cost associated with advanced sterilization technologies and materials, and concerns regarding the environmental impact of certain packaging materials, could temper growth. Major players like Amcor, 3M, and DuPont are continuously investing in research and development to introduce novel packaging solutions and expand their global presence, highlighting the competitive landscape and the strategic importance of technological innovation and market reach. The Asia Pacific region, driven by its large population and expanding healthcare sector, is anticipated to be a key growth engine, alongside established markets in North America and Europe.

Here's a unique report description for Sterilized Medical Packaging, incorporating your specified elements:
Report Title: Global Sterilized Medical Packaging Market: A Comprehensive Analysis (2019-2033) - Navigating Innovation and Demand in a $XX Billion Landscape
Report Description:
This in-depth report provides a comprehensive examination of the global sterilized medical packaging market, a critical sector vital for healthcare safety and efficacy. The market, projected to reach a significant $XXX billion by 2025, is undergoing a dynamic transformation driven by escalating healthcare demands, technological advancements, and stringent regulatory frameworks. Our analysis spans a detailed study period from 2019 to 2033, with a specific focus on the base year of 2025, offering granular insights into market dynamics. The historical period (2019-2024) has laid the groundwork for the forecast period (2025-2033), allowing for robust predictions and strategic recommendations. We delve into the intricate interplay of various segments, including Thermoform Trays, Bottles, Vials, and Ampoules, and their pivotal role in the world sterilized medical packaging production. Furthermore, we meticulously analyze the applications in Surgical Instruments and Pharmaceutical sectors, understanding their unique packaging requirements and market influences. This report is an indispensable resource for stakeholders seeking to understand current trends, identify growth opportunities, and mitigate challenges within this rapidly evolving industry.
The sterilized medical packaging market is characterized by a strong undercurrent of innovation and evolving consumer needs, fundamentally reshaping how healthcare products are protected and delivered. A paramount trend is the increasing demand for advanced barrier properties, moving beyond traditional materials to incorporate sophisticated films and laminates that offer enhanced protection against microbial contamination, moisture, and oxygen. This is particularly crucial for high-value and sensitive medical devices and pharmaceuticals. The integration of smart packaging solutions, such as temperature indicators and tamper-evident seals, is gaining significant traction. These features not only ensure product integrity but also provide valuable real-time information to healthcare providers and patients, fostering greater trust and reducing the risk of compromised products reaching the end-user. Sustainability is no longer a secondary consideration but a driving force behind material innovation. Manufacturers are actively exploring and adopting eco-friendly alternatives, including recyclable polymers, biodegradable materials, and reduced-plastic solutions, without compromising on the critical sterilization and protection requirements. This shift is driven by both regulatory pressures and growing consumer awareness. The rise of personalized medicine and biologics necessitates specialized packaging solutions. This includes sterile pre-filled syringes, vials for cell and gene therapies, and custom-designed trays for complex surgical kits. The complexity of these products demands packaging that maintains sterility throughout their lifecycle and offers ease of use for healthcare professionals. Furthermore, the trend towards miniaturization in medical devices is influencing packaging design, requiring smaller, more efficient, and precisely engineered sterile packaging formats. The seamless integration of packaging with the supply chain is another key trend, with an emphasis on traceability and end-to-end visibility. This involves utilizing technologies like RFID and barcodes to track sterilized products from manufacturing to the point of care, ensuring a secure and efficient supply chain. The ongoing digital transformation within the healthcare sector is also impacting packaging, with a growing focus on automated sterilization processes and packaging line integration.
Several powerful forces are collectively propelling the global sterilized medical packaging market forward, underpinning its substantial growth trajectory. The most significant driver is the ever-increasing global healthcare expenditure and the expanding access to healthcare services worldwide. As populations grow and the prevalence of chronic diseases rises, the demand for sterile medical devices, pharmaceuticals, and diagnostic kits escalates, directly translating into a higher requirement for sterilized packaging. The continuous innovation in medical devices and pharmaceutical formulations also plays a crucial role. Breakthroughs in minimally invasive surgery, advanced drug delivery systems, and novel biologics necessitate highly specialized, sterile packaging solutions that can maintain the integrity and efficacy of these cutting-edge products throughout their shelf life and during transport. Stringent regulatory standards and mandates from health authorities across the globe, such as the FDA and EMA, are instrumental in driving the adoption of robust and reliable sterilized packaging. These regulations emphasize patient safety, demanding high levels of sterility assurance, barrier protection, and material compliance, thereby fostering a market for advanced packaging solutions. The growing preference for single-use medical devices, driven by concerns over hospital-acquired infections and the desire for enhanced convenience and efficiency, is another significant growth catalyst. Single-use products inherently require individual sterile packaging, creating a substantial and expanding market segment. Furthermore, the global shift towards decentralized healthcare delivery, including home healthcare and remote patient monitoring, is spurring the demand for sterile packaging that is user-friendly, tamper-evident, and capable of maintaining sterility outside of traditional hospital settings.
Despite its robust growth, the sterilized medical packaging market faces several significant challenges and restraints that can temper its expansion. One of the primary hurdles is the escalating cost of raw materials. Fluctuations in the prices of polymers, specialized films, and other packaging components can impact profit margins for manufacturers and potentially lead to increased costs for healthcare providers. The complex and often multi-layered nature of sterilized medical packaging, while necessary for optimal protection, can create difficulties in end-of-life disposal. This poses a significant environmental challenge, and the push for sustainable solutions, while a growth driver, also represents a restraint as industries grapple with developing cost-effective and truly biodegradable or recyclable alternatives that meet stringent sterilization requirements. The rigorous and ever-evolving regulatory landscape, while beneficial for patient safety, can also be a significant restraint. Obtaining regulatory approvals for new packaging materials or designs can be a time-consuming, expensive, and complex process, potentially slowing down innovation and market entry for new products. The risk of counterfeiting in the pharmaceutical and medical device industries presents another challenge. Sterilized medical packaging plays a crucial role in preventing counterfeits, but sophisticated counterfeiters can still attempt to replicate packaging, necessitating continuous advancements in anti-counterfeiting technologies and secure supply chain management. Furthermore, the specialized nature of sterilized medical packaging requires significant investment in sophisticated manufacturing equipment and sterile processing facilities. This high capital expenditure can be a barrier to entry for smaller companies and can limit the scalability of production for some players. Finally, ensuring consistent quality and sterility across large-scale production runs requires rigorous quality control measures, and any lapse in these processes can lead to product recalls and reputational damage.
Several key regions and segments are poised to dominate the global sterilized medical packaging market, driven by a confluence of demographic, economic, and technological factors.
Dominant Regions:
Dominant Segments:
These regions and segments are characterized by high R&D investment, a concentration of key industry players, robust regulatory frameworks that encourage quality, and a continuously growing demand for safe and effective healthcare products.
The sterilized medical packaging industry is experiencing robust growth fueled by several key catalysts. The escalating global demand for healthcare services, driven by an aging population and the increasing prevalence of chronic diseases, is a primary growth engine. Furthermore, advancements in medical technology and the introduction of novel pharmaceuticals, biologics, and minimally invasive surgical devices necessitate increasingly sophisticated and reliable sterile packaging solutions. Stringent regulatory requirements worldwide, prioritizing patient safety and product integrity, also act as a powerful catalyst, pushing manufacturers towards higher quality and more secure packaging options. The growing trend of single-use medical devices, propelled by infection control concerns and convenience, directly translates into increased demand for individual sterile packaging.
This report offers unparalleled comprehensive coverage of the sterilized medical packaging market. It delves into the intricate details of market segmentation, analyzing the dynamics of types like Thermoform Trays, Bottles, Vials, and Ampoules, and their contribution to the world sterilized medical packaging production. The application analysis focuses on critical sectors such as Surgical Instruments and Pharmaceuticals, understanding their unique packaging imperatives. The report meticulously covers the Study Period: 2019-2033, with a foundational analysis of the Base Year: 2025 and the Forecast Period: 2025-2033, building upon the Historical Period: 2019-2024. It provides crucial insights into market trends, growth drivers, challenges, and opportunities, equipping stakeholders with the knowledge to make informed strategic decisions in this vital and evolving industry.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.22% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.22%.
Key companies in the market include Amcor, 3M, DuPont, WestPharma, SteriPack, Wipak, Placon, Riverside Medical, Oracle Packaging, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Sterilized Medical Packaging," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Sterilized Medical Packaging, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.