1. What is the projected Compound Annual Growth Rate (CAGR) of the Pad Printing Ink for Medical Products?
The projected CAGR is approximately 3.2%.
 Pad Printing Ink for Medical Products
Pad Printing Ink for Medical ProductsPad Printing Ink for Medical Products by Type (Solvent-based, UV Curing), by Application (PPE, Medical Device), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
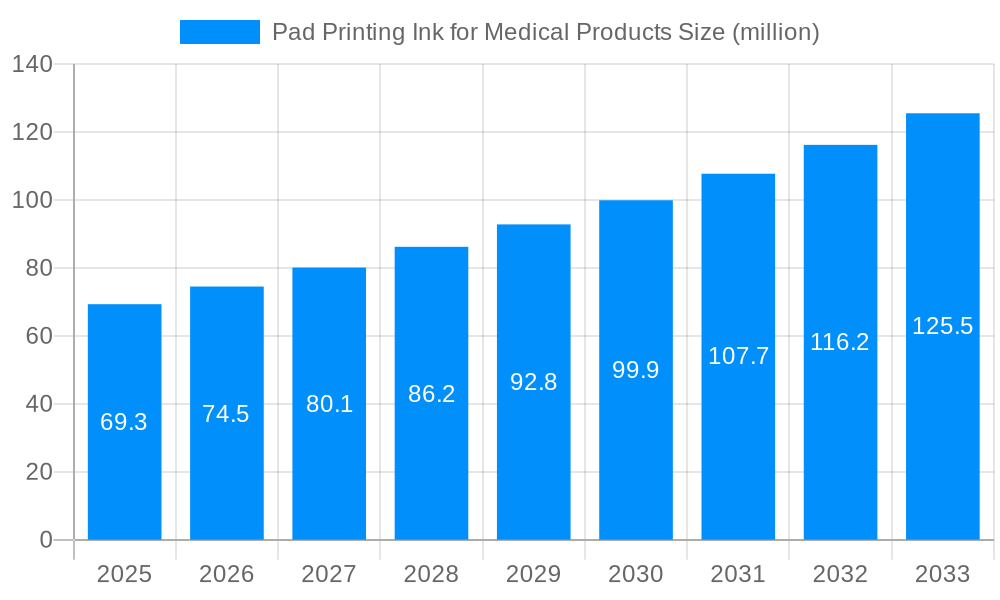
The global Pad Printing Ink for Medical Products market is projected for steady growth, driven by the increasing demand for sophisticated medical devices and enhanced healthcare infrastructure worldwide. With an estimated market size of $69.3 million in the base year of 2025, the sector is poised to expand at a Compound Annual Growth Rate (CAGR) of 3.2% throughout the forecast period from 2025 to 2033. This growth is fundamentally supported by the escalating need for high-precision printing on a variety of medical components, including personal protective equipment (PPE) and intricate medical devices. The inherent requirements for biocompatibility, durability, and resistance to sterilization processes in medical inks necessitate advanced formulations, thereby fostering innovation and market expansion. Technological advancements in printing techniques and ink formulations that offer improved adhesion, chemical resistance, and vibrant color reproduction are key enablers of this upward trajectory.
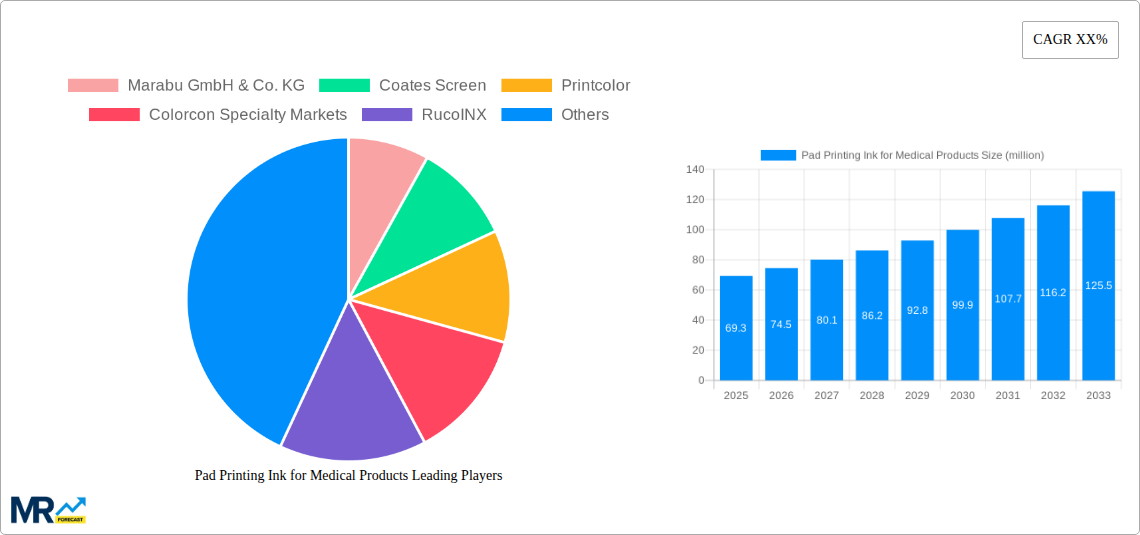

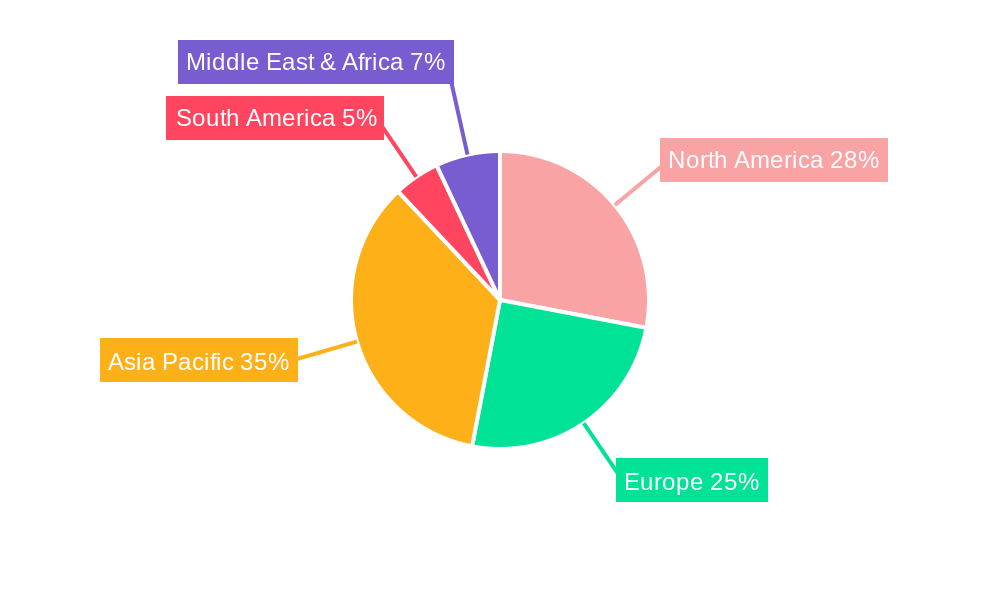
The market dynamics are further shaped by distinct segmentations. In terms of type, both solvent-based and UV curing inks play crucial roles, catering to diverse application needs and regulatory compliance standards within the medical industry. Solvent-based inks offer versatility, while UV curing inks provide rapid drying and enhanced durability, often preferred for their environmental benefits and efficient production cycles. The applications are primarily concentrated on PPE, a segment that has seen significant expansion due to global health concerns, and medical devices, which demand precise and lasting markings for identification, branding, and functional indicators. Leading companies such as Marabu GmbH & Co. KG, Coates Screen, and Inkcups are at the forefront, investing in research and development to meet the stringent quality and safety standards of the medical sector. Geographically, North America and Europe are significant markets, with Asia Pacific emerging as a high-growth region due to its expanding healthcare sector and increasing manufacturing capabilities.

Here is a comprehensive report description for "Pad Printing Ink for Medical Products," incorporating the provided information and structure:
The global pad printing ink market for medical products is poised for significant expansion, with projections indicating a substantial rise in market valuation from an estimated USD 180 million in 2024 to reach an impressive USD 350 million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of approximately 7.2% during the forecast period of 2025-2033. This growth trajectory is underpinned by a confluence of factors, primarily driven by the escalating demand for sophisticated medical devices and the critical need for accurate, durable, and biocompatible marking solutions. The historical period, from 2019 to 2024, witnessed a steady upward trend, setting a robust foundation for future expansion. Key market insights reveal a growing preference for UV curing inks due to their rapid drying times, enhanced durability, and lower environmental impact compared to traditional solvent-based alternatives. The increasing adoption of advanced printing technologies in the healthcare sector, coupled with stringent regulatory requirements for product traceability and patient safety, further fuels the demand for high-performance pad printing inks. The market is also witnessing a paradigm shift towards inks that offer superior adhesion to a diverse range of medical-grade substrates, including plastics, metals, and specialized polymers. Furthermore, the rise in personalized medicine and the need for intricate labeling on smaller, more complex medical components are creating new avenues for innovation and market penetration. The COVID-19 pandemic, while presenting initial supply chain disruptions, also accelerated the demand for certain medical products, particularly Personal Protective Equipment (PPE), thereby indirectly stimulating the market for related printing consumables. The focus on miniaturization and the development of novel drug delivery systems are also expected to contribute to the market's growth in the coming years, as these applications often require precise and indelible markings. The industry is actively investing in research and development to formulate inks that meet evolving biocompatibility standards and offer enhanced resistance to sterilization processes, ensuring the integrity and functionality of medical devices throughout their lifecycle.
Several potent forces are collectively propelling the growth of the pad printing ink market for medical products. Foremost among these is the ever-increasing global demand for advanced medical devices, ranging from intricate surgical instruments and diagnostic equipment to implantable devices and drug delivery systems. As healthcare infrastructure expands and technological innovation accelerates, so too does the need for precise and reliable marking of these critical components. Pad printing offers a versatile and cost-effective solution for applying logos, serial numbers, batch codes, and other essential information onto a wide array of irregularly shaped and often small medical products. Furthermore, stringent regulatory mandates across major healthcare markets worldwide, such as those enforced by the FDA in the United States and the EMA in Europe, necessitate robust traceability and identification systems for medical devices. This regulatory pressure directly translates into a higher demand for high-quality, indelible inks that can withstand various sterilization methods and environmental conditions without compromising legibility or biocompatibility. The growing emphasis on patient safety and the prevention of counterfeit medical products also underscore the importance of secure and verifiable marking solutions, which pad printing inks effectively provide. The increasing adoption of automation in medical manufacturing processes further supports the use of pad printing, as it integrates seamlessly into automated production lines, enhancing efficiency and reducing manual intervention.
Despite the promising growth outlook, the pad printing ink for medical products market faces certain challenges and restraints that could temper its expansion. A significant hurdle is the stringent regulatory landscape surrounding medical product labeling and the materials used in their production. Ensuring that pad printing inks meet rigorous biocompatibility standards and adhere to ISO 10993 guidelines for medical device materials can be a complex and time-consuming process, often requiring extensive testing and certification. This can lead to higher development costs and longer time-to-market for new ink formulations. Furthermore, the evolving nature of sterilization techniques employed for medical devices, including gamma irradiation and ethylene oxide sterilization, demands inks with exceptional resistance and stability. Developing inks that can consistently withstand these harsh processes without degradation or loss of adhesion presents a continuous technical challenge. The cost of raw materials, particularly specialized pigments and resins required for medical-grade inks, can also be a significant restraint, impacting overall ink pricing and profitability. Another factor is the increasing competition from alternative marking technologies, such as laser marking and inkjet printing, which offer different advantages and may be preferred for specific applications or substrates. The need for specialized equipment and trained personnel to operate pad printing machinery can also be a barrier to entry for smaller manufacturers. Lastly, global economic uncertainties and potential disruptions in the supply chain for raw materials can pose risks to the consistent availability and pricing of pad printing inks.
The Medical Device segment, encompassing a vast array of products from diagnostic equipment and surgical tools to implantable devices and drug delivery systems, is projected to be the dominant force in the pad printing ink for medical products market. This dominance stems from the sheer volume and diversity of medical devices manufactured globally, each requiring precise and durable marking for identification, traceability, and regulatory compliance. Within this segment, the UV Curing ink type is expected to witness the most significant growth and hold a substantial market share. UV curing inks offer distinct advantages crucial for medical applications, including rapid curing times that boost production efficiency, superior scratch and chemical resistance essential for product durability, and a significantly lower volatile organic compound (VOC) content, aligning with increasing environmental regulations and workplace safety standards. These inks also provide excellent adhesion to a broad spectrum of medical-grade plastics and metals commonly used in device manufacturing.
Key Regions Dominating the Market:
North America: This region, particularly the United States, stands as a powerhouse in the medical device industry, driven by a robust healthcare system, significant R&D investments, and a highly regulated market that emphasizes product safety and traceability. The presence of numerous leading medical device manufacturers, coupled with a strong demand for advanced healthcare solutions, positions North America as a key driver for pad printing ink adoption. The stringent FDA regulations necessitate reliable marking solutions for a wide range of medical products, further bolstering the demand for specialized inks.
Europe: With a well-established and innovative medical technology sector, countries like Germany, Switzerland, France, and the UK are significant contributors to the global market. Europe’s strong focus on product quality, safety, and sustainability, along with the presence of major players in the pharmaceutical and medical device industries, fuels the demand for high-performance pad printing inks. The unified regulatory framework within the EU also streamlines market access for compliant products.
Asia Pacific: This region is emerging as a rapidly growing market due to the increasing healthcare expenditure, rising disposable incomes, and a burgeoning domestic medical device manufacturing industry in countries like China, India, and South Korea. The cost-effectiveness of manufacturing in these regions attracts global players, leading to a greater demand for printing consumables. Furthermore, government initiatives aimed at improving healthcare access and infrastructure are expected to further stimulate the medical device market and, consequently, the demand for pad printing inks.
Within the Medical Device segment, specific sub-applications such as diagnostic instruments, surgical tools, and wearable medical devices are particularly driving the demand for advanced pad printing solutions. The increasing complexity and miniaturization of these devices require inks that can be applied with high precision and offer exceptional durability to withstand repeated use, cleaning, and sterilization cycles. The shift towards UV Curing inks is largely driven by their ability to meet these stringent requirements, offering rapid curing, enhanced chemical resistance, and a safer environmental profile compared to solvent-based alternatives.
The pad printing ink for medical products industry is experiencing robust growth fueled by several key catalysts. The increasing prevalence of chronic diseases and an aging global population are driving a surge in demand for a wider array of medical devices, from diagnostics to long-term care equipment. This directly translates into a greater need for precise and durable marking solutions. Furthermore, advancements in material science for medical devices, leading to the use of novel polymers and biocompatible metals, require the development of specialized inks that can ensure excellent adhesion and durability on these diverse substrates. Regulatory bodies worldwide are also continuously enhancing their traceability requirements for medical products, making indelible marking a non-negotiable aspect of manufacturing, thus boosting demand for compliant inks.
This report offers a meticulous examination of the pad printing ink for medical products market, providing invaluable insights for stakeholders. The comprehensive analysis delves into market size and forecasts, with estimations reaching USD 350 million by 2033. It meticulously tracks market dynamics, exploring the drivers, restraints, and opportunities shaping the sector from the historical period of 2019-2024 through to the forecast period of 2025-2033. The report provides granular segmentation by ink type (Solvent-based, UV Curing) and application (PPE, Medical Device), highlighting key growth areas and dominant segments. Furthermore, it scrutinizes the competitive landscape, profiling leading players and their strategic initiatives, alongside an overview of significant market developments. The report also sheds light on regional market analysis, identifying key geographic areas with significant growth potential. This in-depth coverage equips businesses with the knowledge to make informed strategic decisions and capitalize on the evolving opportunities within this critical market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 3.2% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 3.2%.
Key companies in the market include Marabu GmbH & Co. KG, Coates Screen, Printcolor, Colorcon Specialty Markets, RucoINX, Inkcups, ITW, Encres DUBUIT, Proell, Tampoprint.
The market segments include Type, Application.
The market size is estimated to be USD 69.3 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Pad Printing Ink for Medical Products," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Pad Printing Ink for Medical Products, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.