1. What is the projected Compound Annual Growth Rate (CAGR) of the New Crown Colloidal Gold Antigen Detection Kit?
The projected CAGR is approximately 8.5%.
 New Crown Colloidal Gold Antigen Detection Kit
New Crown Colloidal Gold Antigen Detection KitNew Crown Colloidal Gold Antigen Detection Kit by Type (Recombinant N Protein, Recombinant S Protein), by Application (Retail Pharmacies, Hospital, Online Sales Platforms), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
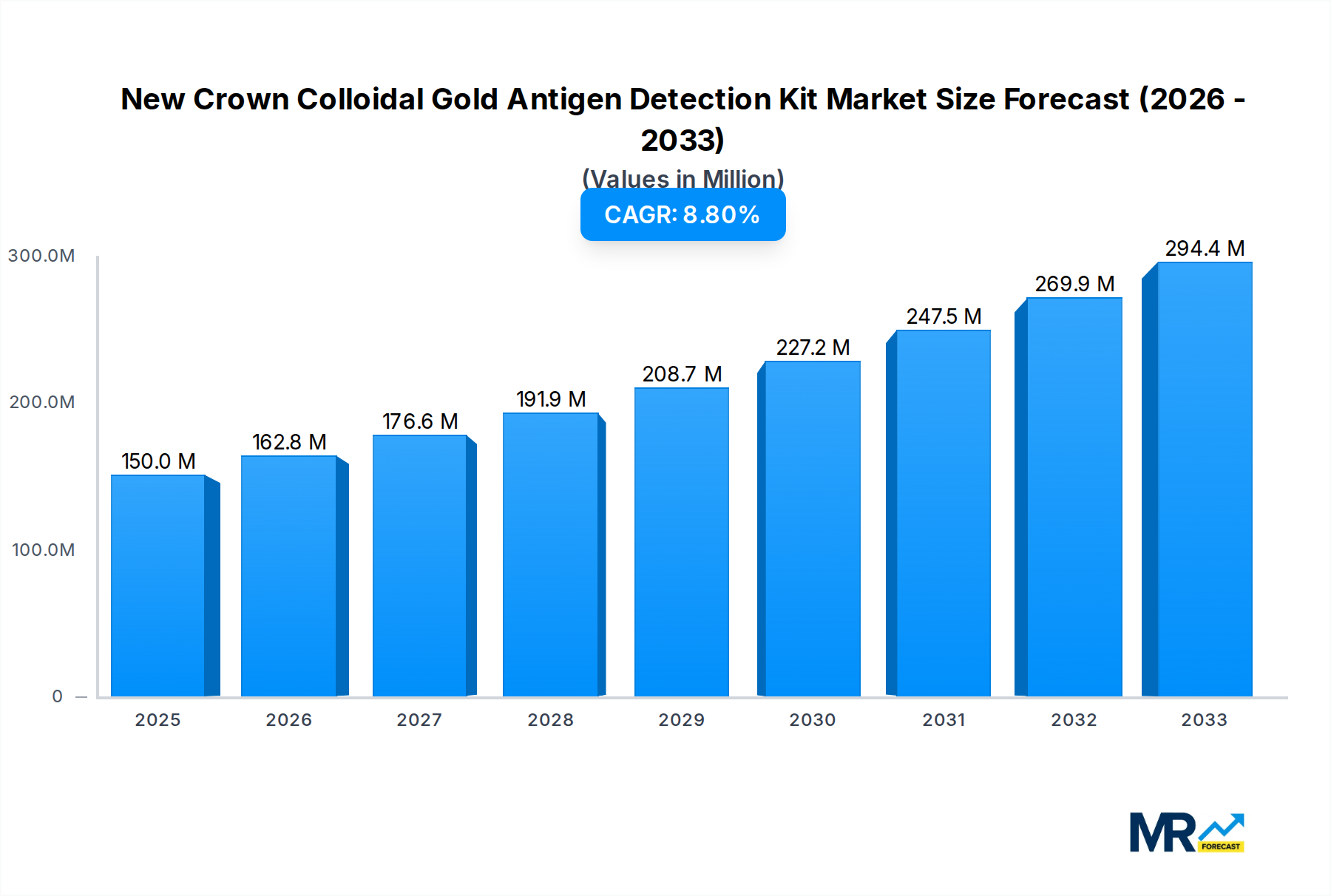
The global market for New Crown Colloidal Gold Antigen Detection Kits is poised for substantial growth, projected to reach approximately \$150 million in market size by 2025, with a robust Compound Annual Growth Rate (CAGR) of 8.5% expected throughout the forecast period of 2025-2033. This expansion is primarily fueled by the increasing demand for rapid and accessible diagnostic solutions for COVID-19. The inherent advantages of colloidal gold antigen tests, including their speed, ease of use, and cost-effectiveness, make them a crucial tool in decentralized testing environments and for high-throughput screening. Key market drivers include the ongoing need for infectious disease surveillance, the rise of point-of-care testing (POCT), and government initiatives promoting widespread diagnostic accessibility. The market is also benefiting from advancements in detection sensitivity and specificity, along with the integration of these kits into various healthcare and retail settings.

The market segmentation reveals a diverse landscape, with Recombinant N Protein and Recombinant S Protein representing the primary types of antigens utilized, each offering distinct diagnostic capabilities. On the application front, Retail Pharmacies are emerging as a significant channel, complementing traditional Hospital settings and the rapidly growing Online Sales Platforms. This diversification in distribution and application underscores the market's adaptability to evolving consumer preferences and healthcare delivery models. Key trends include the development of multiplex assays capable of detecting multiple respiratory pathogens simultaneously, and the increasing focus on at-home testing solutions. Restraints, such as the potential for false negatives or positives and the dynamic regulatory landscape, are being addressed through continuous technological innovation and stringent quality control measures by leading manufacturers like Roche Molecular Systems, Inc., Beijing Wantai Biological Pharmacy Enterprise Co.,Ltd., and Lepu Medical Technology (Beijing) Co.,Ltd., who are at the forefront of driving market advancements and ensuring product reliability.
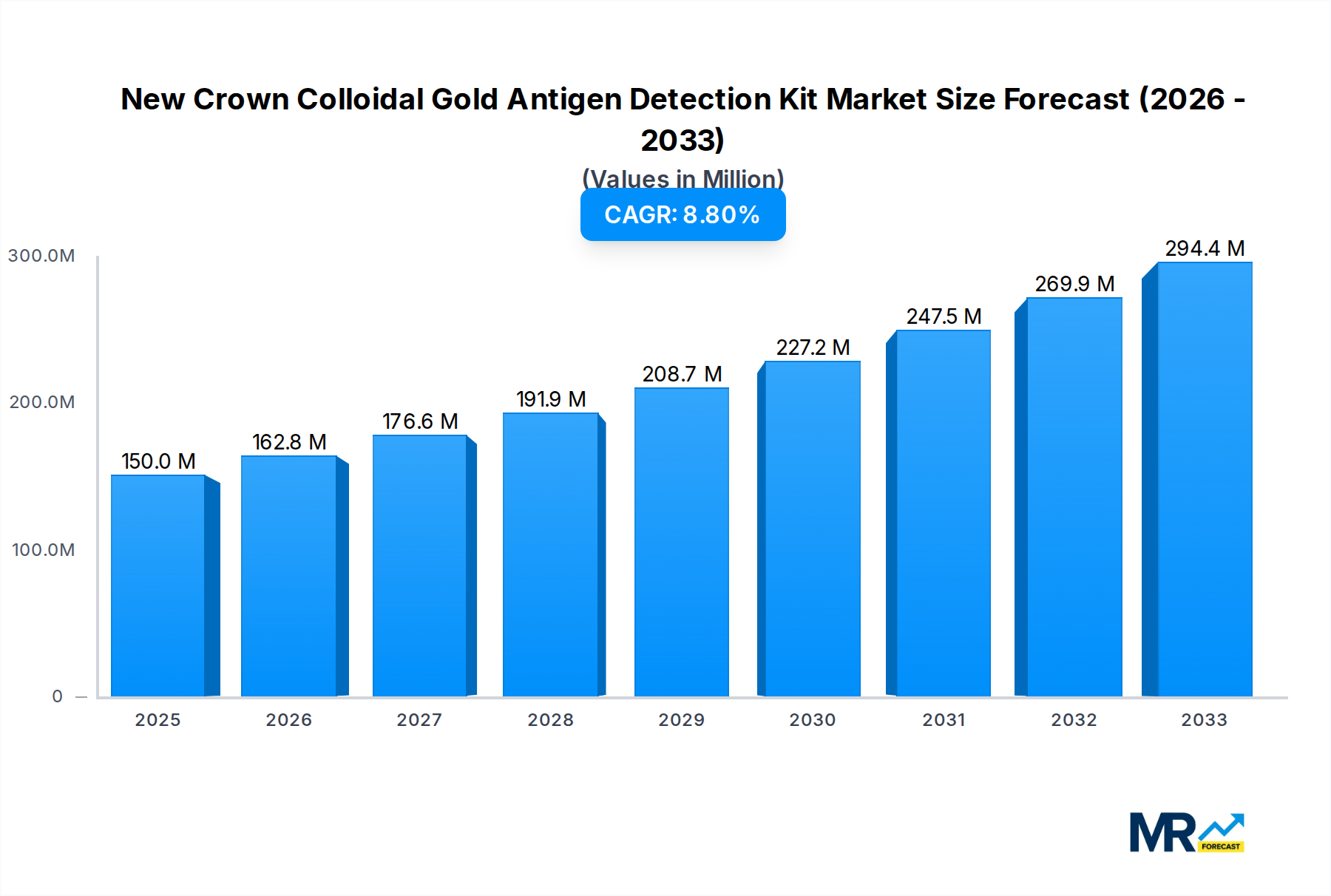

The global market for New Crown Colloidal Gold Antigen Detection Kits is poised for significant evolution, driven by persistent public health concerns and advancements in diagnostic technologies. Over the study period of 2019-2033, with a base year of 2025 and a forecast period extending to 2033, the market is expected to witness a dynamic interplay of demand, technological innovation, and regulatory landscapes. In 2025, the estimated market size is projected to be in the hundreds of millions, reflecting the established presence and ongoing utility of these kits. The historical period from 2019-2024 laid the groundwork, characterized by the initial surge in demand during the pandemic and subsequent refinements in manufacturing and product offerings. As we move into the forecast period, the market is anticipated to stabilize but maintain a strong presence due to factors such as the emergence of new variants, the ongoing need for rapid screening in various settings, and the increasing integration of point-of-care diagnostics. The underlying technology, colloidal gold-based antigen detection, offers a compelling combination of affordability, speed, and ease of use, making it an indispensable tool for initial diagnostics. The market's trajectory will also be shaped by evolving public health strategies, including vaccination campaigns and surveillance programs, which will indirectly influence the demand for reliable and accessible antigen testing solutions. Furthermore, the competitive landscape is robust, with numerous established players and emerging innovators continuously striving to enhance sensitivity, specificity, and user experience. The projected market performance underscores the sustained importance of these kits in managing infectious disease outbreaks and supporting public health infrastructure globally. The continuous adaptation of manufacturers to meet varying regulatory requirements and market demands will be crucial in maintaining this upward, albeit evolving, trend. The sheer volume of potential tests conducted daily across the globe, from individual at-home testing to mass screening initiatives, points towards a market that, while mature in its core technology, still possesses considerable room for growth and innovation within its specific application areas.
The New Crown Colloidal Gold Antigen Detection Kit market is propelled by a confluence of critical factors that ensure its continued relevance and growth. The persistent threat of emerging respiratory viruses and the ongoing need for rapid, accessible diagnostic tools remain the primary drivers. Public health agencies worldwide continue to advocate for widespread testing to facilitate early detection, isolation, and containment strategies, thereby mitigating the spread of infections. Furthermore, the increasing decentralization of healthcare, moving towards point-of-care testing, significantly boosts the demand for user-friendly and cost-effective solutions like colloidal gold antigen kits. These kits empower individuals and healthcare professionals in diverse settings, from retail pharmacies and hospitals to remote clinics, to obtain quick results without requiring specialized laboratory infrastructure. The affordability of colloidal gold technology, when compared to more complex molecular assays, makes it a viable option for mass screening programs and for individuals with limited healthcare access. Technological advancements have also played a crucial role, with manufacturers continuously improving the sensitivity and specificity of their kits, thereby enhancing their diagnostic accuracy. This ongoing refinement addresses earlier limitations and builds greater confidence among users and healthcare providers. The increasing recognition of the benefits of early diagnosis, including timely treatment initiation and reduced transmission, further underpins the market's growth trajectory.
Despite its robust growth, the New Crown Colloidal Gold Antigen Detection Kit market faces several challenges and restraints that could moderate its expansion. A primary concern is the inherent limitation in sensitivity compared to molecular diagnostic methods, such as RT-PCR. While rapid and cost-effective, colloidal gold antigen tests can sometimes yield false-negative results, particularly in individuals with low viral loads, which can lead to a false sense of security and potentially contribute to further transmission. This diagnostic accuracy concern necessitates careful interpretation of results and often requires confirmatory testing with more sensitive methods, adding to the overall cost and complexity. Regulatory hurdles and varying approval processes across different countries can also impede market entry and widespread adoption. Manufacturers must navigate diverse compliance requirements, which can be time-consuming and resource-intensive. Furthermore, the evolving landscape of infectious disease management, including the development of more advanced diagnostic technologies and therapeutic interventions, could potentially diminish the reliance on antigen tests over the long term. Price sensitivity in certain markets and intense competition among a multitude of manufacturers can also lead to price erosion, impacting profit margins and potentially hindering investment in research and development. The recall of certain kits due to quality control issues or performance discrepancies can also erode consumer and regulatory confidence, posing a significant challenge to market sustainability.
The global New Crown Colloidal Gold Antigen Detection Kit market demonstrates a dynamic interplay between regional demand and specific product segments.
Dominant Segments:
Application: Retail Pharmacies: Retail pharmacies have emerged as a pivotal distribution channel, significantly contributing to market dominance. Their widespread accessibility and role as a first point of contact for individuals seeking rapid testing solutions have driven substantial demand. The convenience of over-the-counter availability allows for prompt access to diagnostic tools, facilitating immediate screening for suspected infections. This segment benefits from the general public's increasing awareness and proactive approach to health monitoring, where pharmacies serve as a trusted and convenient location for acquiring essential diagnostic kits. The ability for individuals to purchase and perform tests at home, followed by a consultation if necessary, has cemented the role of retail pharmacies in the broader public health strategy, leading to high sales volumes in this channel.
Type: Recombinant N Protein: The Recombinant N Protein segment is a key driver of market share, largely due to its effectiveness in detecting the nucleocapsid protein of the New Crown virus. The nucleocapsid protein is generally more abundant in the early stages of infection compared to the spike protein, making Recombinant N Protein-based assays highly sensitive for early detection. This characteristic is crucial for rapid identification of infected individuals, enabling timely isolation and intervention. Manufacturers have focused significant research and development efforts on optimizing N protein detection, leading to a robust portfolio of highly accurate and reliable N protein-based antigen kits. The widespread adoption of these kits in various diagnostic settings, driven by their superior performance in capturing early-stage infections, solidifies this segment's dominance.
Dominant Regions:
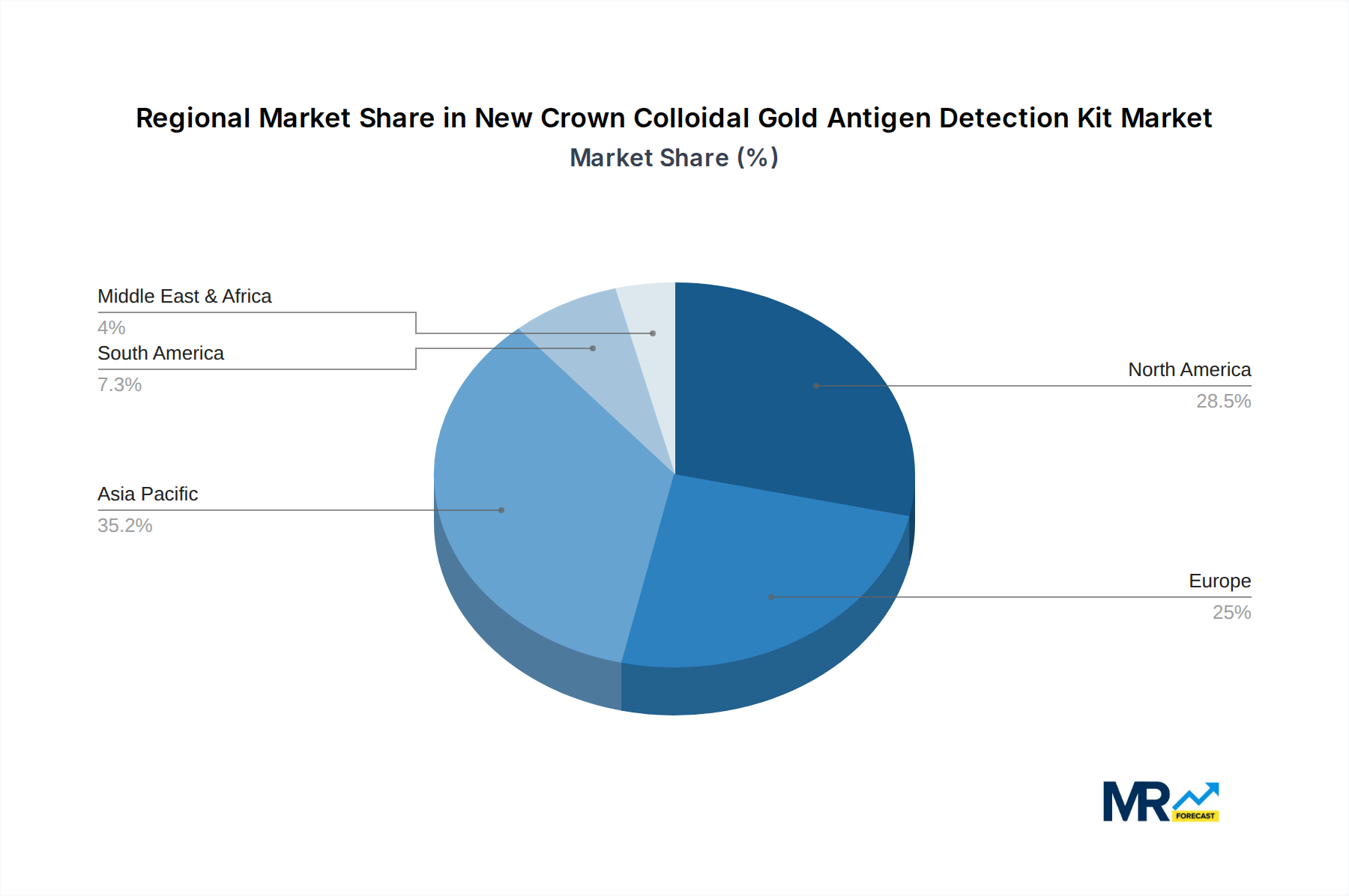
North America: North America, particularly the United States, has consistently been a leading market for New Crown Colloidal Gold Antigen Detection Kits. This dominance is attributable to several factors, including a well-established healthcare infrastructure, high public awareness regarding infectious diseases, and proactive government initiatives supporting widespread testing. The significant investments in public health surveillance and the rapid adoption of point-of-care diagnostics have fueled substantial demand. The presence of leading diagnostic companies and research institutions in the region further contributes to innovation and market growth. Furthermore, the regulatory environment in countries like the United States, while stringent, has also facilitated the swift review and approval of diagnostic products that meet performance standards. The strong emphasis on individual health monitoring and the convenience offered by over-the-counter testing options in retail pharmacies have also played a crucial role in establishing North America as a dominant region. The sheer volume of diagnostic tests conducted, coupled with a preference for rapid and accessible solutions, has created a sustained demand, making this region a powerhouse in the colloidal gold antigen detection kit market.
Europe: Europe represents another significant market for these kits, driven by robust public health systems and a commitment to infectious disease control. Many European countries have implemented comprehensive testing strategies, including mass screening programs and readily accessible testing facilities. The emphasis on early detection and containment, coupled with a growing demand for home-use diagnostic solutions, has further bolstered the market. The presence of major pharmaceutical and biotechnology companies within the region, coupled with a well-developed distribution network, ensures the availability and widespread use of these kits. The regulatory framework in Europe, managed by entities like the European Medicines Agency, also provides a structured pathway for product approval, fostering market growth.
The New Crown Colloidal Gold Antigen Detection Kit industry is experiencing significant growth catalysts that are shaping its future trajectory. The persistent threat of new viral variants and the ongoing need for rapid, accessible diagnostic tools remain paramount. Public health mandates and recommendations for regular testing, especially in high-risk settings such as schools, workplaces, and healthcare facilities, continue to drive demand. Furthermore, the increasing adoption of point-of-care testing and home-use diagnostics, fueled by convenience and cost-effectiveness, is a major growth driver. As diagnostic capabilities decentralize, colloidal gold kits are perfectly positioned to meet these evolving needs.
This report offers a comprehensive analysis of the New Crown Colloidal Gold Antigen Detection Kit market, providing in-depth insights into its dynamics and future outlook. It meticulously examines market trends, growth drivers, and challenges, utilizing robust data and projections. The report delves into key segments such as Recombinant N Protein and Recombinant S Protein types, and analyzes their market share and growth potential. It also scrutinizes application segments including Retail Pharmacies, Hospitals, and Online Sales Platforms, identifying dominant channels and their evolving roles. Industry developments and significant advancements in diagnostic technology are highlighted, offering a forward-looking perspective on innovation. The report further provides detailed profiles of leading companies and their contributions to the market, along with an assessment of regional market dominance, particularly in North America and Europe. This comprehensive coverage equips stakeholders with the necessary information to understand the current landscape and strategize for future opportunities in this critical segment of the diagnostic market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 8.5%.
Key companies in the market include PHASE Scientific International, Ltd., Roche Molecular Systems, Inc., Genabio Diagnostics Inc., Watmind USA, Zybio Inc., BBI Solutions, Wondfo, vazyme, Beijing Hotgen Biotech Co.,Ltd., Beijing Wantai Biological Pharmacy Enterprise Co.,Ltd., Lepu Medical Technology (Beijing) Co.,Ltd., Wuhan EasyDiagnosis Biomedicine Co.,Ltd., Zhejiang Orient Gene Biotech Co., Ltd, Shandong Kanghua Biotechnology Co., Ltd., Shenzhen YHLO Biotech Co., Ltd., SHANGHAI KEHUA BIO-ENGINEERING CO.,LTD., Shanghai ZJ Bio-Tech Co., Ltd., Cangzhou Yongkang Medical Devices Co.,Ltd., Amazing Biotech Co., Ltd., BIOTEKE CORPORATION (WUXI) CO., LTD, Genrui Biotech Inc, GenSure Biotech Inc, Getein Biotech, Inc, Glallergen CO., LTD., Guangdong Hecin Scientific, Inc., Hangzhou Laihe Biotech Co., Hangzhou Lysun Biotechnology Co. Ltd, Hoyotek Biomedical Co.,Ltd., Jiangsu Diagnostics Biotechnology Co., Ltd, Joinstar Biomedical Technology Co. Ltd, .
The market segments include Type, Application.
The market size is estimated to be USD 150 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "New Crown Colloidal Gold Antigen Detection Kit," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the New Crown Colloidal Gold Antigen Detection Kit, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.