1. What is the projected Compound Annual Growth Rate (CAGR) of the Peripheral Specialty Balloon?
The projected CAGR is approximately 6.75%.
 Peripheral Specialty Balloon
Peripheral Specialty BalloonPeripheral Specialty Balloon by Type (Scoring Balloon, Cuttinging Balloon, Chocolate Balloon, Shock Wave Balloon), by Application (Hospitals, Clinics, Outpatient Surgery Centers), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
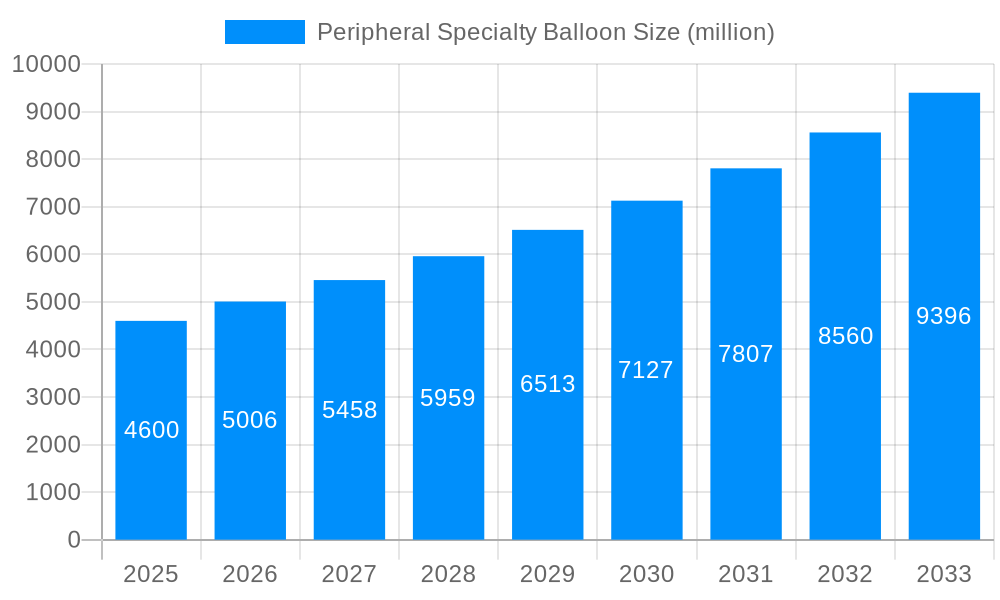
The global Peripheral Specialty Balloon market is poised for significant expansion, projected to reach an estimated USD 4.6 billion by 2025, fueled by a robust Compound Annual Growth Rate (CAGR) of 8.78%. This growth is primarily driven by the increasing prevalence of peripheral artery diseases (PAD) globally, attributed to lifestyle factors such as poor diet, lack of exercise, and rising rates of diabetes and obesity. Advances in interventional cardiology and the development of innovative specialty balloon technologies, including scoring balloons for plaque modification and cutting balloons for precise lesion crossing, are further propelling market demand. These sophisticated devices offer minimally invasive treatment options, leading to reduced patient recovery times and improved clinical outcomes, thereby encouraging wider adoption in hospitals, clinics, and outpatient surgery centers.
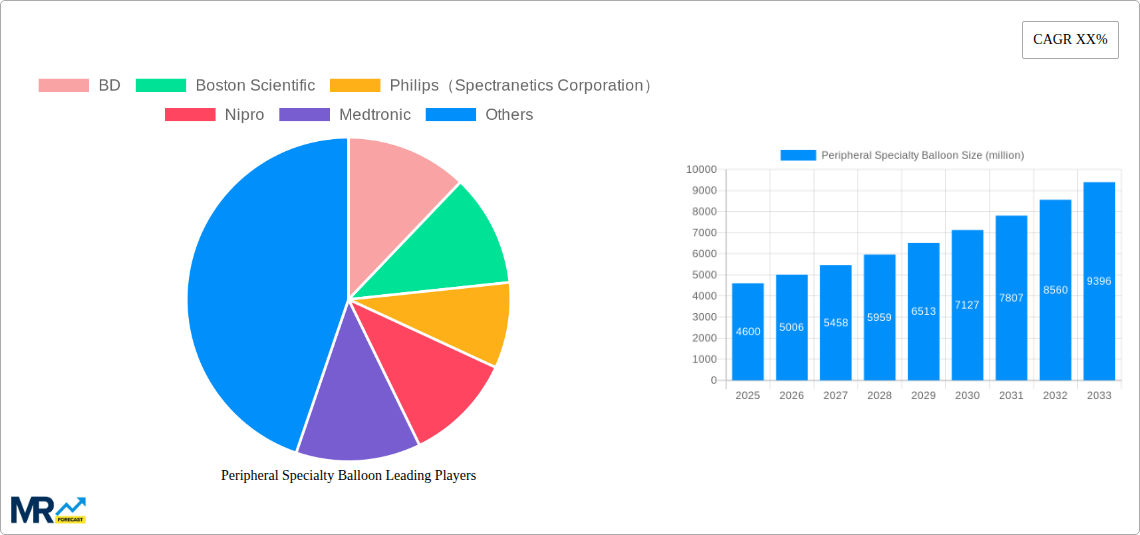

The market dynamics are further shaped by a strong emphasis on technological innovation and strategic collaborations among key players. Companies like BD, Boston Scientific, Philips (Spectranetics Corporation), Medtronic, and Shockwave Medical are actively investing in research and development to introduce next-generation specialty balloons with enhanced efficacy and safety profiles. While the expanding patient pool and technological advancements present substantial opportunities, certain restraints such as the high cost of advanced devices and the need for skilled healthcare professionals could pose challenges. However, the growing healthcare expenditure in emerging economies and the continuous efforts to improve access to advanced cardiovascular treatments are expected to counterbalance these limitations, ensuring sustained market growth throughout the forecast period of 2025-2033. The Asia Pacific region, particularly China and India, is anticipated to emerge as a high-growth area due to increasing healthcare infrastructure development and a burgeoning patient population.

Here's a comprehensive report description on Peripheral Specialty Balloons, incorporating your specified elements:
The global Peripheral Specialty Balloon market is poised for substantial expansion, projected to reach an estimated $7.8 billion by the Base Year of 2025. This robust growth trajectory is further anticipated to continue through the Forecast Period of 2025-2033, painting a picture of a dynamic and increasingly vital segment within interventional cardiology and radiology. XXX The historical period from 2019-2024 witnessed steady adoption, driven by an aging global population, a rising prevalence of peripheral artery disease (PAD), and continuous technological advancements in balloon catheter design and materials. During the Study Period of 2019-2033, the market will undergo significant evolution, characterized by a shift towards less invasive treatment modalities and an increasing demand for specialized balloons that offer enhanced precision and efficacy in complex vascular interventions. The Base Year of 2025 serves as a critical inflection point, with innovative technologies like shock wave balloons and advanced scoring and cutting balloons gaining significant traction, addressing unmet needs in treating challenging lesions. The Estimated Year of 2025 reflects this burgeoning potential, with market players investing heavily in research and development to cater to the growing demand for personalized and effective treatment options for peripheral vascular conditions. The market's expansion is directly linked to the increasing rates of diabetes, hypertension, and obesity, which are primary contributors to the development of PAD, thereby fueling the need for advanced interventional devices. Furthermore, the growing emphasis on improving patient outcomes and reducing hospital stays is driving the adoption of minimally invasive procedures, where specialty balloons play a pivotal role. The increasing healthcare expenditure in emerging economies, coupled with a growing awareness of advanced treatment options, also contributes to the market's upward momentum.
Several key drivers are propelling the Peripheral Specialty Balloon market forward. Foremost is the escalating global burden of Peripheral Artery Disease (PAD), a condition characterized by the narrowing or blockage of arteries in the limbs, most commonly the legs. This chronic disease affects millions worldwide, particularly the elderly and those with underlying risk factors like diabetes, hypertension, and smoking. The increasing prevalence of these comorbidities directly translates into a larger patient pool requiring intervention, and specialty balloons offer crucial tools for effective treatment. Secondly, the continuous wave of technological innovation is a significant catalyst. Manufacturers are relentlessly developing balloons with enhanced features, such as improved flexibility, steerability, and lesion crossing capabilities. The introduction of scoring balloons with precise micro-blades and cutting balloons with excimer laser or micro-razor technology allows for the controlled dissection of fibrotic or calcified lesions, which were previously difficult to treat. Furthermore, the advent of shock wave balloons, which utilize acoustic pressure waves to break down calcified plaque, is revolutionizing the treatment of severely calcified lesions, offering a less traumatic and more effective alternative to traditional angioplasty. The growing preference for minimally invasive procedures over open surgeries, driven by reduced patient trauma, shorter recovery times, and lower healthcare costs, also significantly bolsters the demand for peripheral specialty balloons. This shift towards endovascular interventions aligns perfectly with the capabilities and applications of these advanced balloon technologies.
Despite the robust growth, the Peripheral Specialty Balloon market is not without its challenges and restraints. A primary concern is the high cost associated with some of the more advanced and specialized balloon technologies. This can limit their accessibility, particularly in resource-constrained healthcare settings and emerging economies, where cost-effectiveness is a significant factor. Furthermore, the reimbursement landscape for advanced interventional procedures can be complex and vary by region, potentially impacting physician adoption and patient access. Another significant restraint is the presence of stringent regulatory hurdles. The development and approval of new medical devices, especially those involving novel technologies, require extensive clinical trials and rigorous safety and efficacy evaluations, which can be time-consuming and expensive for manufacturers. Moreover, there is a continuous need for physician training and education to ensure the proper and optimal utilization of these sophisticated devices. Inadequate training can lead to suboptimal outcomes and potentially hinder market penetration. The risk of complications associated with interventional procedures, although generally low with advanced balloons, remains a concern for both patients and healthcare providers. Factors such as restenosis (re-narrowing of the artery) and dissection can necessitate further interventions, impacting patient satisfaction and increasing healthcare costs. Finally, the competitive intensity within the market, with numerous established players and emerging companies, can lead to pricing pressures and necessitate significant investments in marketing and R&D to maintain market share.
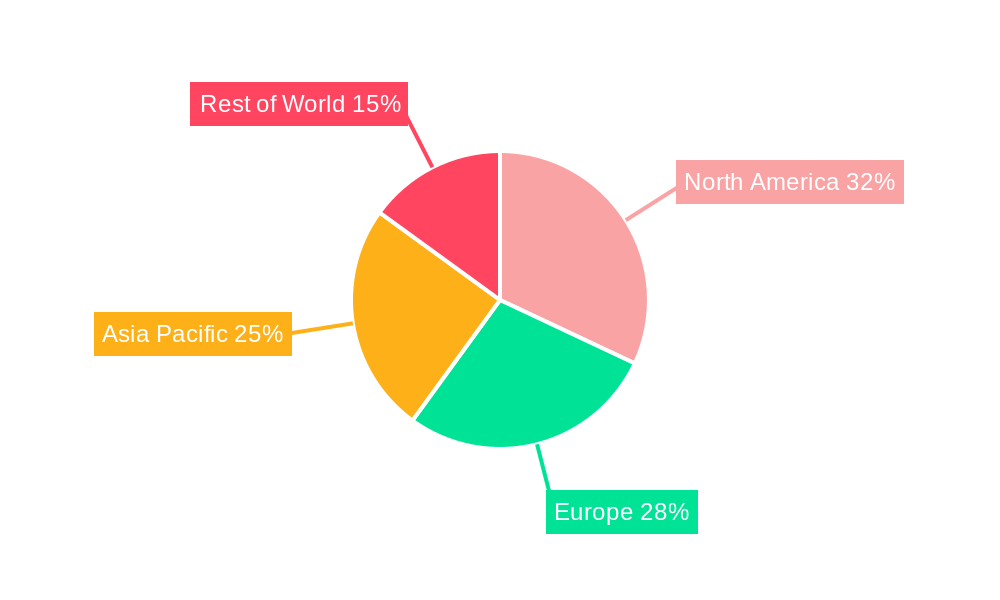
The North America region, driven by the United States, is anticipated to be a dominant force in the Peripheral Specialty Balloon market during the Study Period of 2019-2033. This dominance is underpinned by several factors, including a high prevalence of Peripheral Artery Disease (PAD) due to an aging population, widespread adoption of advanced healthcare technologies, and significant investments in research and development by leading medical device companies. The robust healthcare infrastructure, coupled with favorable reimbursement policies for interventional procedures, further solidifies North America's leading position.
Within this dominant region, the Hospitals segment is expected to be the largest application segment. Hospitals are equipped with the necessary infrastructure, specialized surgical teams, and advanced imaging capabilities to perform complex peripheral interventions. The majority of PAD patients are initially diagnosed and treated in hospital settings, making them the primary point of access for specialty balloon procedures. The availability of a wide range of specialty balloons in hospital formularies and the established protocols for their use further contribute to the segment's dominance.
In terms of balloon types, the Shock Wave Balloon segment is projected to exhibit the most rapid growth and capture significant market share. The increasing prevalence of severely calcified lesions, which are challenging to treat with conventional balloons, is a primary driver for shock wave balloons. Their ability to effectively fracture calcified plaque, facilitate stent delivery, and improve procedural success rates is highly valued by interventionalists. Companies like Shockwave Medical have pioneered this technology and are experiencing substantial market penetration.
The Scoring Balloon segment is also expected to witness considerable growth. These balloons, equipped with micro-blades or scoring elements, provide controlled dissection of fibrotic and calcified lesions, improving angioplasty outcomes and reducing the risk of dissection. Their versatility in treating a wide range of lesion types makes them a valuable tool for interventionalists and contributes to their increasing adoption.
The Cutting Balloon segment, while established, will continue to hold a significant market share due to its effectiveness in treating complex lesions, particularly in the femoropopliteal arteries. Its ability to create controlled micro-incisions in the arterial wall allows for better plaque modification and improved stent deployment.
However, the Chocolate Balloon segment, while offering unique benefits in navigating tortuous anatomy and treating dissections, might experience slower growth compared to shock wave and scoring balloons due to the emergence of more advanced technologies addressing calcified lesions more directly.
The market's expansion in North America is also influenced by the proactive approach of regulatory bodies in approving innovative devices, allowing for faster market entry of new technologies. The presence of a highly skilled workforce of interventional cardiologists and radiologists adept at utilizing these specialized balloons further fuels the demand. The increasing number of outpatient surgery centers catering to less complex procedures also presents a growing avenue for specialty balloons, although hospitals remain the primary setting for comprehensive vascular interventions. The growing awareness among patients about advanced treatment options and the shift towards limb salvage procedures in cases of critical limb ischemia also contribute to the sustained demand for effective peripheral intervention tools.
Several key growth catalysts are shaping the Peripheral Specialty Balloon industry. The rapidly aging global population is a significant demographic driver, as the incidence of PAD escalates with age. Furthermore, the rising prevalence of chronic diseases like diabetes, hypertension, and obesity, all major risk factors for PAD, directly expands the patient pool requiring intervention. Continuous technological advancements, leading to the development of more sophisticated and effective balloons, are also crucial. Innovations such as shock wave, scoring, and advanced angioplasty balloons offer improved lesion crossing, plaque modification, and overall procedural success rates. The growing preference for minimally invasive procedures over traditional open surgeries, due to reduced patient trauma and faster recovery, further fuels the demand for these endovascular tools.
This comprehensive report offers an in-depth analysis of the Peripheral Specialty Balloon market, covering the Study Period of 2019-2033 with a Base Year of 2025. It delves into market dynamics, segmentations, and regional landscapes, providing valuable insights for stakeholders. The report meticulously examines driving forces such as the escalating burden of PAD and continuous technological innovation, while also addressing challenges like high costs and regulatory complexities. Key regions and segments, including the dominance of North America and the significant role of Hospitals and Shock Wave Balloons, are thoroughly explored. Leading players and their contributions are highlighted, alongside significant developments shaping the sector. This report is designed to equip industry participants with the knowledge necessary to navigate this evolving market and capitalize on emerging opportunities.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.75% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6.75%.
Key companies in the market include BD, Boston Scientific, Philips(Spectranetics Corporation), Nipro, Medtronic, Acrostak, Shockwave Medical, BIOTRONIK, Lepu Medical, OrbusNeich, DK Medtech, Sinomed, Brosmed.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Peripheral Specialty Balloon," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Peripheral Specialty Balloon, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.