1. What is the projected Compound Annual Growth Rate (CAGR) of the NTRK Fusion Gene Positive Advanced Solid Tumor?
The projected CAGR is approximately 11.48%.
 NTRK Fusion Gene Positive Advanced Solid Tumor
NTRK Fusion Gene Positive Advanced Solid TumorNTRK Fusion Gene Positive Advanced Solid Tumor by Type (/> Therapeutics, Diagnostics), by Application (/> Hospitals and Clinics, Cancer Centers, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The market for NTRK Fusion Gene Positive Advanced Solid Tumors is poised for substantial growth, projected to reach an estimated \$11.13 billion by 2025, driven by a robust CAGR of 11.48%. This expansion is primarily fueled by advancements in targeted therapies and diagnostic tools that enable precise identification of NTRK gene fusions. The increasing prevalence of these specific genetic alterations across various advanced solid tumors, coupled with a growing understanding of their therapeutic implications, is a significant driver. Furthermore, the development of novel diagnostic assays, including next-generation sequencing (NGS) and immunohistochemistry (IHC), is expanding the diagnostic landscape and improving patient stratification, thereby boosting market demand. The growing emphasis on personalized medicine and the subsequent increase in clinical trials investigating NTRK inhibitors further contribute to market momentum. This dynamic landscape is characterized by a strong focus on early detection and tailored treatment strategies, which are crucial for improving patient outcomes in complex oncological cases.
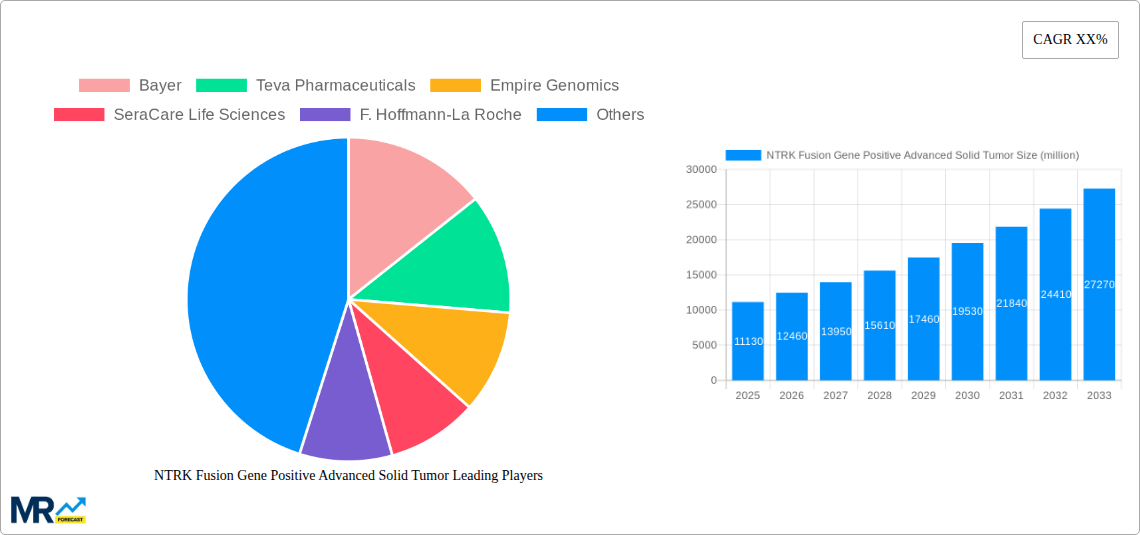

The market is segmented into therapeutics and diagnostics, with both segments experiencing significant innovation. Therapeutics, particularly NTRK inhibitors, represent a major area of growth, offering highly effective treatment options for patients with NTRK fusion-positive tumors. Diagnostics, including advanced molecular testing, are critical for patient identification and are witnessing rapid adoption in hospitals, clinics, and specialized cancer centers. While North America currently leads the market, driven by early adoption of precision oncology and a strong research infrastructure, the Asia Pacific region is expected to exhibit the fastest growth due to increasing healthcare investments, rising cancer incidence, and a burgeoning market for advanced diagnostics. Restraints include the relatively rare incidence of NTRK fusions in the overall cancer population, which can pose challenges for market penetration, and the high cost associated with advanced diagnostic tests and targeted therapies. However, ongoing research, increasing regulatory approvals, and strategic collaborations among key players are expected to overcome these challenges, ensuring sustained market expansion and improved patient care.
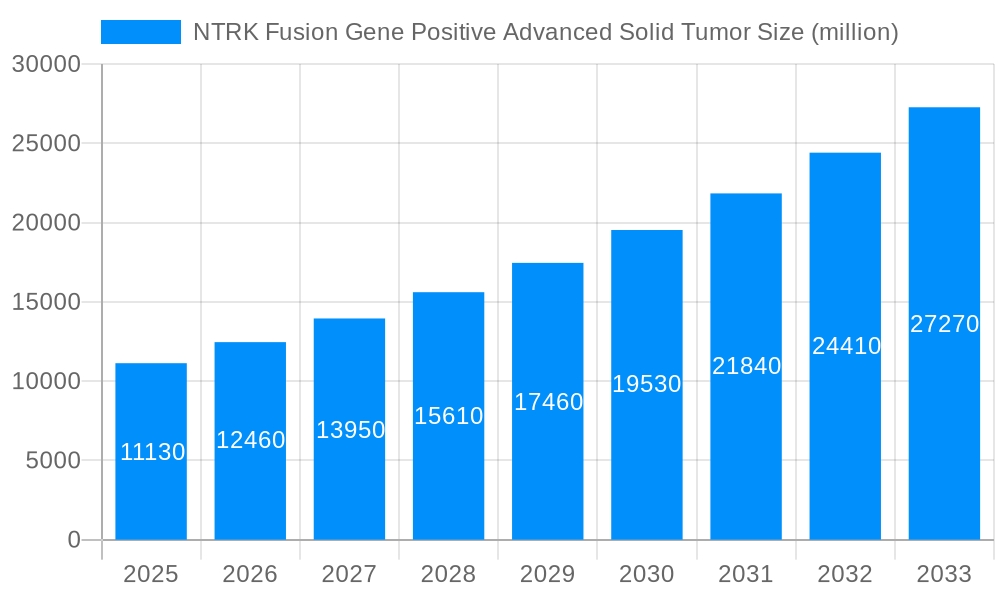

This report offers an in-depth analysis of the global NTRK Fusion Gene Positive Advanced Solid Tumor market, providing a comprehensive overview for stakeholders. Spanning a Study Period of 2019-2033, with a Base Year and Estimated Year of 2025, and a Forecast Period of 2025-2033, this study leverages data from the Historical Period of 2019-2024 to project future market trajectories. The report delves into market dynamics, key growth drivers, existing challenges, and emerging opportunities, offering actionable insights for strategic decision-making. The estimated market size for NTRK Fusion Gene Positive Advanced Solid Tumor is projected to reach $5.5 billion by 2025, with significant growth anticipated in the coming years.
The landscape of NTRK fusion gene-positive advanced solid tumors is experiencing a transformative shift, driven by advancements in precision oncology and the growing understanding of actionable genetic alterations. The increasing incidence of rare tumor types harboring NTRK fusions, coupled with a rising global cancer burden, is creating a fertile ground for market expansion. Over the Study Period of 2019-2033, we anticipate a significant CAGR, pushing the market value well beyond the $5.5 billion mark by 2033. Key trends shaping this market include the widespread adoption of next-generation sequencing (NGS) technologies for comprehensive genomic profiling, leading to earlier and more accurate identification of NTRK fusions. This diagnostic evolution is directly fueling the demand for targeted therapeutics. The efficacy demonstrated by TRK inhibitors in a broad range of NTRK-fusion-positive tumors, regardless of their tissue of origin, has revolutionized treatment paradigms, moving away from histology-specific approaches. This "tumor-agnostic" strategy is a cornerstone of precision medicine and is unlocking new treatment avenues for patients with previously limited options. Furthermore, the growing emphasis on personalized medicine, supported by favorable reimbursement policies and increasing payer acceptance of biomarker-driven therapies, is accelerating market growth. The proactive approach of pharmaceutical companies in developing and launching novel TRK inhibitors, alongside the expansion of diagnostic testing infrastructure, are all contributing to a dynamic and rapidly evolving market. The market is also witnessing an increasing focus on early detection and screening initiatives, particularly within high-risk populations or those with suspected rare cancers, further solidifying the growth trajectory. The growing awareness among oncologists and patients about the significance of NTRK fusions as a therapeutic target is a crucial factor driving market penetration.
Several potent forces are actively propelling the growth of the NTRK fusion gene-positive advanced solid tumor market. Foremost among these is the remarkable clinical efficacy of TRK inhibitors. Drugs like larotrectinib and entrectinib have demonstrated exceptional response rates across a diverse array of NTRK-fusion-positive solid tumors, establishing them as cornerstone therapies and driving significant demand. This success story is a powerful testament to the precision medicine approach. Secondly, the rapid and widespread adoption of advanced diagnostic technologies, particularly next-generation sequencing (NGS), has been instrumental. NGS allows for the simultaneous detection of multiple genetic alterations, including NTRK fusions, in a cost-effective and efficient manner, making comprehensive genomic profiling more accessible to a broader patient population and contributing an estimated $1.2 billion to the diagnostics segment by 2025. This diagnostic capability directly translates to more patients being identified as candidates for targeted therapy. Thirdly, the increasing prevalence of rare and pediatric cancers, many of which are known to harbor NTRK fusions, is creating a significant unmet medical need that targeted therapies are uniquely positioned to address. The unique nature of these fusions, often present in less common tumor types, amplifies the impact of effective targeted treatments. The growing understanding of the molecular underpinnings of cancer, coupled with robust research and development efforts from leading pharmaceutical and diagnostic companies, is constantly expanding the therapeutic arsenal and diagnostic capabilities. This ongoing innovation is a critical engine of market expansion.
Despite the promising growth trajectory, the NTRK fusion gene-positive advanced solid tumor market faces several significant challenges and restraints that could temper its expansion. A primary concern is the rarity of NTRK fusions. While their identification is increasing, they still represent a small fraction of the overall cancer patient population, limiting the addressable market size compared to more common oncological indications, potentially impacting market revenue by an estimated $800 million annually if not addressed. This necessitates highly efficient diagnostic strategies to identify these rare events effectively. Another significant hurdle is the high cost of targeted therapies and advanced diagnostic testing. TRK inhibitors and comprehensive genomic profiling services are often expensive, posing a barrier to access for many patients and healthcare systems, especially in resource-limited regions. The reimbursement landscape, while improving, can still be complex and variable across different countries and insurance providers. Furthermore, the potential for acquired resistance to TRK inhibitors is a growing concern. While initial responses can be dramatic, some patients may develop resistance over time, necessitating the development of next-generation therapies or combination strategies. The limited awareness and understanding among some healthcare professionals regarding NTRK fusions and their implications for treatment can also be a bottleneck. Educating oncologists, pathologists, and other clinicians about the importance of molecular testing and the specific benefits of TRK inhibitors is crucial for market penetration. Finally, the regulatory hurdles and complexities in approving new diagnostic tests and therapies, especially for rare diseases, can lead to prolonged development and launch timelines, impacting market availability.
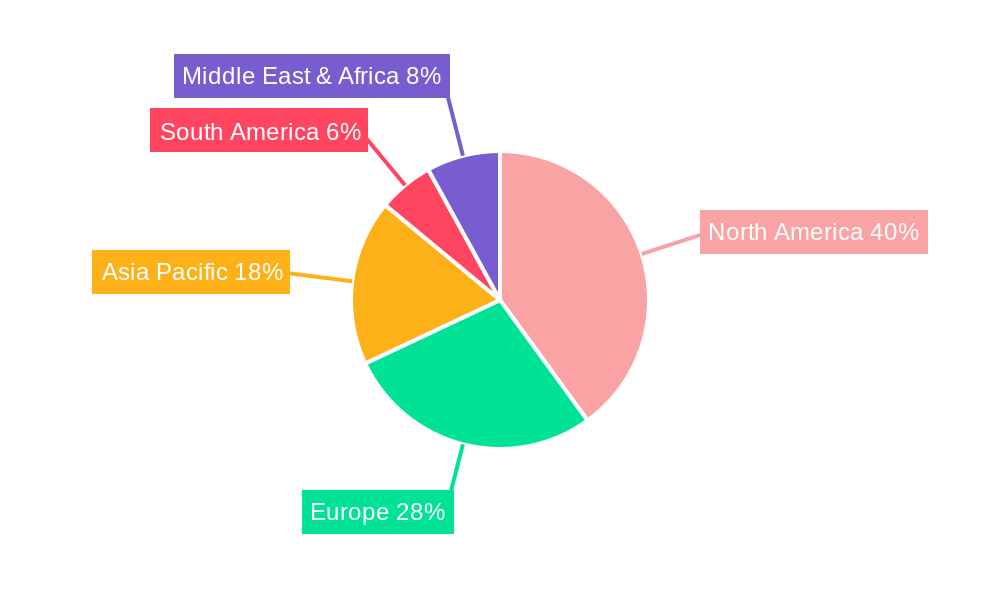
The NTRK Fusion Gene Positive Advanced Solid Tumor market is poised for significant growth across various regions and segments, with certain areas and sectors expected to lead the charge.
Dominant Segments:
Therapeutics: This segment is anticipated to be the primary revenue driver within the NTRK fusion gene-positive advanced solid tumor market. The development and increasing adoption of highly effective TRK inhibitors are central to this dominance. Companies like F. Hoffmann-La Roche, Bayer, and Daiichi Sankyo are investing heavily in novel TRK inhibitors and their pipeline candidates, aiming to capture a significant share of this growing market. The success of existing drugs has paved the way for a robust pipeline of next-generation inhibitors and combination therapies, further solidifying the therapeutics segment's leading position. The market for therapeutics is projected to reach an estimated $3.8 billion by 2025, with steady growth expected thereafter.
Diagnostics: The diagnostics segment is the critical enabler of the therapeutics market. The increasing demand for comprehensive genomic profiling (CGP) using next-generation sequencing (NGS) is fueling the growth of this segment. Companies such as SeraCare Life Sciences, NeoGenomics Laboratories, and Empire Genomics are at the forefront of developing and providing advanced diagnostic solutions, including assay kits and testing services, to accurately identify NTRK fusions. The expanding awareness of NTRK fusions as actionable targets is driving higher utilization of these diagnostic tools, making it an indispensable part of the treatment pathway. The diagnostics segment is expected to contribute an estimated $1.7 billion to the market by 2025.
Dominant Regions/Countries:
North America: This region is expected to maintain its leadership position in the NTRK fusion gene-positive advanced solid tumor market. Factors contributing to this dominance include:
Europe: Europe is another significant market for NTRK fusion gene-positive advanced solid tumors, driven by:
Application Segment Dominance:
The interplay between these dominant segments and regions, driven by technological innovation and a deeper understanding of NTRK fusions, will shape the future of the NTRK fusion gene-positive advanced solid tumor market.
The growth of the NTRK fusion gene-positive advanced solid tumor industry is being significantly catalyzed by several key factors. The continuous innovation and expansion of diagnostic technologies, particularly next-generation sequencing (NGS), are crucial. This enables more precise and accessible identification of NTRK fusions across a broader range of rare and common cancers. Furthermore, the unwavering clinical success of TRK inhibitors in demonstrating high response rates and durable benefits in NTRK-fusion-positive patients is a powerful driver, encouraging wider adoption and investment. The increasing regulatory approvals and favorable reimbursement landscapes for both diagnostics and therapeutics are also removing significant barriers to access and accelerating market penetration.
This report provides unparalleled comprehensive coverage of the NTRK Fusion Gene Positive Advanced Solid Tumor market, offering a 360-degree view for industry participants. It delves deep into market dynamics, meticulously analyzing trends, growth drivers, and challenges. Our analysis extends to a granular examination of key regions and segments, identifying areas of dominance and future potential. The report further highlights significant industry developments, providing a historical context and a forward-looking perspective on innovation. With a rigorous methodology and extensive data analysis, this report equips stakeholders with the insights necessary to navigate this complex and rapidly evolving market, ensuring strategic decision-making and maximizing opportunities for growth and patient benefit.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.48% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 11.48%.
Key companies in the market include Bayer, Teva Pharmaceuticals, Empire Genomics, SeraCare Life Sciences, F. Hoffmann-La Roche, NeoGenomics Laboratories, Glaxosmithkline, OncoDNA, Merck & Co., Daiichi Sankyo, S Deciphera Pharmaceuticals.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "NTRK Fusion Gene Positive Advanced Solid Tumor," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the NTRK Fusion Gene Positive Advanced Solid Tumor, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.