1. What is the projected Compound Annual Growth Rate (CAGR) of the Respiratory Pathogens Test Kits?
The projected CAGR is approximately 5.5%.
 Respiratory Pathogens Test Kits
Respiratory Pathogens Test KitsRespiratory Pathogens Test Kits by Application (Hospitals and Clinical Laboratories, Diagnostic Centers and Screening Facilities, Home and Individual Self-Assessment, World Respiratory Pathogens Test Kits Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
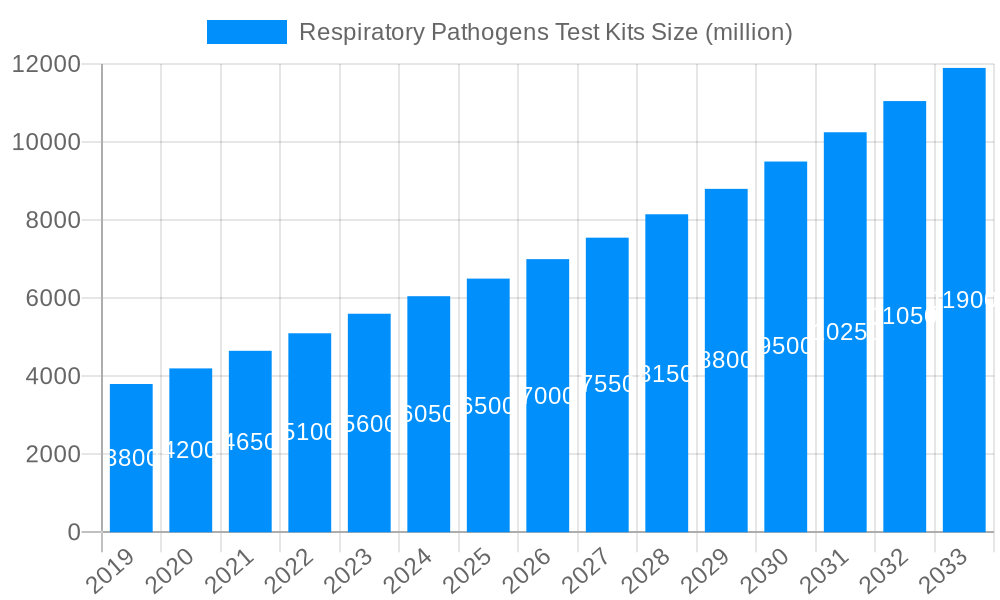
The global respiratory pathogens test kits market is poised for significant expansion, projected to reach an estimated market size of USD 6,500 million by 2025 and grow at a robust Compound Annual Growth Rate (CAGR) of approximately 8.5% through 2033. This upward trajectory is primarily fueled by the escalating prevalence of respiratory infections, heightened public health awareness, and the continuous demand for rapid and accurate diagnostic solutions. The COVID-19 pandemic has undeniably acted as a potent catalyst, underscoring the critical importance of timely pathogen identification for effective disease management and containment. Furthermore, advancements in molecular diagnostic technologies, such as Polymerase Chain Reaction (PCR) and next-generation sequencing (NGS), are enabling the development of more sensitive, specific, and multiplexed testing kits, capable of detecting a wider range of pathogens simultaneously. This technological evolution is a key driver, facilitating earlier and more precise diagnoses, thereby improving patient outcomes and reducing the burden on healthcare systems.
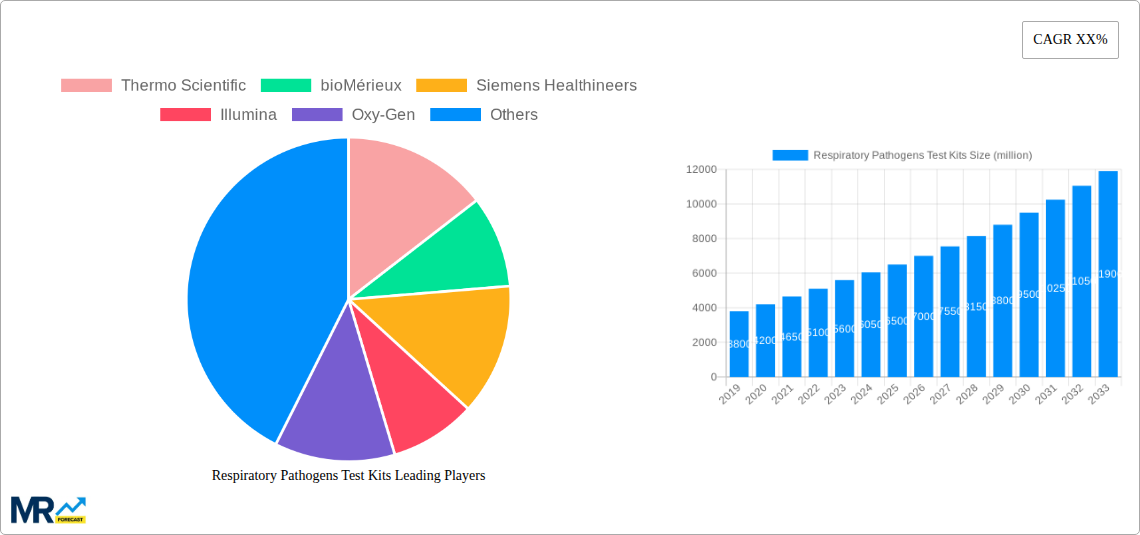

The market's growth is further supported by the increasing adoption of respiratory pathogen testing across diverse healthcare settings. Hospitals and clinical laboratories remain the dominant segment, leveraging these kits for routine diagnosis and patient management. However, there is a discernible surge in demand from diagnostic centers and screening facilities, driven by proactive public health initiatives and screening programs. The burgeoning home and individual self-assessment segment also presents a substantial growth opportunity, propelled by the increasing consumer interest in personalized health monitoring and the convenience offered by point-of-care testing solutions. Geographically, North America and Europe are expected to maintain their leadership positions due to advanced healthcare infrastructure and high research and development investments. Asia Pacific, however, is anticipated to witness the fastest growth, spurred by a large population, increasing disposable income, and a growing focus on improving public health surveillance. Key players such as Thermo Scientific, bioMérieux, and Siemens Healthineers are at the forefront of innovation, actively contributing to market expansion through new product development and strategic collaborations.

Here's a comprehensive report description for Respiratory Pathogens Test Kits, incorporating your specified requirements:
The global market for Respiratory Pathogen Test Kits is poised for significant expansion, projected to reach approximately USD 8,500 million by 2025, with an estimated trajectory towards USD 15,200 million by 2033. This robust growth, spanning the historical period of 2019-2024 and extending through the forecast period of 2025-2033, is underpinned by a confluence of factors, most notably the increased awareness and proactive approach towards infectious respiratory diseases, amplified by recent global health events. The study period of 2019-2033 encapsulates a dynamic evolution in diagnostic methodologies and market penetration. During the historical period, the market experienced a surge driven by the unprecedented demand for COVID-19 testing, which laid the groundwork for broader adoption of rapid and accurate respiratory pathogen detection. This foundational growth established crucial infrastructure and accelerated innovation in multiplex testing capabilities, allowing for the simultaneous identification of multiple respiratory agents. Looking ahead, the market's continued ascent is fueled by the ongoing need for accurate and timely diagnosis of influenza, RSV, and other common and emerging respiratory threats. The transition to advanced molecular diagnostic techniques, such as PCR and isothermal amplification, continues to be a key trend, offering higher sensitivity and specificity compared to traditional methods. Furthermore, the increasing prevalence of chronic respiratory conditions and the aging global population, who are more susceptible to respiratory infections, contribute to a sustained demand for these vital diagnostic tools. The market is witnessing a geographical shift, with emerging economies increasingly investing in healthcare infrastructure, including diagnostic capabilities, thereby broadening the market's reach and potential. The integration of artificial intelligence and machine learning in data analysis from these kits also represents a nascent but promising trend, aiming to improve diagnostic efficiency and epidemiological surveillance. Overall, the respiratory pathogens test kits market is characterized by innovation, increasing demand, and a growing global emphasis on proactive health management.
The relentless upward trajectory of the Respiratory Pathogens Test Kits market is significantly propelled by a multifaceted array of drivers. The persistent threat of novel and re-emerging respiratory pathogens, as starkly demonstrated by recent global pandemics, has permanently elevated the importance of rapid and reliable diagnostic tools. This heightened awareness has translated into sustained government and private sector investment in infectious disease surveillance and control, directly benefiting the market for these essential test kits. Furthermore, the increasing adoption of point-of-care testing (POCT) devices is a pivotal driver. These portable and user-friendly kits allow for quicker results, enabling faster clinical decision-making and reducing the burden on centralized laboratories. The growing demand for multiplex assays, capable of identifying a panel of respiratory pathogens simultaneously from a single sample, is another significant catalyst. This not only improves diagnostic efficiency but also helps in differentiating between viral and bacterial infections, leading to more targeted treatment and reduced antimicrobial resistance. The aging global population and the rising incidence of immunocompromised individuals are also contributing factors, as these demographics are at higher risk of severe respiratory infections, thus necessitating more frequent and accurate testing. The proactive screening initiatives implemented by healthcare systems worldwide, aimed at early detection and containment of outbreaks, further bolster the demand for these kits.
Despite the promising growth outlook, the Respiratory Pathogens Test Kits market is not without its hurdles. One of the primary challenges revolves around the high cost of advanced molecular diagnostic kits, particularly for widespread deployment in resource-limited settings. The initial investment in equipment and consumables can be a significant barrier, hindering accessibility and adoption in certain regions. Furthermore, stringent regulatory approval processes across different geographies can prolong the time-to-market for new test kits, impacting innovation and market entry. The ever-evolving nature of respiratory pathogens, leading to the emergence of new strains and variants, necessitates continuous research and development to update existing kits and develop novel diagnostic solutions, which can be a costly and time-consuming endeavor. Technical expertise required for operating sophisticated molecular diagnostic equipment can also be a restraint, particularly in areas with a shortage of trained laboratory personnel. Moreover, the reimbursement landscape for diagnostic tests can be complex and variable, impacting the profitability and adoption rates for certain types of tests. Finally, concerns regarding the accuracy and sensitivity of some rapid diagnostic tests, especially those intended for home use, can lead to a lack of confidence among end-users and healthcare providers, necessitating rigorous validation and standardization efforts.
The Hospitals and Clinical Laboratories segment is anticipated to exhibit a commanding presence and dominate the Respiratory Pathogens Test Kits market throughout the study period. This dominance stems from several critical factors that solidify their position as the primary consumers and drivers of demand.
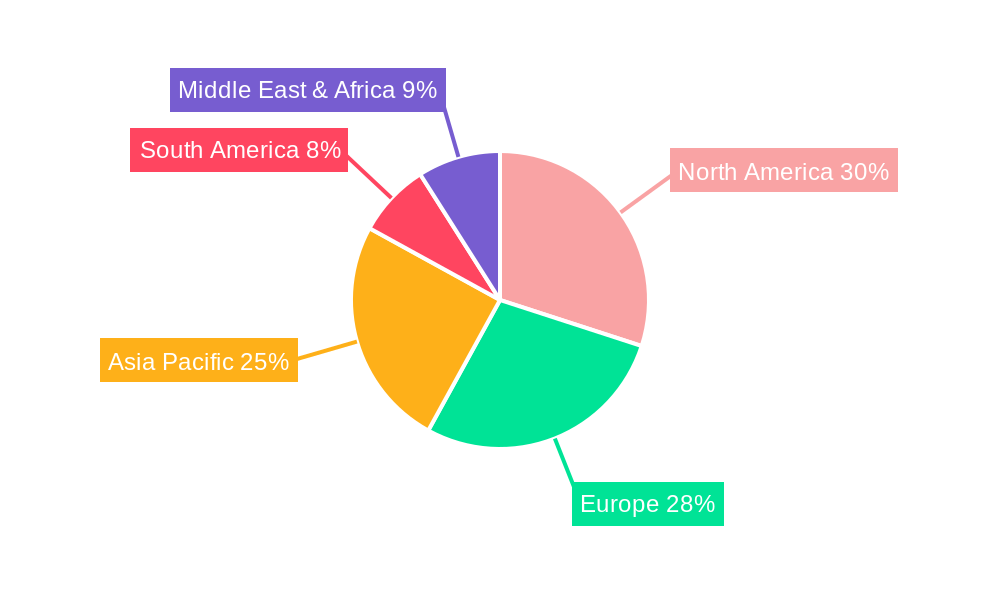
Geographically, North America is projected to lead the market, driven by its advanced healthcare infrastructure, high disposable incomes, and a strong emphasis on public health surveillance. The presence of leading diagnostic companies and a proactive regulatory environment in countries like the United States and Canada further bolsters this region's dominance. Following closely, Europe also represents a significant market, characterized by well-established healthcare systems and a high prevalence of respiratory diseases. The region's commitment to research and development and the implementation of robust screening programs contribute to sustained demand. The Asia Pacific region, while historically lagging, is demonstrating robust growth. Rapidly developing economies, increasing healthcare expenditure, and growing awareness of infectious diseases are propelling the adoption of advanced diagnostic technologies. Countries like China and India are key contributors to this growth, with expanding populations and an increasing focus on public health initiatives. The demand for respiratory pathogen test kits in this region is expected to surge as healthcare infrastructure continues to improve and awareness of the importance of rapid diagnostics grows.
Several key factors act as significant growth catalysts for the Respiratory Pathogens Test Kits industry. The increasing global burden of respiratory diseases, amplified by emerging infectious agents and the growing susceptibility of vulnerable populations, creates a constant and escalating demand for accurate diagnostic solutions. The rapid advancements in molecular diagnostic technologies, leading to more sensitive, specific, and faster test kits, are making these tools more accessible and effective. The growing emphasis on personalized medicine and antimicrobial stewardship also fuels the demand for precise pathogen identification to guide targeted treatments and reduce unnecessary antibiotic use.
This comprehensive report offers an in-depth analysis of the Respiratory Pathogens Test Kits market, providing invaluable insights for stakeholders. It meticulously examines market dynamics, including growth drivers, restraints, and emerging opportunities, with a focus on the period from 2019 to 2033. The report delves into the technological advancements shaping the diagnostic landscape, such as the increasing adoption of PCR, isothermal amplification, and next-generation sequencing. It also provides detailed market segmentation by application, covering hospitals, clinical laboratories, diagnostic centers, and home-use self-assessment kits, along with an analysis of world production and industry developments. The competitive landscape is thoroughly explored, featuring detailed profiles of leading players and their strategic initiatives. This report is an essential resource for understanding the current state and future trajectory of the Respiratory Pathogens Test Kits market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.5%.
Key companies in the market include Thermo Scientific, bioMérieux, Siemens Healthineers, Illumina, Oxy-Gen, Prism Health Lab, TRUPCR, DiaSorin, SML Genetree, GT Molecular, Capitalbio Technology, Wuhan Zhongzhi Biotechnologies.
The market segments include Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Respiratory Pathogens Test Kits," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Respiratory Pathogens Test Kits, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.