1. What is the projected Compound Annual Growth Rate (CAGR) of the Oral Thin-film Drug Delivery?
The projected CAGR is approximately 5.1%.
 Oral Thin-film Drug Delivery
Oral Thin-film Drug DeliveryOral Thin-film Drug Delivery by Type (OTC, Rx), by Application (Hospitals, Drugstores, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
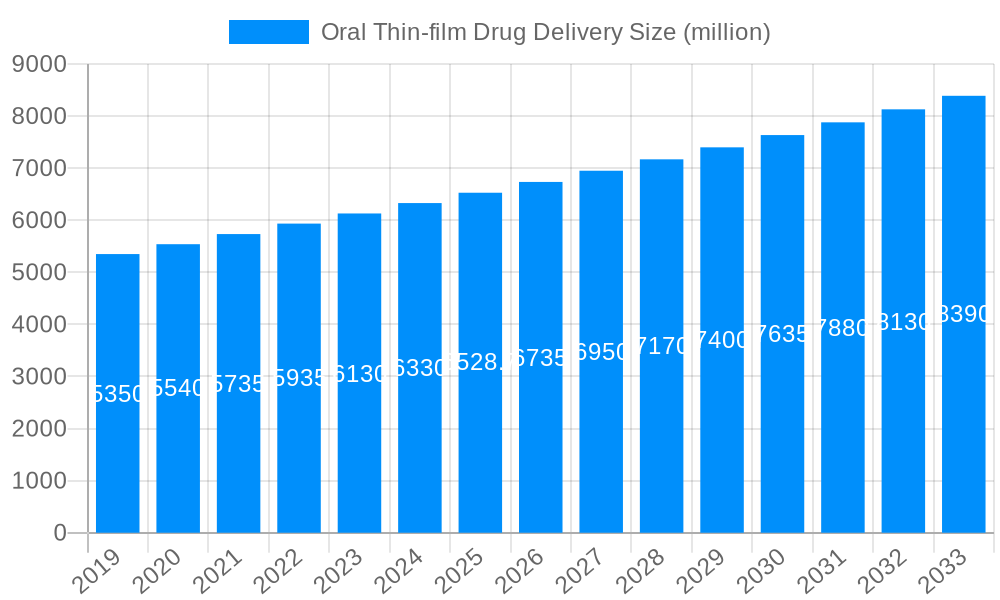
The global Oral Thin-film Drug Delivery market is poised for substantial growth, projected to reach a market size of $6,528.7 million by 2025, with an anticipated Compound Annual Growth Rate (CAGR) of 5.1% through 2033. This robust expansion is driven by the inherent advantages of oral thin films, including rapid drug absorption, improved patient compliance, and discretion, making them an attractive alternative to traditional dosage forms. Key growth drivers include the increasing prevalence of chronic diseases requiring long-term medication, a rising demand for patient-centric drug delivery systems, and ongoing advancements in film formulation and manufacturing technologies. Furthermore, the growing focus on over-the-counter (OTC) medications and the potential for enhanced bioavailability of certain active pharmaceutical ingredients (APIs) are expected to fuel market adoption. The market's segmentation by type into OTC and Rx, and by application into hospitals, drugstores, and others, indicates a diversified demand across various healthcare settings and consumer channels.
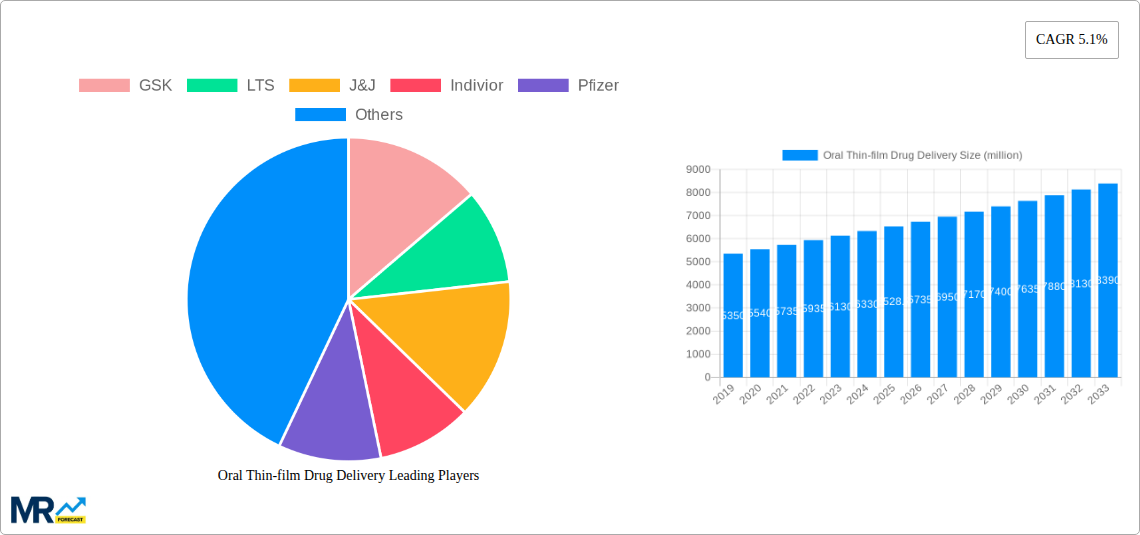

Several influential companies, including GSK, Johnson & Johnson, Indivior, and Pfizer, are actively investing in research and development, along with strategic collaborations and acquisitions, to expand their portfolios and capture a larger market share within the oral thin-film drug delivery space. Emerging trends such as the development of orally disintegrating films for faster onset of action, the incorporation of novel excipients for better drug stability and release profiles, and the exploration of thin films for complex molecules and biologics are shaping the future trajectory of this market. While the market presents significant opportunities, certain restraints, such as the complexity of manufacturing for specific drug molecules and the need for stringent regulatory approvals, may pose challenges. However, the persistent innovation and strategic initiatives by key players are expected to overcome these hurdles, ensuring sustained market expansion and increasing penetration of oral thin-film drug delivery systems in the global pharmaceutical landscape.

This in-depth report offers a meticulous examination of the global Oral Thin-film Drug Delivery market, projecting a robust expansion from approximately \$4,500 million in 2024 to an estimated \$9,800 million by 2033, reflecting a Compound Annual Growth Rate (CAGR) of roughly 9% during the forecast period of 2025-2033. The study encompasses a comprehensive analysis of the market dynamics, from its historical trajectory (2019-2024) through to its future outlook, with 2025 serving as both the base and estimated year for key market assessments. Industry developments, technological advancements, and evolving patient preferences are intricately woven into the analysis, providing a holistic perspective on this burgeoning sector. The report also meticulously details the competitive landscape, highlighting the strategic initiatives and product pipelines of key market participants.
The global Oral Thin-film Drug Delivery market is experiencing a significant and sustained upward trajectory, driven by a confluence of factors that underscore its growing importance in modern pharmaceutical delivery. Over the study period of 2019-2033, the market has witnessed an impressive expansion, with the base year of 2025 projecting a valuation of approximately \$6,500 million, poised for substantial growth to reach an estimated \$9,800 million by 2033. This remarkable growth is largely attributed to the inherent advantages of oral thin films (OTFs) as a drug delivery system. The convenience and ease of administration, requiring no water and dissolving rapidly on the tongue, directly address patient compliance issues, particularly for pediatric and geriatric populations, as well as individuals with dysphagia. This enhanced patient experience translates into improved therapeutic outcomes and a reduced burden on healthcare systems. Furthermore, the discreet nature of OTFs offers a significant advantage in terms of patient acceptance and adherence, especially for sensitive medications. The ability of OTFs to bypass first-pass metabolism in some cases also contributes to improved bioavailability and potentially lower dosages, further enhancing their therapeutic profile. The market's evolution is also marked by continuous innovation in film formulation, leading to the development of films with tailored dissolution rates, improved drug loading capacities, and enhanced stability. This technological advancement is crucial in expanding the therapeutic applications of OTFs across a wider range of drug classes, from simple analgesics to more complex therapeutics. The increasing investment in research and development by major pharmaceutical companies and specialized OTF manufacturers is a testament to the perceived potential of this drug delivery platform. The shift towards personalized medicine and the demand for patient-centric solutions further bolster the growth prospects of OTFs, positioning them as a key component in the future of pharmaceutical dispensing. The market's current valuation, estimated at \$6,500 million in 2025, is expected to see a CAGR of approximately 9% during the forecast period of 2025-2033, underscoring the robust market penetration and adoption anticipated for this innovative drug delivery method.
The relentless expansion of the Oral Thin-film Drug Delivery (OTFD) market is powered by a compelling set of drivers, each contributing to its growing prominence. A primary catalyst is the escalating demand for patient-centric drug delivery systems. The convenience, discretion, and ease of administration offered by OTFD address critical unmet needs, particularly for patient populations who struggle with traditional dosage forms like pills or injections. This improved patient compliance is a significant factor for pharmaceutical companies seeking to maximize therapeutic efficacy and reduce treatment failures. Furthermore, the rapid onset of action that OTFD can provide for certain medications is highly desirable, especially for conditions requiring swift relief. Advances in polymer science and manufacturing technologies have been instrumental in overcoming earlier limitations, enabling the development of films with precise drug release profiles and enhanced stability. This technological evolution allows for the incorporation of a wider range of active pharmaceutical ingredients (APIs), broadening the therapeutic applications of OTFD. The increasing prevalence of chronic diseases, requiring long-term medication adherence, further amplifies the appeal of convenient drug delivery methods like OTFD. The aging global population, with its associated challenges in swallowing pills, also represents a significant demographic driving the adoption of these user-friendly alternatives. Moreover, the growing market for over-the-counter (OTC) medications, where ease of use and immediate accessibility are paramount, provides a fertile ground for OTFD expansion, promising substantial growth from an estimated \$6,500 million in 2025 to an impressive \$9,800 million by 2033.
Despite its promising growth, the Oral Thin-film Drug Delivery (OTFD) market faces several inherent challenges and restraints that temper its expansion. A significant hurdle is the drug loading capacity of OTFs. For potent drugs requiring very low doses, this is less of an issue, but for drugs needing higher dosages, achieving adequate drug loading within the thin film format can be technically challenging and may necessitate larger film sizes, potentially compromising patient acceptance. Taste masking remains a persistent concern, especially for bitter-tasting APIs, as the rapid dissolution on the tongue can expose the patient to unpleasant flavors, necessitating the use of flavor masking agents which can add complexity and cost to the formulation. Stability and shelf-life can also be a constraint, particularly for moisture-sensitive drugs or formulations, requiring careful selection of excipients and packaging to maintain drug integrity over time. Manufacturing costs can also be a restraint. While the simplicity of administration is a patient benefit, the specialized equipment and processes required for film manufacturing can sometimes translate to higher production costs compared to conventional dosage forms, potentially impacting pricing and market penetration, especially in price-sensitive markets. Furthermore, regulatory hurdles associated with novel drug delivery systems can sometimes lead to longer approval timelines, delaying market entry. The development of specific and sensitive analytical methods for quality control of thin films is also an ongoing area of research and development. While the market is projected to grow significantly, these challenges need to be continuously addressed through innovation and strategic partnerships to unlock the full potential of OTFD, which is currently valued at approximately \$6,500 million in 2025.
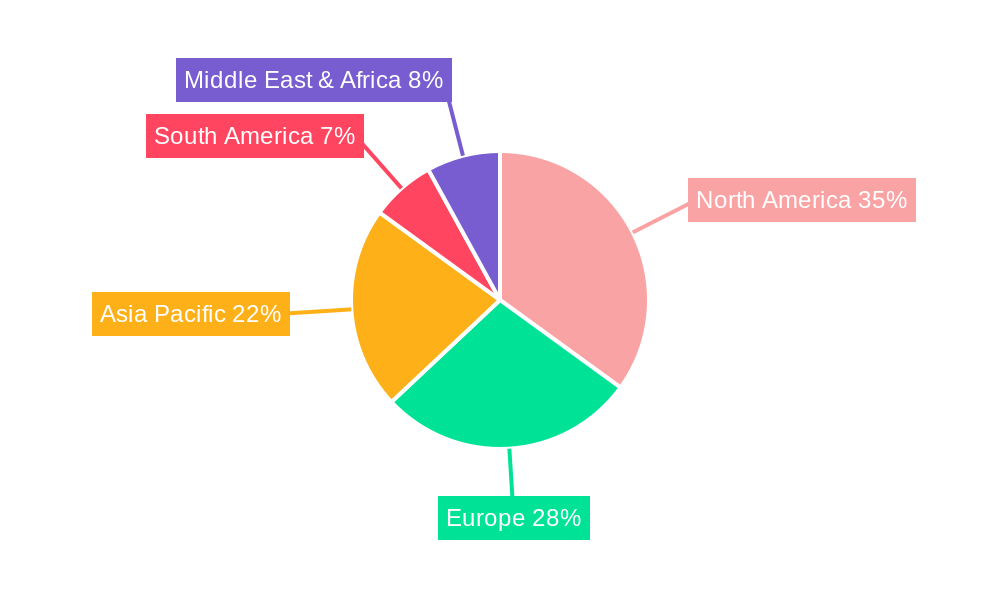
The global Oral Thin-film Drug Delivery market is anticipated to witness dominance by North America and the Rx (Prescription) segment, owing to a synergistic interplay of factors including advanced healthcare infrastructure, high disposable incomes, a robust pharmaceutical research and development ecosystem, and a proactive approach towards adopting novel drug delivery systems.
In North America, particularly the United States, the market's ascendancy is propelled by several key attributes:
The Rx (Prescription) segment is projected to be the dominant force in the OTFD market, driven by the following reasons:
The Oral Thin-film Drug Delivery (OTFD) industry is poised for accelerated growth, driven by several key catalysts. A significant driver is the continuous innovation in film formulation and manufacturing technologies. This includes advancements in polymers, drug encapsulation techniques, and specialized manufacturing processes that enable higher drug loading, improved dissolution profiles, and enhanced stability. Furthermore, the increasing focus on patient-centric healthcare solutions and the growing demand for user-friendly drug delivery methods are creating a substantial market pull for OTFs. The expansion of the OTFD application portfolio into new therapeutic areas, beyond its traditional uses, is another major growth catalyst. This includes novel applications in areas like ophthalmology, otology, and even transdermal delivery through specialized film designs.
This comprehensive report provides a deep dive into the Oral Thin-film Drug Delivery market, projecting its significant growth from an estimated \$6,500 million in 2025 to an impressive \$9,800 million by 2033, with a CAGR of approximately 9% during the forecast period of 2025-2033. It meticulously analyzes the market's historical trends (2019-2024) and future projections, offering valuable insights into market dynamics, technological advancements, and strategic initiatives of key players. The report also elaborates on the driving forces, including the rising demand for patient convenience and adherence, and the challenges such as drug loading capacity and taste masking. It thoroughly investigates the dominant regions and segments, with a particular focus on North America and the Rx segment, explaining their pivotal roles in market expansion. The report is an indispensable resource for stakeholders seeking to understand the current landscape and future trajectory of the Oral Thin-film Drug Delivery market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.1% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.1%.
Key companies in the market include GSK, LTS, J&J, Indivior, Pfizer, Church & Dwight, tesa Labtec, Tapemark, Prestige, MonoSol, BioDelivery, Arx, ZIM, NAL Pharma, AdhexPharma, Aavishkar, IntelGenx Corp, APR, .
The market segments include Type, Application.
The market size is estimated to be USD 6528.7 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "Oral Thin-film Drug Delivery," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Oral Thin-film Drug Delivery, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.