1. What is the projected Compound Annual Growth Rate (CAGR) of the Non-Alcoholic Steatohepatitis Biomarkers?
The projected CAGR is approximately XX%.
 Non-Alcoholic Steatohepatitis Biomarkers
Non-Alcoholic Steatohepatitis BiomarkersNon-Alcoholic Steatohepatitis Biomarkers by Type (/> Hepatic Fibrosis Biomarkers, Serum Biomarkers, Oxidative Stress Biomarkers, Apoptosis Biomarkers, Others), by Application (/> Hospital, Pharmaceutical Company, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Non-Alcoholic Steatohepatitis (NASH) biomarkers market is experiencing robust growth, projected to reach a substantial market size of approximately $1,200 million by 2025. This expansion is driven by the increasing prevalence of obesity and type 2 diabetes, key risk factors for NASH, and a heightened awareness of the disease's potential to progress to cirrhosis and hepatocellular carcinoma. The market is expected to witness a Compound Annual Growth Rate (CAGR) of around 15% from 2025 to 2033, indicating a strong and sustained upward trajectory. This growth is further fueled by significant investments in research and development by leading pharmaceutical and diagnostic companies, aimed at identifying and validating novel biomarkers for early and accurate NASH diagnosis, staging, and treatment response monitoring. The demand for non-invasive diagnostic tools is paramount, as liver biopsy, the current gold standard, is invasive, costly, and carries associated risks. Consequently, the development and commercialization of advanced serum, hepatic fibrosis, and oxidative stress biomarkers are at the forefront of market innovation, catering to the urgent need for more accessible and patient-friendly diagnostic solutions.


The market landscape for NASH biomarkers is characterized by a dynamic competitive environment with key players actively engaged in strategic collaborations, mergers, and acquisitions to expand their product portfolios and geographical reach. Companies like Genfit, Prometheus Laboratories, Siemens Medical Solutions USA, and Quest Diagnostics are at the forefront, offering a range of diagnostic solutions and actively developing new technologies. The market is segmented into various types of biomarkers, including Hepatic Fibrosis Biomarkers, Serum Biomarkers, and Oxidative Stress Biomarkers, each playing a crucial role in different aspects of NASH management. Application-wise, hospitals and pharmaceutical companies represent the primary end-users, leveraging these biomarkers for patient diagnosis, clinical trial stratification, and drug development. Geographically, North America and Europe currently dominate the market due to established healthcare infrastructures and higher spending on research and diagnostics. However, the Asia Pacific region is poised for significant growth, driven by a rapidly increasing patient population and improving healthcare access. The market is poised for significant advancements with ongoing clinical trials and the anticipated approval of new NASH therapies, which will likely boost the demand for effective diagnostic biomarkers.
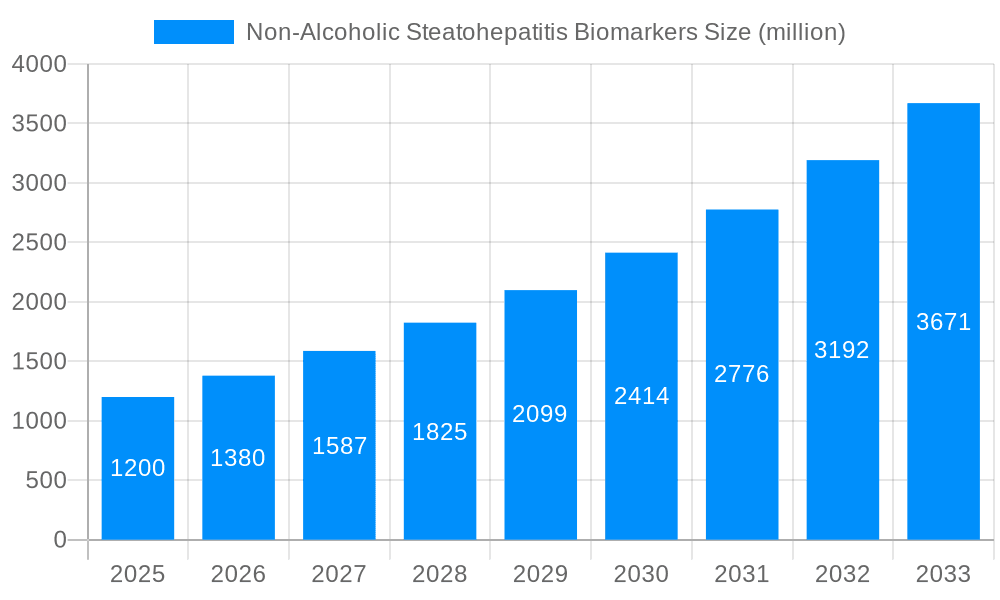

The global Non-Alcoholic Steatohepatitis (NASH) biomarkers market is poised for substantial expansion, projected to reach an estimated value of $3,450.5 million in 2025, with a remarkable upward trajectory towards $7,890.2 million by 2033. This growth is underpinned by a confluence of factors, including the escalating global prevalence of obesity and type 2 diabetes, which are intrinsically linked to NASH development. The study period, spanning from 2019-2033, with a base year of 2025 and a forecast period of 2025-2033, highlights the sustained momentum anticipated in this critical healthcare segment. Historically, from 2019-2024, the market has witnessed steady progress driven by increased research and development investments and a growing understanding of NASH pathogenesis.
The increasing demand for non-invasive diagnostic tools over traditional liver biopsy is a primary driver, offering a safer and more patient-friendly alternative for disease detection and progression monitoring. The market’s segmentation reveals a dynamic landscape. Hepatic Fibrosis Biomarkers are expected to command a significant share due to their crucial role in assessing disease severity and predicting patient outcomes. Serum Biomarkers, owing to their accessibility and ease of collection, will also remain a cornerstone of diagnostic strategies. Oxidative Stress Biomarkers and Apoptosis Biomarkers, while perhaps representing smaller segments currently, are anticipated to witness robust growth as research delves deeper into the molecular mechanisms of NASH. The application of these biomarkers spans across Hospitals, Pharmaceutical Companies, and other healthcare entities, reflecting their broad utility in clinical practice and drug development. The industry is observing a heightened focus on multi-biomarker panels that offer enhanced diagnostic accuracy and prognostic value, moving beyond single-analyte approaches. Furthermore, the integration of advanced analytical techniques and artificial intelligence in biomarker discovery and validation is set to redefine the market's future. The overall trend indicates a maturing market characterized by increasing sophistication, a growing emphasis on personalized medicine, and a strong commitment to addressing the unmet medical needs associated with NASH.
The surge in the Non-Alcoholic Steatohepatitis (NASH) biomarkers market is primarily propelled by the alarming global epidemic of metabolic disorders, particularly obesity and type 2 diabetes, which act as fertile ground for NASH to flourish. As these chronic conditions become more pervasive, so too does the incidence of NASH, consequently amplifying the need for effective diagnostic and monitoring tools. This growing patient population is creating an unprecedented demand for reliable biomarkers that can accurately identify individuals at risk, stage disease progression, and predict treatment response, thereby spurring innovation and investment in this sector. Furthermore, the inherent limitations and invasiveness of the gold-standard liver biopsy are increasingly pushing the medical community towards less intrusive alternatives. The development and validation of non-invasive biomarkers, such as those detectable in serum or breath, are therefore becoming paramount. Patients and clinicians alike are seeking convenient, cost-effective, and reproducible methods for NASH assessment, which directly translates into a burgeoning market for advanced biomarker solutions.
The burgeoning pharmaceutical pipeline focused on NASH therapies is another significant catalyst. With numerous drug candidates undergoing clinical trials, there is a parallel and urgent requirement for validated biomarkers to support patient stratification, efficacy assessment, and safety monitoring. Pharmaceutical companies are actively investing in biomarker research and development to de-risk their drug development programs and to identify patients most likely to benefit from their investigational treatments. This synergy between drug discovery and biomarker science is creating a powerful engine for market growth. Moreover, increasing government and private funding initiatives aimed at understanding and combating liver diseases, including NASH, are further accelerating research and development efforts. These investments are fostering the discovery of novel biomarkers and facilitating their translation into clinically actionable tools, thereby expanding the market's scope and potential.
Despite the robust growth prospects, the Non-Alcoholic Steatohepatitis (NASH) biomarkers market faces several significant challenges and restraints that could temper its expansion. One of the foremost hurdles is the inherent complexity of NASH itself. The disease is a multifactorial condition influenced by genetics, lifestyle, and other co-morbidities, making it difficult to identify a single, universally accurate biomarker. This complexity often necessitates the development and validation of multi-biomarker panels, which can be costly and time-consuming to develop and implement. The lack of a definitive, widely accepted non-invasive biomarker for all stages of NASH also remains a significant restraint. While progress is being made, the current diagnostic landscape still relies, to some extent, on liver biopsy for definitive diagnosis and staging, a procedure that is associated with risks, patient discomfort, and variability in interpretation.
The stringent regulatory approval processes for novel diagnostic tests pose another considerable challenge. Obtaining regulatory clearance from bodies like the FDA or EMA requires extensive validation studies to demonstrate the accuracy, reliability, and clinical utility of new biomarkers. This can lead to lengthy development timelines and significant financial investment, deterring some smaller players from entering the market. Furthermore, issues surrounding reimbursement and payer acceptance can hinder the widespread adoption of new biomarker tests. Healthcare providers and insurance companies need to be convinced of the cost-effectiveness and clinical value of these biomarkers before they are fully integrated into routine clinical practice, which can be a slow and arduous process. The fragmentation of the market, with numerous companies developing diverse biomarker panels and technologies, also presents a challenge in terms of standardization and comparability of results across different platforms.
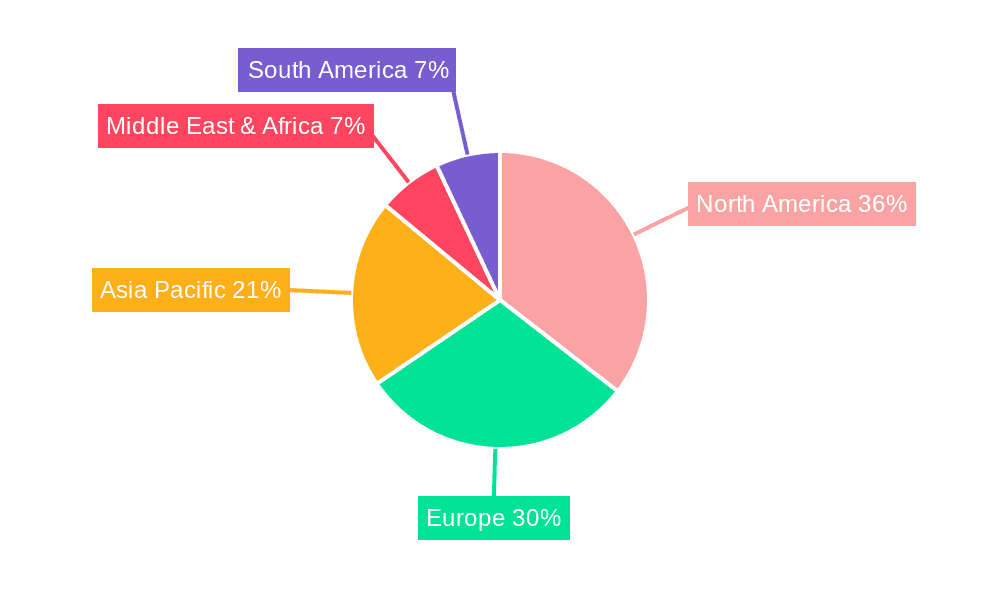
The global Non-Alcoholic Steatohepatitis (NASH) Biomarkers market is poised for substantial growth, with certain regions and segments expected to take the lead. North America, particularly the United States, is anticipated to be a dominant force due to its robust healthcare infrastructure, high prevalence of metabolic diseases, and significant investments in research and development. The presence of leading pharmaceutical companies and established diagnostic laboratories in this region, such as Quest Diagnostics, Laboratory Corporation of America Holdings, and Siemens Medical Solutions USA, further solidifies its leading position. The country's proactive approach to tackling chronic diseases and the increasing awareness among both healthcare professionals and the general public regarding NASH contribute to a high demand for advanced diagnostic solutions. Furthermore, favorable regulatory pathways and substantial funding for clinical trials of NASH therapeutics in the US create a fertile ground for biomarker innovation and adoption.
Among the various segments, Hepatic Fibrosis Biomarkers are projected to exhibit the strongest growth and dominate the market. This dominance stems from the critical need to accurately assess the extent of liver fibrosis, which is a key determinant of disease prognosis and the risk of developing advanced liver conditions like cirrhosis and hepatocellular carcinoma. Identifying and staging fibrosis non-invasively is a major unmet clinical need, and biomarkers that can reliably achieve this will see widespread adoption. Companies like Genfit, Prometheus Laboratories, and Perspectum Diagnostics are actively developing and marketing advanced fibrosis markers that are gaining traction in clinical practice. Following closely, Serum Biomarkers will also represent a significant and rapidly growing segment. Their ease of collection, minimal invasiveness, and scalability make them highly attractive for widespread screening and routine monitoring of NASH. The development of panels of serum markers that can simultaneously assess inflammation, oxidative stress, and fibrosis is a key trend within this segment. Siemens Medical Solutions USA and Quest Diagnostics are well-positioned to capitalize on this segment with their broad diagnostic platforms.
The Pharmaceutical Company segment, in terms of application, is expected to be a major driver of growth. As pharmaceutical giants like AstraZeneca Plc., Pfizer, and Bristol-Myers Squibb Company intensify their efforts in developing NASH therapeutics, the demand for reliable biomarkers to support drug development – from patient selection in clinical trials to monitoring treatment efficacy and safety – will escalate dramatically. These companies are actively collaborating with biomarker developers and investing in research to identify and validate the most predictive markers. Emerging players like NGM Biopharmaceuticals, Gilead Sciences, and Boehringer Ingelheim are also significant contributors to this space. The growing understanding of the underlying pathophysiology of NASH, including its links to Oxidative Stress Biomarkers and Apoptosis Biomarkers, suggests that these segments, while currently smaller, will witness significant expansion as novel diagnostic and therapeutic targets are identified. This intricate interplay between regional strengths, segment-specific needs, and the evolving landscape of NASH research and treatment is shaping the dominant forces within the global NASH biomarkers market.
The Non-Alcoholic Steatohepatitis (NASH) biomarkers industry is experiencing significant growth catalysts, primarily driven by the escalating global burden of obesity and diabetes, which are the leading precursors to NASH. This growing patient pool necessitates more effective and non-invasive diagnostic tools. Furthermore, the increasing focus on early disease detection and intervention, coupled with the limitations of traditional liver biopsy, is creating a strong demand for reliable biomarker-based solutions. The robust pipeline of NASH therapeutics under development by major pharmaceutical companies, including Pfizer and AstraZeneca Plc., acts as a powerful catalyst, as these companies require validated biomarkers for patient stratification and efficacy assessment in clinical trials.
This comprehensive report on Non-Alcoholic Steatohepatitis (NASH) Biomarkers provides an in-depth analysis of market dynamics, trends, and future outlook. It meticulously examines the growth drivers, including the rising prevalence of metabolic diseases and the demand for non-invasive diagnostics, alongside potential challenges like regulatory hurdles and reimbursement issues. The report offers granular insights into key market segments, such as Hepatic Fibrosis Biomarkers and Serum Biomarkers, and their projected growth trajectories. It also explores the application of these biomarkers across various sectors, with a particular focus on the crucial role they play in Pharmaceutical Company drug development. The study delves into the competitive landscape, profiling leading players and highlighting significant developments and technological advancements expected throughout the forecast period. This report serves as an indispensable resource for stakeholders seeking to understand and navigate the evolving NASH biomarkers market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Genfit, Prometheus Laboratories, Siemens Medical Solutions USA, BioPredictive, Quest Diagnostics, AstraZeneca Plc., Laboratory Corporation of America Holdings, Pfizer, Bristol-Myers Squibb Company, NGM Biopharmaceuticals, Gilead Sciences, Perspectum Diagnostics, One Way Liver SL, Enterome SA, Echosens SA, Cisbio Bioassays SAS, Celerion Inc., Xeptagen SpA, Boehringer Ingelheim, .
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "Non-Alcoholic Steatohepatitis Biomarkers," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Non-Alcoholic Steatohepatitis Biomarkers, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.