1. What is the projected Compound Annual Growth Rate (CAGR) of the Sterile Auto Disable Syringes?
The projected CAGR is approximately 5.4%.
 Sterile Auto Disable Syringes
Sterile Auto Disable SyringesSterile Auto Disable Syringes by Type (0.05-1ML, 2ML, 3ML, 5ML, 10ML, Others, World Sterile Auto Disable Syringes Production ), by Application (Hospitals, Clinics, Others, World Sterile Auto Disable Syringes Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for Sterile Auto Disable Syringes is poised for substantial growth, projected to reach an estimated 15.27 billion USD by 2025, with a robust Compound Annual Growth Rate (CAGR) of 8.4% anticipated throughout the forecast period of 2025-2033. This expansion is primarily fueled by the increasing global emphasis on infection prevention and control, particularly in healthcare settings. The inherent safety features of auto-disable syringes, designed to prevent reuse and the subsequent transmission of bloodborne pathogens, are driving their adoption in vaccination campaigns, chronic disease management, and routine medical procedures. Key market drivers include rising healthcare expenditure, a growing prevalence of infectious diseases, and government initiatives promoting the use of single-use medical devices. The market is segmented by type, with 0.05-1ML and 2ML syringes representing significant volume due to their widespread use in pediatric and adult vaccinations respectively. Hospitals and clinics constitute the dominant application segments, reflecting the core demand from direct patient care environments.
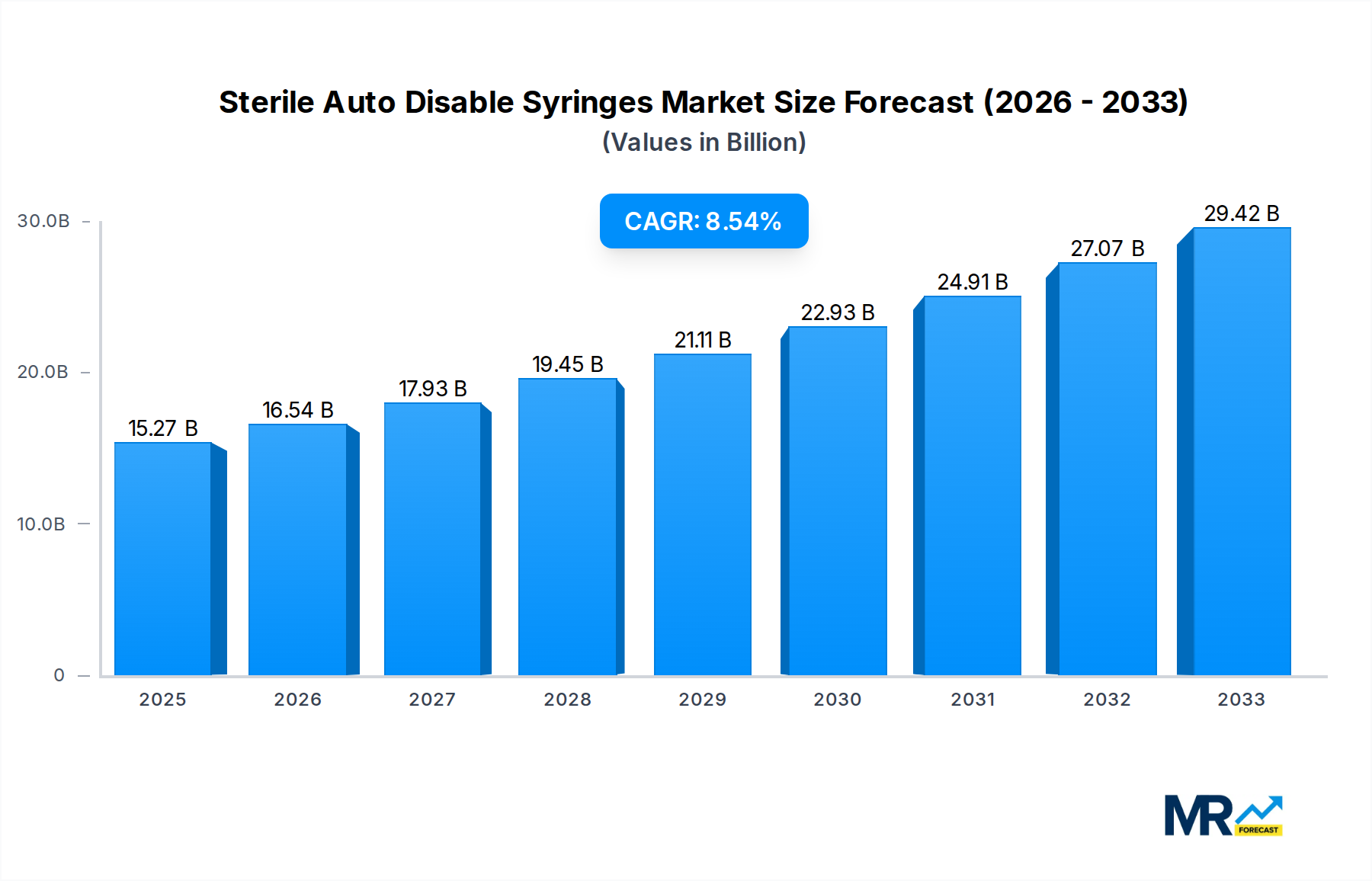

Emerging economies, particularly in the Asia Pacific region, are expected to witness the fastest growth, driven by expanding healthcare infrastructure, increasing awareness of safe injection practices, and a large, underserved population requiring access to sterile medical devices. Major players like BD, Terumo, and Vogt Medical are actively investing in research and development, expanding their manufacturing capacities, and forming strategic partnerships to cater to the escalating demand. However, the market faces certain restraints, including stringent regulatory approvals for medical devices and potential price sensitivity in some developing regions. Nevertheless, the overarching trend towards enhanced patient safety and the imperative to curb healthcare-associated infections are expected to overpower these challenges, ensuring a positive trajectory for the Sterile Auto Disable Syringes market in the coming years. The focus on affordability and accessibility will also be crucial for market players aiming to capture the full potential of this dynamic sector.

Here's a report description on Sterile Auto Disable Syringes, incorporating the requested information and structure:
The global Sterile Auto Disable (SAD) syringes market is experiencing robust and sustained growth, a trend anticipated to continue throughout the forecast period of 2025-2033. Driven by an increasing awareness of injection safety and the critical need to prevent the transmission of bloodborne diseases, the market is projected to reach several billion dollars in value by 2033. XXX, a leading market research firm, highlights key insights indicating a strong upward trajectory. The historical period from 2019-2024 laid a solid foundation, witnessing steady adoption of SAD syringes driven by public health initiatives and stricter regulatory frameworks in developed economies. The base year of 2025 marks a pivotal point, with estimated production volumes and market value set to surge. A significant driver is the escalating prevalence of chronic diseases requiring regular parenteral drug administration, such as diabetes and various autoimmune conditions. Furthermore, the push for universal healthcare access and immunization programs, particularly in emerging economies, is significantly augmenting demand. The inherent safety features of auto-disable syringes, designed to prevent reuse after a single injection, directly address the critical issue of needle-stick injuries and subsequent infection risks, making them a preferred choice for healthcare providers globally. The market's expansion is also being fueled by advancements in manufacturing technologies that enhance product quality and cost-effectiveness, making these essential safety devices more accessible. Innovations in materials science and ergonomic design are further contributing to user comfort and efficiency.
The global Sterile Auto Disable (SAD) syringes market is propelled by a confluence of powerful driving forces, each contributing to its accelerated expansion. Paramount among these is the unwavering global commitment to enhancing injection safety protocols. The devastating consequences of needle-stick injuries and the subsequent spread of infections like HIV and Hepatitis B and C have created an urgent imperative for governments and healthcare organizations worldwide to adopt safer medical devices. SAD syringes, by design, eliminate the possibility of reuse, thereby directly mitigating these risks. This inherent safety feature has made them indispensable in clinical settings, particularly in vaccination campaigns and routine medical procedures. Furthermore, the increasing global burden of chronic diseases necessitating frequent injections, such as diabetes, rheumatoid arthritis, and other autoimmune disorders, directly translates into a higher demand for disposable and safe injection devices. As healthcare access expands, especially in developing nations, the need for affordable and safe syringes becomes even more pronounced. Regulatory mandates from health authorities globally, advocating for the use of safety-engineered syringes, are also playing a crucial role in driving market growth. These regulations often stipulate the use of non-reusable syringes in various healthcare settings, thereby creating a favorable market environment for SAD syringes.
Despite the robust growth trajectory, the Sterile Auto Disable (SAD) syringes market is not without its challenges and restraints that could potentially temper its expansion. A primary concern revolves around the cost differential between standard disposable syringes and SAD syringes. While the long-term benefits of preventing infections are undeniable, the initial procurement cost of SAD syringes can be a deterrent for resource-constrained healthcare facilities and governments, particularly in low-income countries. This cost sensitivity can lead to a slower adoption rate in these regions, despite the pressing need for improved injection safety. Moreover, the manufacturing process for SAD syringes can be more complex and require specialized equipment compared to conventional syringes. This can lead to higher production costs and potentially create supply chain vulnerabilities, especially during periods of surge demand or global disruptions. The awareness and training gap regarding the correct usage and disposal of SAD syringes also presents a challenge. While the syringes are designed for safety, improper handling or a lack of understanding of their auto-disable mechanism could still lead to suboptimal safety outcomes. Finally, the existence of alternative safety-engineered syringes, such as those with needle protection features, can create a competitive landscape. While SAD syringes offer a distinct advantage in preventing reuse, other technologies might appeal to specific market segments or address particular concerns, thus fragmenting demand to some extent.
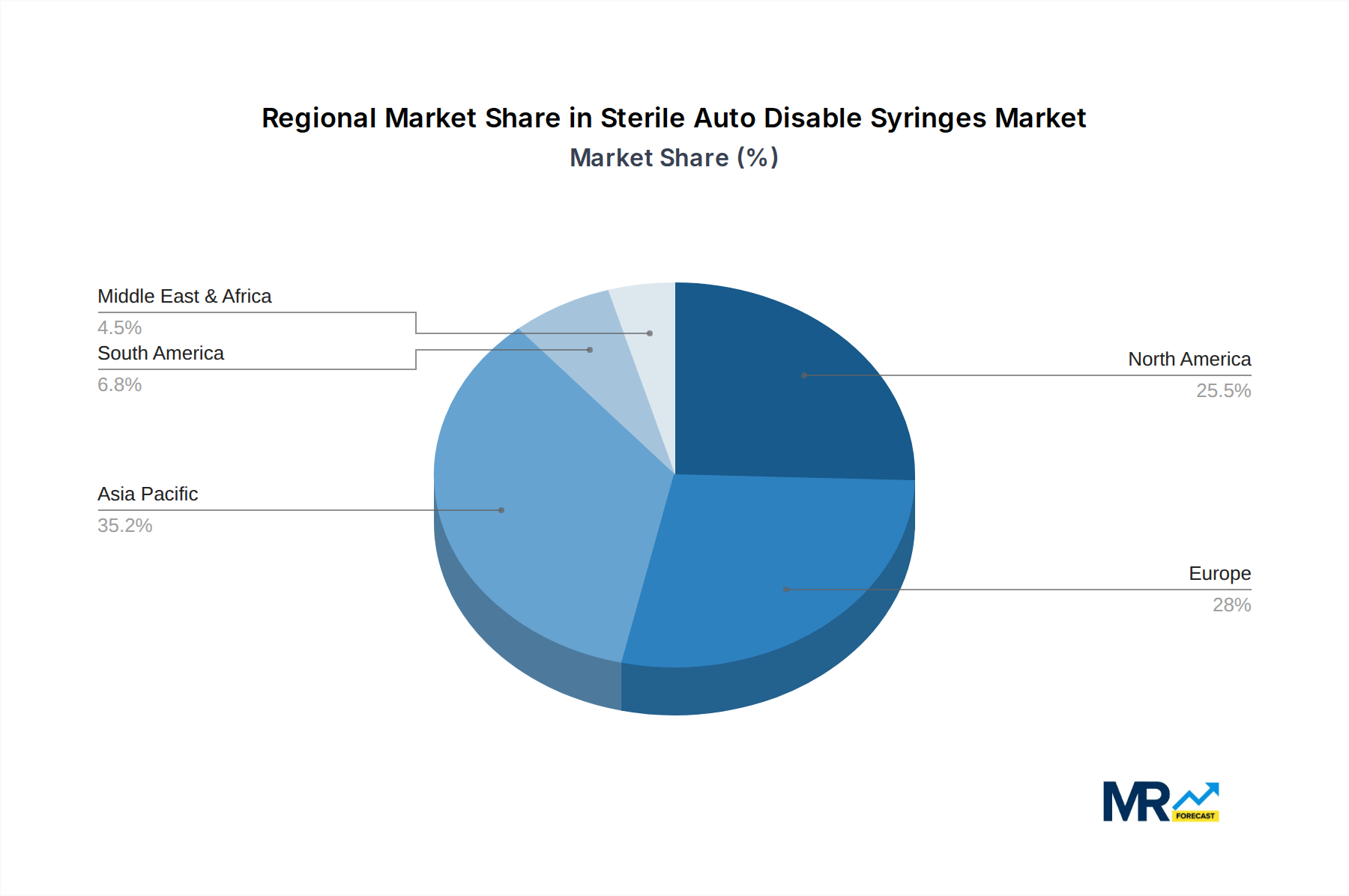
The global Sterile Auto Disable (SAD) syringes market exhibits distinct regional dominance and segment leadership, shaped by a complex interplay of healthcare infrastructure, regulatory frameworks, and disease prevalence.
Dominant Region: North America and Europe
Dominant Segment: 3ML and 5ML Syringes (Type)
Dominant Application: Hospitals
The interplay between these dominant regions, segments, and applications creates a powerful market dynamic, ensuring sustained growth and innovation in the Sterile Auto Disable Syringes sector.
The Sterile Auto Disable (SAD) syringes industry is experiencing several key growth catalysts. The persistent global focus on preventing healthcare-associated infections (HAIs) and bloodborne pathogens directly fuels demand for these safety-engineered devices. Furthermore, the ongoing expansion of immunization programs worldwide, particularly for routine childhood vaccinations and emerging public health needs, is a significant driver. The increasing prevalence of chronic diseases requiring regular parenteral drug administration, such as diabetes and autoimmune disorders, also contributes to sustained market growth. Advancements in manufacturing technology are leading to more cost-effective production, making SAD syringes more accessible to a wider range of healthcare settings.
This comprehensive report provides an in-depth analysis of the global Sterile Auto Disable (SAD) syringes market from the historical period of 2019-2024 through to the forecast period of 2025-2033, with 2025 serving as the base and estimated year. It meticulously examines market trends, driving forces, challenges, and significant regional and segmental analyses. The report details key industry developments and offers a thorough competitive landscape analysis, featuring leading players in the industry. It is designed to offer stakeholders a holistic understanding of the market's past performance, current standing, and future trajectory, enabling informed strategic decision-making.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.4% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.4%.
Key companies in the market include BD, Terumo, Vogt Medical, SPM Medicare, Shandong Ande Healthcare Apparatus, Azur Medical, Guangdong Intmed Medical Appliance, Guangdong Haiou Medical Apparatus, Hunan Luzhou Huikang, Shanxi XinHuaMei Medical Device, Shanghai Kindly Enterprise Development Group, Anhui Tiankang Medical Technology, Shandong Weigao Group, Zhejiang Yusheng Medical Instrument, Wuxi Yushou Medical Appliances, Hunan Pingan Medical Device Technology, Jiangxi Hongda Medical Equipment, Berpu Medical, Zhejiang Lingyang Medical Apparatus, Zhejiang KangKang Medical-Devices, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Sterile Auto Disable Syringes," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Sterile Auto Disable Syringes, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.