1. What is the projected Compound Annual Growth Rate (CAGR) of the New Coronary Pneumonia Nucleic Acid Detection Kit?
The projected CAGR is approximately 6.6%.
 New Coronary Pneumonia Nucleic Acid Detection Kit
New Coronary Pneumonia Nucleic Acid Detection KitNew Coronary Pneumonia Nucleic Acid Detection Kit by Application (Hospital, Clinic, Government), by Type (Anterior Nasal Swab Kit, Oropharyngeal Swab Kit, Nasopharyngeal Swab Kit), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for COVID-19 Nucleic Acid Detection Kits is poised for significant expansion. Driven by persistent demand for accurate and rapid diagnostics, the market is projected to reach a size of 15,000 million by 2025, expanding at a Compound Annual Growth Rate (CAGR) of 6.6% through 2033. This growth is attributed to the ongoing prevalence of respiratory illnesses, critical public health management, and increasing demand for point-of-care testing. Advancements in nucleic acid amplification technologies like RT-PCR and isothermal amplification are boosting kit reliability and accessibility for hospitals, clinics, and screening programs. The development of multiplexed assays for detecting multiple respiratory pathogens also enhances diagnostic efficiency and market potential.
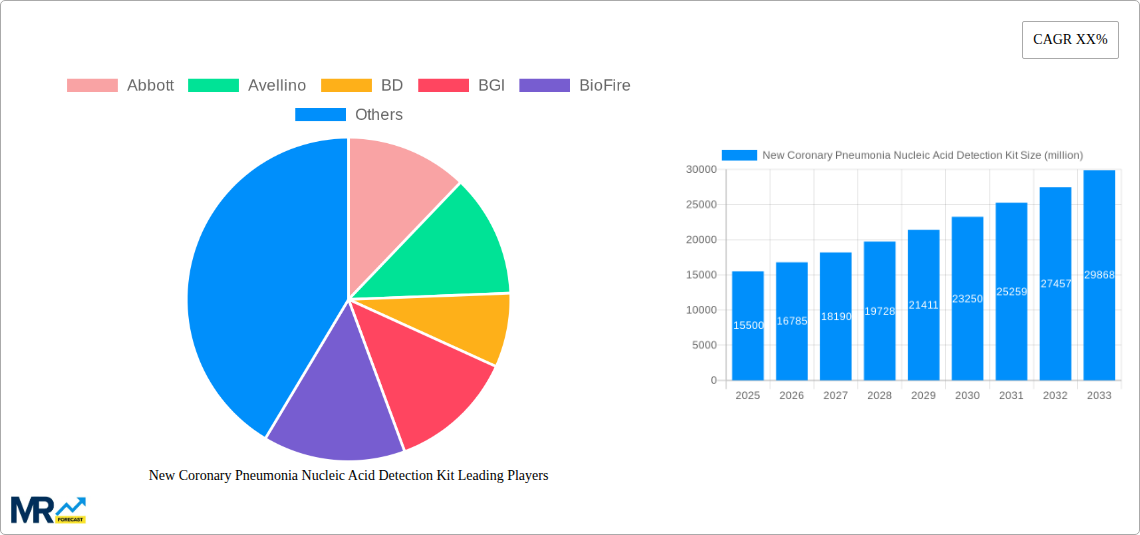

Key market drivers include substantial R&D investments by leading companies, leading to innovative products with faster turnaround times and lower costs. The growing adoption of these kits in clinical and non-clinical settings, such as workplaces and travel hubs, is expanding market penetration. Challenges include stringent regulatory approvals and the evolving nature of the virus, requiring continuous methodological updates. Geographically, North America and Europe lead due to advanced healthcare infrastructure, while the Asia Pacific region, particularly China and India, presents substantial growth opportunities driven by increasing healthcare expenditure and demand in densely populated areas. The market is segmented by application (hospitals, clinics, government) and by kit type (anterior nasal swab, oropharyngeal swab, nasopharyngeal swab).
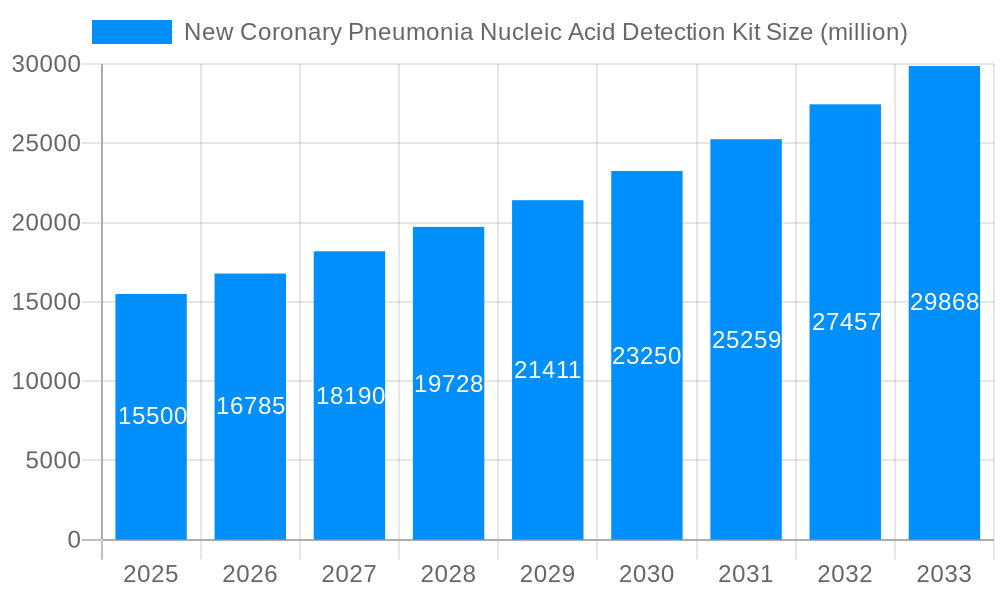

The global COVID-19 Nucleic Acid Detection Kit market is set for significant evolution. The base year of 2025 provides a crucial baseline for understanding its trajectory. By 2025, the market is expected to reach 15,000 million, with further expansion projected throughout the forecast period ending in 2033. This growth is supported by sustained demand for accurate COVID-19 diagnostics. The infrastructure and manufacturing expertise developed during the pandemic have enabled broader applications and more efficient production, leading to increased market penetration and product diversification.
Decentralized testing, facilitated by rapid point-of-care (POC) solutions, is a significant trend. As healthcare systems prioritize efficiency, demand for kits suitable for clinics and home-use settings is rising. Innovations in swab types and assay designs focus on ease of use and speed. The market also emphasizes multiplexing capabilities for simultaneous detection of multiple respiratory pathogens, including SARS-CoV-2, influenza, and RSV, enhancing diagnostic value. Technological advancements in real-time PCR and isothermal amplification continue to improve sensitivity and specificity, ensuring diagnostic tool reliability. The interplay of public health initiatives, government funding, and commercial strategies will shape market expansion.
The New Coronary Pneumonia Nucleic Acid Detection Kit market is experiencing sustained growth driven by a confluence of critical factors. Foremost among these is the ongoing need for surveillance and early detection of SARS-CoV-2, even as pandemic-level restrictions ease. The virus continues to evolve, and the potential for new variants necessitates continuous monitoring, thereby sustaining the demand for accurate nucleic acid detection kits. Public health agencies worldwide remain committed to maintaining robust diagnostic capacities to respond effectively to future outbreaks or surges in cases. This sustained commitment translates into ongoing procurement and deployment of these testing solutions.
Furthermore, the increasing integration of molecular diagnostics into routine healthcare practices plays a pivotal role. The COVID-19 pandemic accelerated the adoption and acceptance of nucleic acid testing, demonstrating its value beyond just emergency response. Healthcare providers are now more inclined to incorporate these tests for differential diagnosis of respiratory illnesses, patient management, and infection control protocols within hospitals and clinics. The advancements in assay technology, leading to faster turnaround times and improved sensitivity, are also crucial. Innovations in platforms, such as those enabling point-of-care testing, are making diagnostics more accessible and convenient for a wider range of settings, from small clinics to remote locations. Finally, government initiatives and investments aimed at strengthening national public health infrastructure and preparedness for future pandemics are providing a significant financial impetus, ensuring a stable market for these essential diagnostic tools.
Despite the positive outlook, the New Coronary Pneumonia Nucleic Acid Detection Kit market is not without its hurdles. A significant challenge lies in the evolving landscape of the virus and public health priorities. As the perceived severity of COVID-19 diminishes in certain populations and governments shift their focus and funding, the demand for testing could potentially decline more rapidly than anticipated. This can lead to oversupply and price pressures for manufacturers. Another restraint is the intense competition within the market, which has led to a commoditization of certain basic testing kits, impacting profit margins for companies. The need for continuous innovation to stay ahead of the curve and differentiate offerings is therefore paramount.
Moreover, regulatory complexities and the time required for new product approvals can pose a barrier to entry and slow down the introduction of novel technologies. Manufacturers must navigate varying regulatory frameworks across different regions, which can be a costly and time-consuming process. The cost of raw materials and manufacturing, particularly for specialized reagents and components, can also fluctuate, impacting the overall cost-effectiveness of the kits. Finally, the emergence of alternative diagnostic technologies, such as rapid antigen tests or serological assays, which may offer lower costs or faster results for certain applications, could potentially erode market share for nucleic acid detection kits in some segments, although nucleic acid tests remain the gold standard for definitive diagnosis.
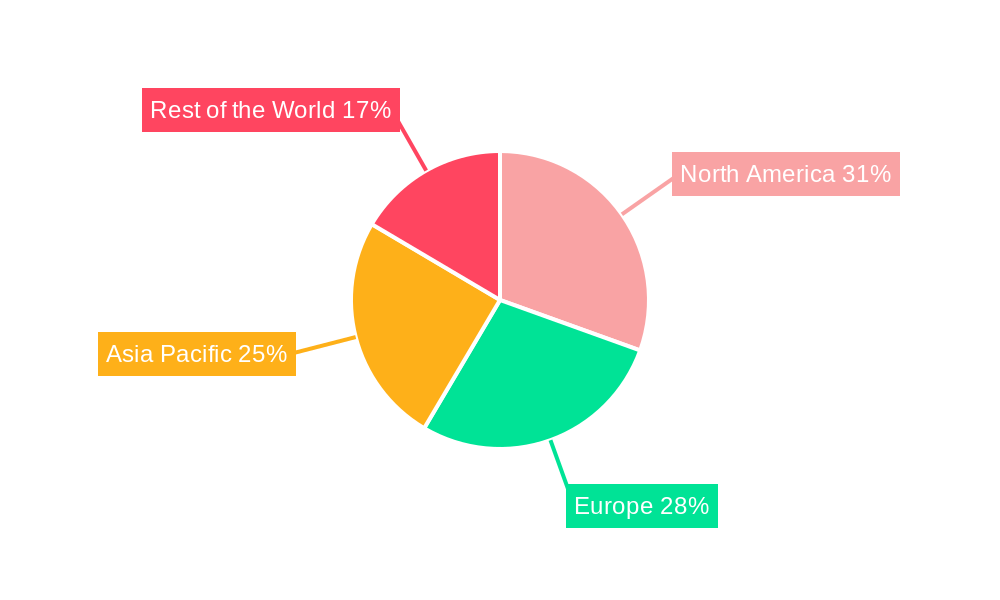
The New Coronary Pneumonia Nucleic Acid Detection Kit market is projected to witness significant regional and segmental dominance, driven by a combination of healthcare infrastructure, population density, and governmental policies.
Dominant Regions/Countries:
Dominant Segments:
Application: Hospital: Hospitals are anticipated to continue to be the largest application segment for New Coronary Pneumonia Nucleic Acid Detection Kits. This is attributed to several factors:
Type: Nasopharyngeal Swab Kit: While other swab types are gaining traction, the Nasopharyngeal Swab Kit is expected to maintain a significant market share, particularly in clinical settings for definitive diagnosis.
The synergy between these dominant regions and segments will shape the overall market dynamics, with North America and Europe likely leading in technological adoption and high-value kit utilization, while the Asia Pacific region offers immense growth potential due to its large population and increasing healthcare investments. Within applications, hospitals will remain the cornerstone, and nasopharyngeal swabs will continue to be a crucial type of kit for accurate diagnosis.
The growth of the New Coronary Pneumonia Nucleic Acid Detection Kit industry is fueled by several key catalysts. The continued global circulation of SARS-CoV-2 and its emerging variants necessitates ongoing diagnostic vigilance and surveillance, ensuring a baseline demand. Furthermore, the increasing integration of molecular diagnostics into routine public health strategies and routine healthcare pathways for respiratory illness management is expanding the market beyond pandemic-specific needs. Advancements in point-of-care (POC) testing technologies, enabling faster results and decentralized testing in clinics and remote settings, are significantly driving adoption and accessibility. Government investments in pandemic preparedness and strengthening healthcare infrastructure provide crucial financial support and market stability. Finally, the development of multiplex assays that can detect multiple respiratory pathogens simultaneously is enhancing the utility and value proposition of these kits.
The comprehensive coverage of the New Coronary Pneumonia Nucleic Acid Detection Kit report offers an in-depth analysis of market dynamics, trends, and future projections. It provides valuable insights into the market size and growth potential, detailing projections from the 2019-2024 Historical Period through to the 2025-2033 Forecast Period, with 2025 serving as the crucial Base Year and Estimated Year. The report meticulously examines the driving forces, such as the persistent need for surveillance and the integration of molecular diagnostics into routine healthcare, alongside the challenges, including evolving public health priorities and market competition. It identifies key regions and countries poised for market dominance, alongside critical application and type segments that will shape demand. Furthermore, the report highlights significant industry developments and lists the leading players in the market, providing a holistic view for stakeholders. This comprehensive approach ensures that businesses, researchers, and policymakers have the necessary information to make informed strategic decisions within this vital sector of the diagnostics industry.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 6.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 6.6%.
Key companies in the market include Abbott, Avellino, BD, BGI, BioFire, BioMedomics, Biomerieux, CDC, Cepheid, Chembio, Co-Diagnostics Inc., CTK Biotech, Danaher, DiaSorin, Epitope Diagnostics, Inc., Grifols, Hologic, Chungdo Pharm Co., Ltd., .
The market segments include Application, Type.
The market size is estimated to be USD 15000 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "New Coronary Pneumonia Nucleic Acid Detection Kit," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the New Coronary Pneumonia Nucleic Acid Detection Kit, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.