1. What is the projected Compound Annual Growth Rate (CAGR) of the Personalized Cell Therapeutic?
The projected CAGR is approximately 24.6%.
 Personalized Cell Therapeutic
Personalized Cell TherapeuticPersonalized Cell Therapeutic by Type (Embryonic Stem Cells, Adult Stem Cell, Induced Pluripotent Cells, Other), by Application (Cardiovascular Diseases, Neurological Disorders, Diabetes, Oncology, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Personalized Cell Therapeutic market is poised for extraordinary expansion, driven by significant advancements in regenerative medicine and a growing demand for tailored treatment strategies. Valued at approximately \$17.59 billion, the market is projected to experience a robust Compound Annual Growth Rate (CAGR) of 24.6% over the forecast period, indicating substantial future value. This surge is primarily fueled by the escalating prevalence of chronic diseases such as cardiovascular disorders, neurological conditions, and diabetes, for which cell-based therapies offer novel and effective solutions. The increasing sophistication of stem cell technologies, including embryonic stem cells, adult stem cells, and induced pluripotent stem cells (iPSCs), along with their expanding applications in oncology and other life-threatening diseases, are key accelerators. Furthermore, a growing understanding of cellular mechanisms and the development of personalized approaches to treatment are creating a fertile ground for market growth, attracting considerable investment and innovation from leading pharmaceutical and biotechnology firms.
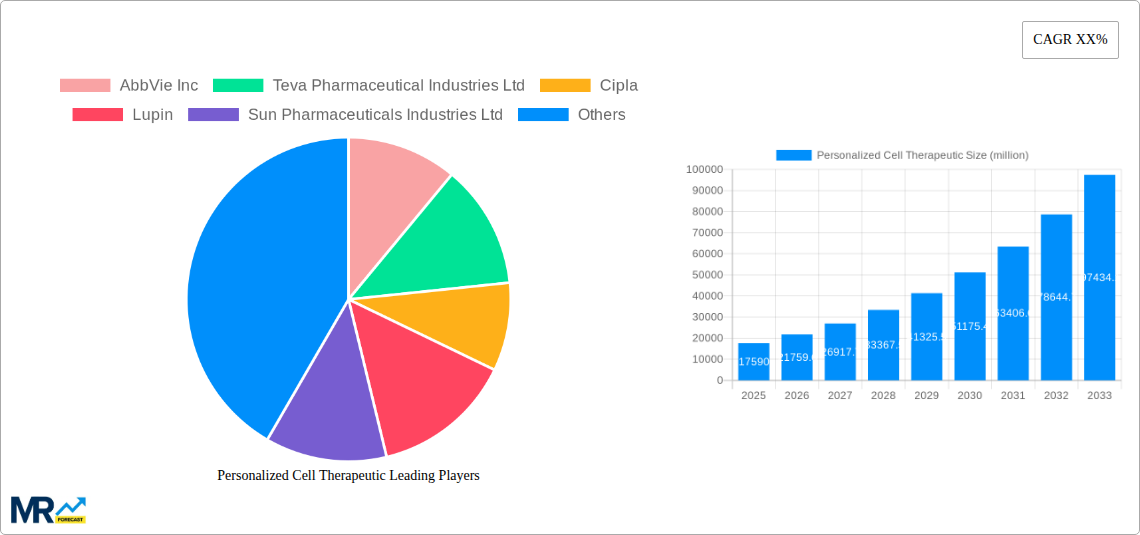

The competitive landscape is characterized by intense research and development activities, strategic collaborations, and mergers and acquisitions aimed at expanding product pipelines and market reach. Prominent players like Pfizer, AstraZeneca, Roche, and Amgen are actively investing in personalized cell therapies, seeking to leverage their established expertise and infrastructure. The market's growth is further supported by favorable regulatory environments and increasing healthcare expenditure globally, particularly in developed economies. While challenges such as high manufacturing costs and stringent regulatory approvals exist, the transformative potential of personalized cell therapies in addressing unmet medical needs is undeniable, positioning this market as a critical frontier in modern healthcare. The strategic focus on developing targeted therapies for specific patient populations and genetic profiles will continue to define the trajectory of this dynamic and rapidly evolving sector.
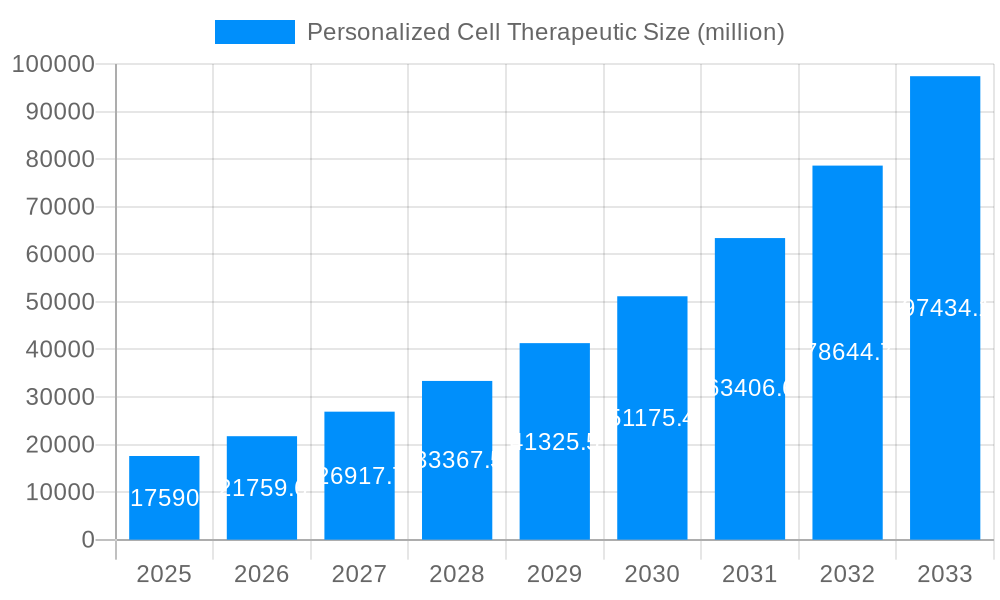

The Personalized Cell Therapeutic market is poised for exponential growth, projected to reach an astonishing $185.5 billion by 2033. This seismic shift in healthcare is driven by an unprecedented understanding of cellular mechanisms and the ability to harness these for targeted therapeutic interventions. The historical period (2019-2024) laid the foundational groundwork, witnessing significant advancements in research and development, particularly in the realm of induced pluripotent stem cells (iPSCs). These versatile cells, reprogrammed from adult cells, offer immense potential for patient-specific therapies, minimizing immunogenicity and maximizing efficacy. The base year, 2025, marks a critical inflection point, with the market already demonstrating robust momentum and setting the stage for sustained expansion throughout the forecast period (2025-2033). Key market insights reveal a strong preference for Induced Pluripotent Cells as the dominant cell type, attributed to their ethical sourcing and broad applicability. Furthermore, Oncology applications are at the forefront, leveraging cell therapies to revolutionize cancer treatment with CAR-T therapies and other immunotherapies. However, the market is not monolithic; while oncology leads, the burgeoning potential in treating Neurological Disorders is a significant emerging trend, with early-stage clinical trials showing promising results for conditions like Parkinson's and Alzheimer's. The increasing prevalence of chronic diseases, coupled with a growing demand for less invasive and more effective treatment modalities, is fueling this transformative market. The integration of advanced gene-editing technologies, such as CRISPR-Cas9, is further accelerating the development of next-generation cell therapies, enabling precise correction of genetic defects and enhancement of therapeutic cell functions. This synergy between cell biology, genetics, and advanced manufacturing techniques is creating a powerful ecosystem for personalized medicine. The market's trajectory is also shaped by evolving regulatory landscapes and increasing investment from both public and private sectors, signifying a collective belief in the paradigm-shifting capabilities of personalized cell therapeutics.
The personalized cell therapeutic market's remarkable ascent is being propelled by a confluence of powerful forces. A fundamental driver is the ever-deepening scientific understanding of human biology at the cellular level. This knowledge allows researchers to identify specific cellular pathways involved in disease pathogenesis and to engineer cells with precise therapeutic functions. Complementing this scientific progress is the surging demand for more effective and less toxic treatment options, particularly for chronic and life-threatening conditions. Patients and healthcare providers are actively seeking alternatives to conventional therapies that often come with significant side effects or limited efficacy. The advent of advanced gene-editing technologies, such as CRISPR-Cas9, has been a game-changer, enabling the precise modification of cellular genomes to correct defects or impart new therapeutic capabilities. This technological leap has significantly expanded the potential applications of cell therapies. Furthermore, substantial and sustained investment from venture capital firms, pharmaceutical giants, and government funding bodies underscores the industry's immense promise and de-risks novel therapeutic development. As R&D pipelines mature and promising clinical trial results emerge, this financial impetus continues to grow, fueling innovation and accelerating the translation of laboratory breakthroughs into clinical realities.
Despite its immense promise, the personalized cell therapeutic market faces significant hurdles that temper its unbridled growth. A primary challenge lies in the high cost of development and manufacturing. Creating patient-specific cell therapies is an intricate and resource-intensive process, involving complex biological manipulations, stringent quality control, and specialized infrastructure. This translates into prohibitively high treatment costs for many patients, posing a significant barrier to widespread accessibility and adoption. The complex regulatory pathways for these novel therapies also present a considerable challenge. Regulatory bodies are still developing frameworks to assess the safety and efficacy of these highly personalized and dynamic treatments, leading to lengthy and unpredictable approval processes. Scalability of manufacturing remains another critical bottleneck. Producing personalized cell therapies on a large scale to meet growing demand requires significant investment in manufacturing capacity and the development of efficient, reproducible processes. Furthermore, clinical efficacy and long-term safety data are still being gathered for many cell therapy applications. While early results are promising, robust evidence demonstrating sustained clinical benefit and absence of long-term adverse events is crucial for widespread clinical acceptance and reimbursement. Finally, patient and physician education and adoption are essential. Understanding the complexities of cell therapies and integrating them into existing treatment paradigms requires significant educational efforts and overcoming potential skepticism.
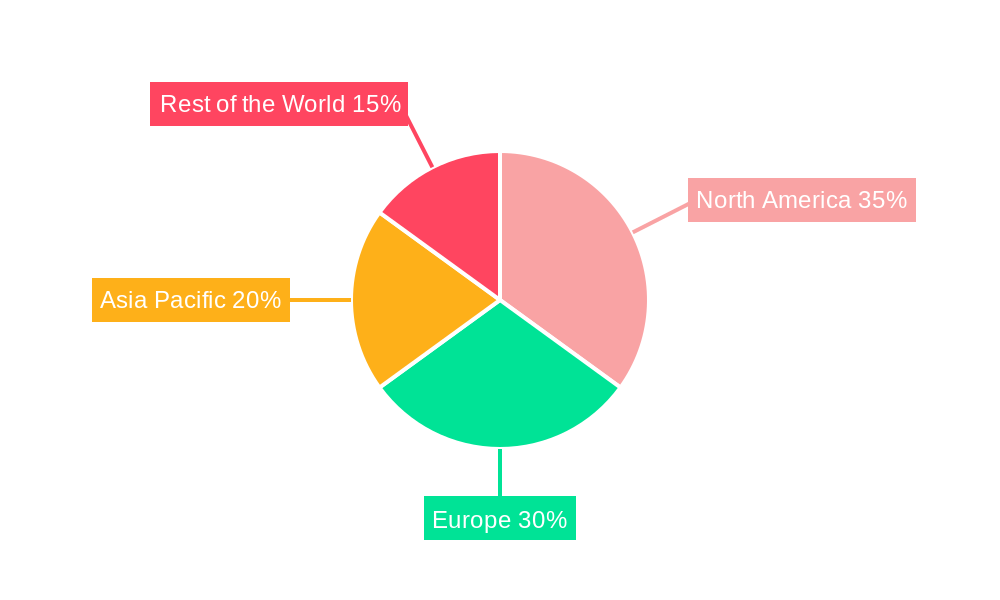
The global personalized cell therapeutic market is witnessing a dynamic interplay of regional dominance and segment leadership. Throughout the study period of 2019-2033, with 2025 as the base and estimated year, and an extensive forecast period of 2025-2033, the North America region, particularly the United States, is projected to emerge as a dominant force. This leadership is underpinned by several factors:
Alongside regional dominance, specific segments are set to drive market growth. Among the Type of cells, Induced Pluripotent Cells (iPSCs) are poised to lead.
In terms of Application, Oncology will continue to be the primary segment driving market revenue, with a projected market value exceeding $80 billion by 2033.
However, the segment of Neurological Disorders is exhibiting the most rapid growth potential, moving from a nascent stage to a significant contributor by the end of the forecast period.
The Other application segment, encompassing rare diseases and chronic conditions beyond cardiovascular and diabetes, will also contribute to market diversification, driven by the inherent flexibility of cell-based approaches. The integration of Other industry developments, such as advancements in ex vivo expansion techniques and sophisticated delivery systems, will further propel the growth and adoption of personalized cell therapeutics across these diverse applications.
The personalized cell therapeutic industry is fueled by several key growth catalysts. The relentless pace of scientific innovation, particularly in cell engineering and gene editing, allows for the development of increasingly sophisticated and effective therapies. A growing global burden of chronic diseases, including oncology and neurological disorders, creates a significant unmet medical need, driving demand for novel treatments. Supportive regulatory frameworks and expedited approval pathways for groundbreaking therapies are also crucial. Furthermore, substantial investment from venture capital and established pharmaceutical companies provides the financial backbone for extensive research and development. Public awareness and patient advocacy are also playing an increasing role in pushing for access to these potentially life-saving treatments.
This comprehensive report delves into the intricate landscape of personalized cell therapeutics, offering an in-depth analysis of market dynamics, technological advancements, and future projections. It meticulously examines the historical trajectory from 2019-2024, providing a solid foundation for understanding the current market status in the estimated year of 2025. The report leverages cutting-edge analytical tools to forecast market growth and identify key trends throughout the extensive forecast period of 2025-2033, projecting a market value reaching an impressive $185.5 billion. Detailed insights into the driving forces, including scientific breakthroughs and increasing demand for advanced treatments, are provided. Furthermore, the report critically assesses the significant challenges and restraints, such as high manufacturing costs and complex regulatory hurdles, that the industry must overcome. The analysis extends to pinpointing the key regions and segments poised for dominance, with a particular focus on the leading role of North America and the burgeoning significance of induced pluripotent cells and oncology applications. It also highlights the rapidly growing potential in neurological disorders. Growth catalysts, leading players, and significant industry developments are meticulously documented, providing a holistic view of this transformative sector. The report serves as an indispensable resource for stakeholders seeking to navigate and capitalize on the opportunities within the rapidly evolving personalized cell therapeutic market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 24.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 24.6%.
Key companies in the market include AbbVie Inc, Teva Pharmaceutical Industries Ltd, Cipla, Lupin, Sun Pharmaceuticals Industries Ltd, Hikma Pharmaceuticals PLC, AstraZeneca, GSK Plc, Pfizer Inc, Amneal Pharmaceuticals LLC, Alvogen, F. Hoffmann-La Roche Ltd, Amgen Inc, Jazz Pharmaceuticals, Inc, Amicus Therapeutics, Inc, MeiraGTx Limited, Rocket Pharmaceuticals, Inc, Gilead Sciences, Inc.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Personalized Cell Therapeutic," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Personalized Cell Therapeutic, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.