1. What is the projected Compound Annual Growth Rate (CAGR) of the Non-PVC Infusion Set?
The projected CAGR is approximately 10.7%.
 Non-PVC Infusion Set
Non-PVC Infusion SetNon-PVC Infusion Set by Type (TPE, Thermoplastic Polyurethane (TPU), Others, World Non-PVC Infusion Set Production ), by Application (Hospital, Clinic, Others, World Non-PVC Infusion Set Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Non-PVC Infusion Set market is projected for significant expansion, currently valued at $14.37 billion. This growth is primarily driven by the increasing demand for safer, high-performance medical devices and a regulatory push to phase out PVC-based products due to DEHP plasticizer concerns. The adoption of alternative materials like Thermoplastic Polyurethane (TPU) is rising due to their superior biocompatibility, flexibility, and chemical resistance. An expanding healthcare sector, particularly in emerging economies, coupled with an aging global population, is also fueling the need for advanced infusion therapies and non-PVC alternatives, enhancing patient safety and reducing long-term health risks.
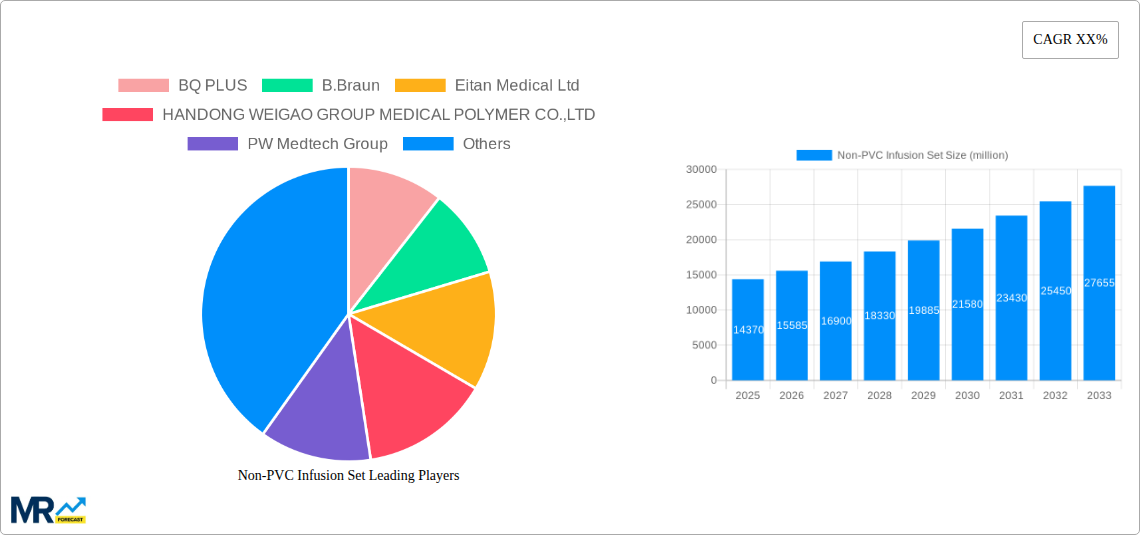

Key industry players are actively innovating and expanding their product portfolios. The market is forecast to experience a robust Compound Annual Growth Rate (CAGR) of approximately 10.7%. Advancements in material science are expected to yield more cost-effective and high-performance non-PVC infusion sets. Emerging markets in the Asia Pacific and Rest of the World regions are anticipated to be key growth centers, supported by improving healthcare infrastructure and increasing disposable incomes. While initial manufacturing costs and regulatory approvals present challenges, these are being mitigated by technological progress and the paramount importance of patient safety, ensuring a positive market outlook.
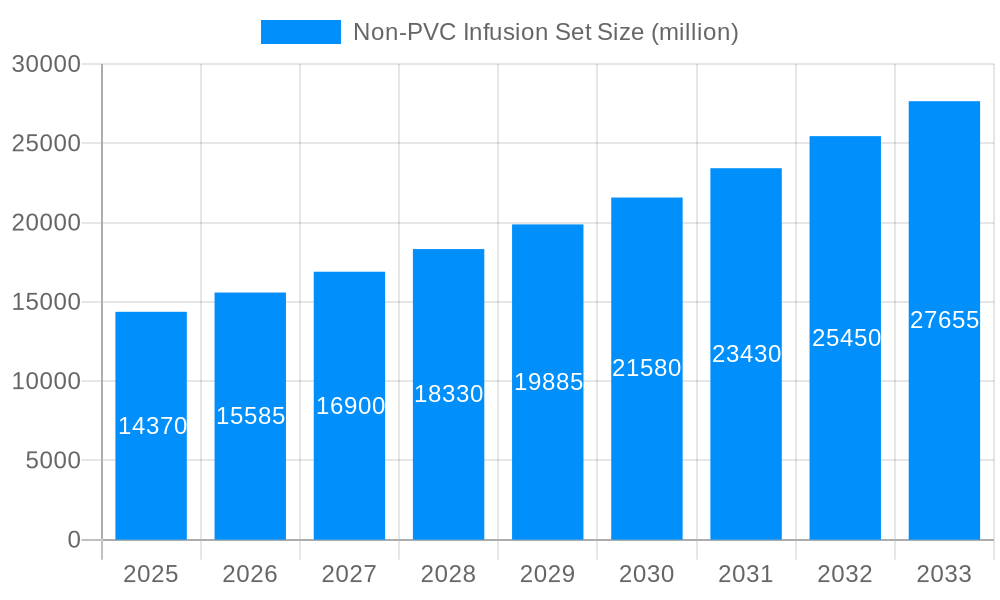

This report provides an in-depth analysis of the global Non-PVC Infusion Set market, offering critical insights for stakeholders. The study covers the base year of 2025 and includes a forecast period to 2033. We analyze production volumes and market dynamics, utilizing millions of units for production figures. This report is essential for understanding market trends, growth drivers, restraints, and identifying dominant regions and segments within the Non-PVC Infusion Set industry.
The global Non-PVC Infusion Set market is experiencing a significant paradigm shift, driven by an increasing awareness of the health and environmental implications associated with traditional PVC-based medical devices. XXX. The demand for non-PVC alternatives is soaring as healthcare providers and regulatory bodies prioritize patient safety and sustainability. This surge is largely attributed to the inherent toxicity of plasticizers like DEHP, commonly used in PVC, which can leach into intravenous fluids and pose potential health risks, including endocrine disruption and adverse effects on organ development. Consequently, there's a palpable move towards materials like Thermoplastic Elastomers (TPE) and Thermoplastic Polyurethane (TPU), which offer superior biocompatibility and reduced environmental impact. Production of Non-PVC Infusion Sets is projected to witness robust growth, with estimates suggesting an expansion from an estimated 250 million units in 2025 to well over 450 million units by 2033. This growth trajectory is underpinned by increasing healthcare expenditure, a rising prevalence of chronic diseases necessitating long-term infusion therapies, and a growing preference for single-use, disposable medical supplies. Furthermore, technological advancements in material science are leading to the development of more advanced, cost-effective, and functionally superior non-PVC infusion sets, further stimulating market penetration. The market is also observing a trend towards specialized infusion sets designed for specific drug delivery systems, such as chemotherapy drugs and biologics, where material compatibility is paramount. The shift away from PVC is not merely a trend but a fundamental reorientation of the medical device industry towards safer and more responsible manufacturing practices, directly influencing production strategies and investment decisions across the value chain.
The Non-PVC Infusion Set market is being propelled by a confluence of powerful factors, chief among them being the escalating global demand for safer and more biocompatible medical devices. The increasing awareness among healthcare professionals and patients about the potential health hazards associated with DEHP leaching from PVC infusion sets is a primary driver. This concern is amplified by regulatory pressures and initiatives from health organizations worldwide to restrict or ban the use of DEHP, pushing manufacturers towards alternative materials. Furthermore, the growing prevalence of chronic diseases and an aging global population necessitate longer and more frequent infusion therapies, thereby increasing the overall demand for infusion sets. The development of advanced non-PVC materials like TPE and TPU, which offer improved flexibility, clarity, and chemical resistance, is also a significant catalyst. These materials not only address safety concerns but also enhance the functionality and performance of infusion sets. The expanding healthcare infrastructure, particularly in emerging economies, coupled with increasing healthcare spending, is creating a larger market for disposable medical devices, including non-PVC infusion sets. Moreover, the environmental sustainability aspect of non-PVC materials, being often recyclable or incinerable with fewer toxic byproducts, aligns with the growing global emphasis on eco-friendly healthcare practices, further bolstering their adoption. The robust production of non-PVC infusion sets, anticipated to grow from an estimated 250 million units in 2025 to over 450 million units by 2033, directly reflects these driving forces.
Despite the robust growth prospects, the Non-PVC Infusion Set market faces several challenges and restraints that could temper its expansion. A primary hurdle is the higher cost of raw materials for non-PVC alternatives like TPE and TPU compared to traditional PVC. This increased production cost can translate into higher prices for the final product, potentially limiting adoption in cost-sensitive markets or healthcare systems with budget constraints. Established manufacturing infrastructure and investment in PVC production also pose a challenge, as transitioning to new materials requires significant capital expenditure for new machinery, process re-engineering, and employee retraining. Perceived performance differences and long-term clinical data for some non-PVC materials, especially when compared to the decades of established use of PVC, can lead to hesitancy among some healthcare providers. While biocompatibility is a major advantage, ensuring equivalent drug compatibility and long-term stability for a wide range of pharmaceuticals within non-PVC materials requires rigorous testing and validation. Regulatory hurdles and the time taken for new material approvals can also slow down market entry and adoption of novel non-PVC infusion sets. Furthermore, supply chain complexities and the availability of raw materials for non-PVC alternatives can be a concern, particularly as demand surges. The global production of Non-PVC Infusion Sets, projected to increase significantly, will need to navigate these challenges to achieve its full potential, with market growth potentially being capped at around 450 million units by 2033 if these restraints are not adequately addressed.
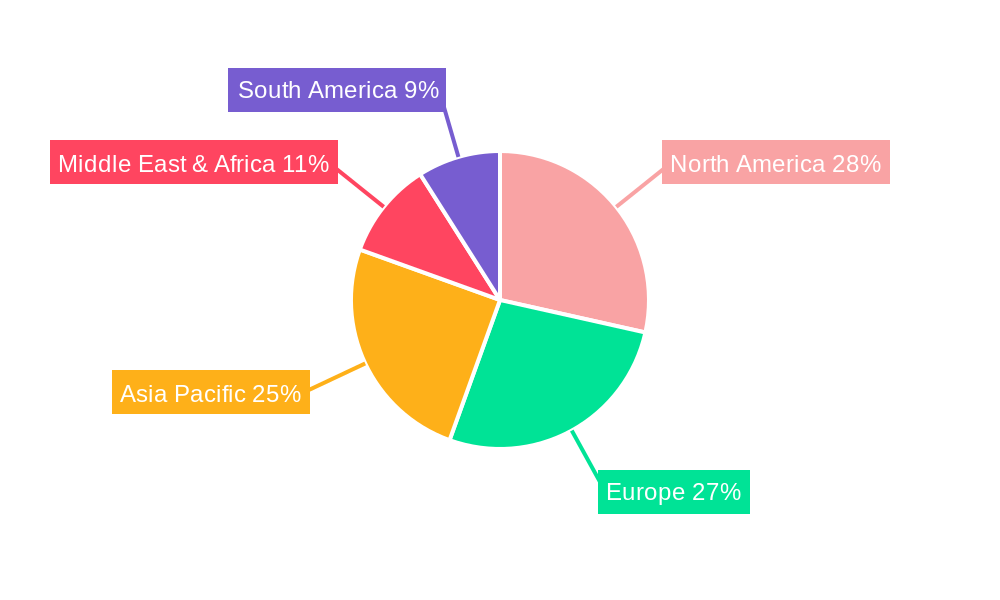
The global Non-PVC Infusion Set market is poised for significant regional and segmental dominance, with North America and Europe expected to lead due to their advanced healthcare infrastructure, stringent regulatory environments, and high patient awareness regarding healthcare product safety. The United States, in particular, is anticipated to be a major consumer, driven by a strong emphasis on patient outcomes and the proactive adoption of innovative medical technologies.
Dominant Regions/Countries:
Dominant Segments:
Type: Thermoplastic Polyurethane (TPU)
Application: Hospital
World Non-PVC Infusion Set Production:
The Non-PVC Infusion Set industry is experiencing powerful growth catalysts. The primary driver is the escalating global health consciousness and regulatory pressure to phase out harmful plasticizers like DEHP from medical devices. This is complemented by the increasing prevalence of chronic diseases and an aging population, which leads to a greater demand for long-term infusion therapies. Furthermore, advancements in material science have yielded superior, safer, and more cost-effective non-PVC alternatives like TPE and TPU, enhancing product performance and patient comfort. The growing healthcare expenditure, particularly in emerging economies, and the expansion of healthcare infrastructure are creating new markets and opportunities for these advanced infusion sets.
This report offers an unparalleled depth of analysis into the Non-PVC Infusion Set market, providing stakeholders with a comprehensive understanding of its current landscape and future trajectory. It meticulously details market sizes, production volumes (in millions of units), segmentation by material type (TPE, TPU, Others) and application (Hospital, Clinic, Others), and regional dynamics. The report delves into the intricate interplay of driving forces, challenges, and growth catalysts that shape the industry. With a detailed historical analysis from 2019-2024, a robust base year of 2025, and an extensive forecast period extending to 2033, this report equips businesses with the critical data and insights needed for strategic decision-making, market entry, and competitive positioning in the ever-evolving Non-PVC Infusion Set sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.7% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 10.7%.
Key companies in the market include BQ PLUS, B.Braun, Eitan Medical Ltd, HANDONG WEIGAO GROUP MEDICAL POLYMER CO.,LTD, PW Medtech Group, Shandong Xinhua Pharmaceutical Company Limited, TianJin Hanaco Medical Co., Ltd, .
The market segments include Type, Application.
The market size is estimated to be USD 2.44 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Non-PVC Infusion Set," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Non-PVC Infusion Set, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.