1. What is the projected Compound Annual Growth Rate (CAGR) of the Sterile Liquid Transfer Port?
The projected CAGR is approximately 6.7%.
 Sterile Liquid Transfer Port
Sterile Liquid Transfer PortSterile Liquid Transfer Port by Type (Single Component System, Two Component System, World Sterile Liquid Transfer Port Production ), by Application (Laboratory, Biotechnology, Pharmaceutical, Others, World Sterile Liquid Transfer Port Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
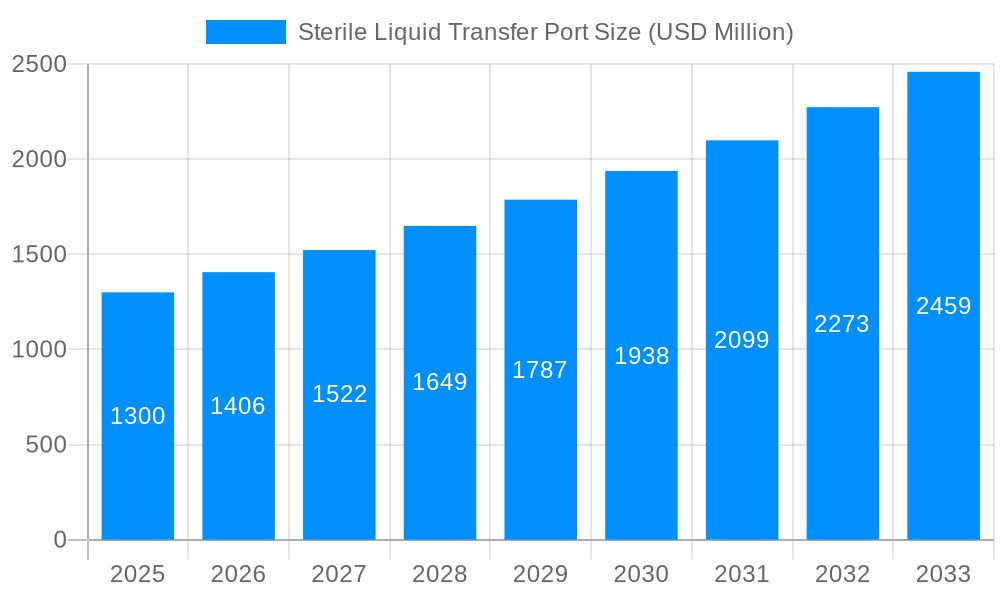
The global sterile liquid transfer port market is poised for significant expansion, projected to reach approximately $1.3 billion in 2025. This growth is underpinned by a robust Compound Annual Growth Rate (CAGR) of 8.1%, indicating sustained and dynamic market evolution throughout the forecast period of 2025-2033. The increasing demand for sterile liquid handling solutions across critical sectors like pharmaceuticals, biotechnology, and advanced laboratories is the primary driver. These industries are at the forefront of developing new therapies, diagnostic tools, and biopharmaceuticals, all of which necessitate stringent aseptic conditions for liquid transfer to prevent contamination and ensure product integrity. The inherent need for secure, reliable, and contamination-free connections in these sensitive applications fuels the adoption of advanced sterile liquid transfer port technologies.

The market's expansion is further propelled by ongoing technological innovations and a growing emphasis on regulatory compliance. Companies are investing in developing single and two-component systems that offer enhanced ease of use, improved sterility assurance, and greater efficiency in fluid management. The rising prevalence of biopharmaceutical manufacturing, coupled with the increasing complexity of drug development pipelines, directly translates into a greater requirement for sophisticated sterile transfer solutions. While the market exhibits strong growth, potential restraints could include the high initial investment costs associated with some advanced systems and the need for specialized training for optimal usage. However, the paramount importance of product safety and efficacy in the healthcare and life sciences sectors is expected to outweigh these challenges, ensuring a consistent upward trajectory for the sterile liquid transfer port market.

This comprehensive report delves into the dynamic global market for Sterile Liquid Transfer Ports, meticulously analyzing trends, driving forces, challenges, and future prospects. With a robust study period spanning 2019-2033, this research provides an in-depth understanding of the market's trajectory, utilizing 2025 as both the base and estimated year, and projecting growth through the 2025-2033 forecast period. The historical context from 2019-2024 offers valuable insights into past performance and evolutionary shifts. The report quantifies the market's significant expansion, with World Sterile Liquid Transfer Port Production projected to reach an astounding $X.XX Billion by 2033, indicating a Compound Annual Growth Rate (CAGR) of Y.YY% during the forecast period.
The global Sterile Liquid Transfer Port market is experiencing a significant evolutionary surge, driven by an increasing demand for aseptic processing across various critical industries. The core trend revolves around the growing emphasis on contamination control and the reduction of human intervention in liquid handling processes. As the pharmaceutical and biotechnology sectors continue to push the boundaries of drug discovery and development, the need for reliable, sterile, and efficient methods of transferring sensitive liquids becomes paramount. This has fueled a steady adoption of advanced sterile liquid transfer port technologies designed to maintain product integrity and prevent microbial ingress. Furthermore, the market is witnessing a pronounced shift towards disposable or single-use sterile transfer systems, particularly within the burgeoning biotechnology landscape. This trend is motivated by the desire to minimize the risk of cross-contamination, streamline cleaning and validation processes, and enhance overall operational flexibility, especially in early-stage research and pilot-scale manufacturing. The increasing complexity of biologics and advanced therapies, often requiring intricate and multi-step transfer protocols, further amplifies the demand for highly engineered and customizable sterile liquid transfer port solutions. Regulatory stringency, particularly concerning Good Manufacturing Practices (GMP), is also a powerful catalyst, compelling manufacturers to invest in and implement validated sterile transfer systems that meet the highest industry standards. The market is also observing a rise in the integration of smart technologies, such as real-time monitoring and data logging capabilities within transfer ports, offering enhanced traceability and quality assurance. This continuous innovation landscape underscores the market's responsiveness to evolving industry needs and its commitment to safeguarding product quality and patient safety. The projected market value, expected to exceed $X.XX Billion by 2033, directly reflects these burgeoning trends and the indispensable role sterile liquid transfer ports play in modern biopharmaceutical operations.
Several potent forces are collectively propelling the growth of the global Sterile Liquid Transfer Port market. Foremost among these is the relentless expansion of the pharmaceutical and biotechnology industries. The escalating research and development activities, coupled with the growing pipeline of novel biologics, vaccines, and cell and gene therapies, necessitate highly controlled and sterile manufacturing environments. Sterile liquid transfer ports are indispensable components in these processes, ensuring the aseptic integrity of precious biological materials throughout their journey from bioreactors to downstream processing. Another significant driver is the increasing global healthcare expenditure and the rising prevalence of chronic diseases, which translate into a higher demand for pharmaceuticals and, consequently, for the advanced manufacturing technologies that produce them. The stringent regulatory landscape across major pharmaceutical markets, mandating rigorous contamination control and aseptic processing, further reinforces the demand for high-quality sterile liquid transfer ports. Companies are compelled to invest in these solutions to comply with regulatory bodies like the FDA and EMA, thereby mitigating risks of product recalls and ensuring patient safety. The growing trend towards personalized medicine and the development of smaller batch sizes for specialized therapies also contribute to the market's expansion. Flexible and adaptable sterile transfer solutions are essential for handling these diverse production requirements efficiently. Moreover, advancements in material science and engineering have led to the development of more robust, biocompatible, and user-friendly sterile liquid transfer ports, enhancing their appeal and adoption across a wider range of applications.
Despite the robust growth trajectory, the Sterile Liquid Transfer Port market faces several challenges and restraints that could potentially temper its expansion. One of the primary hurdles is the high initial cost associated with acquiring and implementing advanced sterile liquid transfer systems. This can be a significant barrier for smaller research institutions and emerging biopharmaceutical companies with limited capital budgets. The complexity of integrating these systems into existing manufacturing workflows can also be a challenge, often requiring specialized expertise and infrastructure modifications. Furthermore, the need for rigorous validation and qualification procedures to ensure compliance with regulatory standards adds to the overall cost and time investment. The stringent requirements for sterility assurance necessitate thorough testing and documentation, which can be resource-intensive. Another concern is the potential for supply chain disruptions, particularly in the sourcing of specialized components and materials required for sterile transfer ports. Geopolitical factors, trade disputes, and unforeseen global events can impact the availability and cost of essential raw materials, thereby affecting production timelines and market stability. The rapid pace of technological innovation also presents a challenge. Manufacturers must continuously invest in research and development to keep pace with evolving industry demands and emerging technologies, which can strain resources. Finally, the availability of alternative, albeit less sophisticated, liquid transfer methods in certain low-resource settings, coupled with the inherent resistance to adopting entirely new methodologies, can pose a restraint to market penetration in some regions.
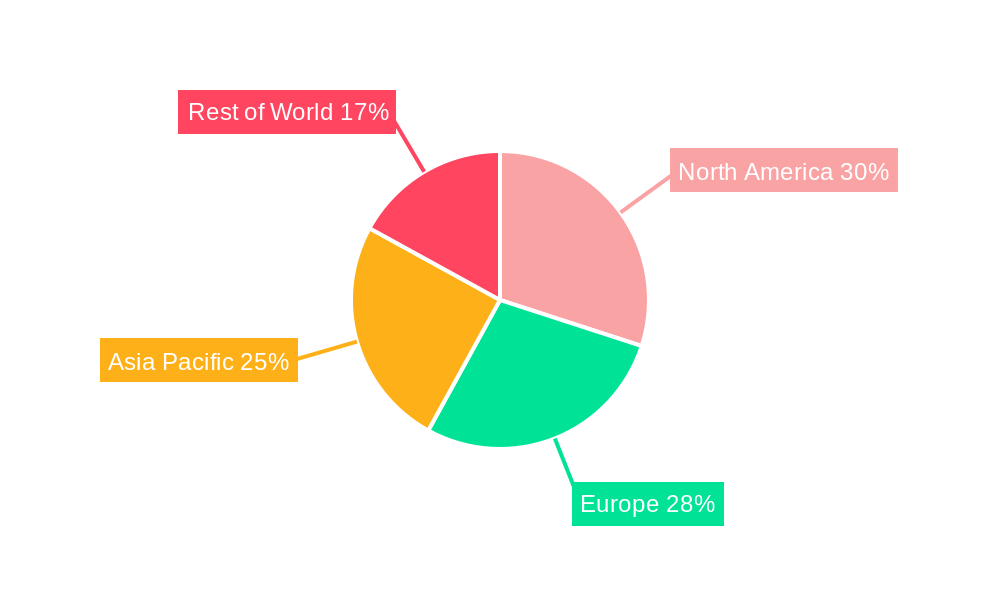
The global Sterile Liquid Transfer Port market is poised for significant regional dominance and segment growth, with North America and Europe expected to lead due to their well-established pharmaceutical and biotechnology ecosystems, coupled with stringent regulatory frameworks.
North America:
Europe:
Asia Pacific:
Other Regions (Latin America, Middle East & Africa):
Segment Dominance:
The Pharmaceutical and Biotechnology application segments are collectively expected to dominate the global Sterile Liquid Transfer Port market throughout the forecast period. The increasing complexity of biopharmaceutical manufacturing, the rise of biologics, and the stringent requirements for aseptic processing in these sectors directly translate into a sustained and growing demand for sophisticated sterile liquid transfer solutions. Within the Type segment, both Single Component Systems and Two Component Systems will witness significant growth. Single Component Systems offer simplicity and cost-effectiveness for certain applications, while Two Component Systems provide enhanced security and sterility assurance, making them indispensable for high-value and highly sensitive biopharmaceutical production. The overall World Sterile Liquid Transfer Port Production is intricately tied to the health and expansion of these core application segments.
Several key factors act as potent growth catalysts for the Sterile Liquid Transfer Port industry. The relentless innovation in biopharmaceutical drug development, particularly the rise of complex biologics, cell therapies, and gene therapies, necessitates increasingly sophisticated and reliable aseptic transfer methods. Furthermore, the ongoing globalization of pharmaceutical manufacturing and the establishment of new production facilities in emerging markets are creating significant demand for sterile transfer solutions. Growing investments by contract development and manufacturing organizations (CDMOs) in advanced aseptic processing capabilities also act as a strong catalyst. The increasing focus on patient safety and regulatory compliance worldwide, driven by agencies like the FDA and EMA, compels manufacturers to adopt and upgrade to high-quality sterile liquid transfer ports.
This report offers an all-encompassing analysis of the Sterile Liquid Transfer Port market, providing stakeholders with invaluable insights to inform strategic decision-making. It meticulously details market segmentation by type, application, and region, offering granular data on the projected size and growth of each segment. The report includes a comprehensive examination of the competitive landscape, profiling key players and their market strategies. Furthermore, it delves into the technological advancements, regulatory trends, and emerging opportunities shaping the industry's future. The economic impact of the market is quantified, with detailed projections of World Sterile Liquid Transfer Port Production values, and its contribution to the broader pharmaceutical and biotechnology sectors. This in-depth coverage ensures that readers gain a profound understanding of the market dynamics and future outlook.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.7% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6.7%.
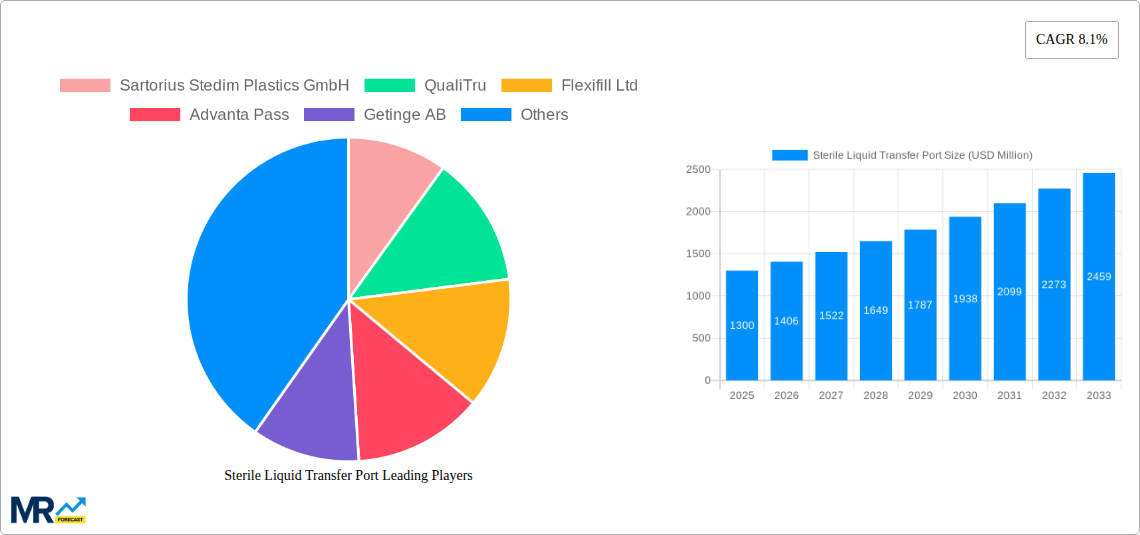
Key companies in the market include Sartorius Stedim Plastics GmbH, QualiTru, Flexifill Ltd, Advanta Pass, Getinge AB, Aseptic Technologies, Syntegon Technology GmbH, Veltek Associates, Inc, Tema Sinergie Spa, Corning Incorporated, Atec Pharmatechnik GmbH, Weber Scientific.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Sterile Liquid Transfer Port," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Sterile Liquid Transfer Port, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.