1. What is the projected Compound Annual Growth Rate (CAGR) of the Neuraminidase Assay Kit?
The projected CAGR is approximately XX%.
 Neuraminidase Assay Kit
Neuraminidase Assay KitNeuraminidase Assay Kit by Type (Colorimetric Testing, Fluorometric Testing, World Neuraminidase Assay Kit Production ), by Application (General Hospitals, Specialty Clinics, World Neuraminidase Assay Kit Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Neuraminidase Assay Kit market is poised for significant expansion, projected to reach an estimated market size of approximately $180 million by 2025, with a projected Compound Annual Growth Rate (CAGR) of around 8%. This robust growth is primarily fueled by the increasing prevalence of influenza and other viral infections, which necessitate accurate and efficient diagnostic tools. The rising demand for rapid and sensitive detection methods, coupled with advancements in assay technologies like fluorometric testing, is a key driver. Furthermore, the growing emphasis on early disease detection and the expanding research and development activities in virology and drug discovery are contributing to market momentum. The market also benefits from the expanding healthcare infrastructure globally and increasing healthcare expenditure, particularly in emerging economies.
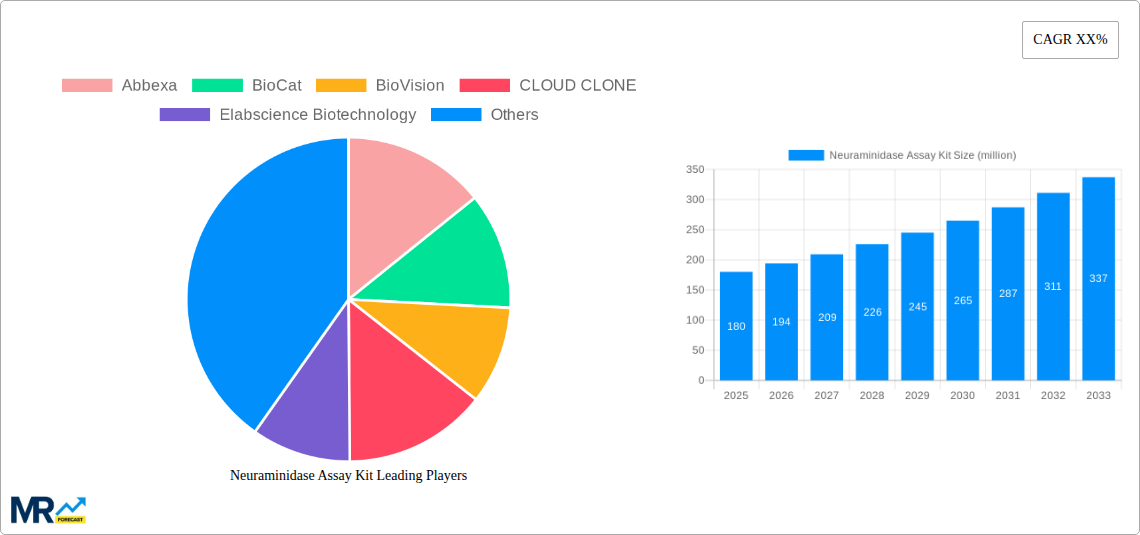

The market is segmented into various types, including Colorimetric Testing and Fluorometric Testing, with Fluorometric Testing expected to gain a larger market share due to its superior sensitivity and speed. Application-wise, the market is dominated by general hospitals and specialty clinics, reflecting the widespread need for neuraminidase assays in clinical diagnostics. While the market presents substantial opportunities, certain restraints such as the high cost of advanced assay kits and stringent regulatory approvals for new diagnostic products could pose challenges. However, the continuous innovation by key players like Abbexa, BioCat, and Sartorius, focusing on developing more cost-effective and user-friendly kits, is expected to mitigate these restraints. Geographically, North America and Europe currently lead the market, but the Asia Pacific region is anticipated to witness the fastest growth due to increasing healthcare investments and a growing patient population.
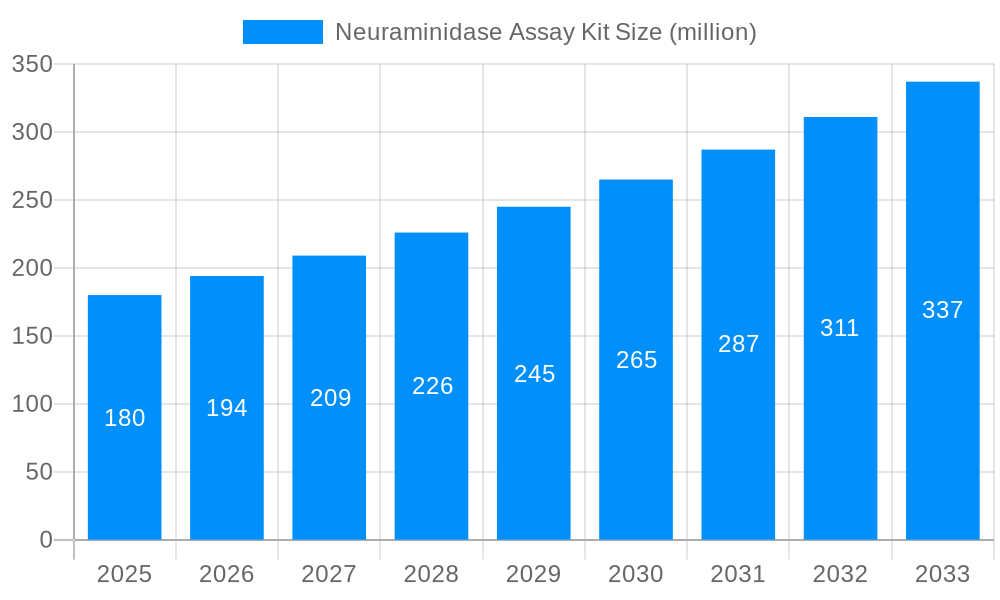

This comprehensive report delves into the dynamic global market for Neuraminidase Assay Kits, offering an in-depth analysis of trends, driving forces, challenges, and future projections. Spanning the historical period of 2019-2024, the base year of 2025, and a robust forecast period extending to 2033, this study provides invaluable insights for stakeholders. The report quantifies market opportunities, estimated at 50 million units in the base year of 2025, with projections anticipating a significant expansion.
The Neuraminidase Assay Kit market is experiencing a significant evolutionary phase, characterized by technological advancements and expanding applications, as evidenced by the projected market size of 75 million units by 2033. A key trend is the increasing adoption of Fluorometric Testing methods. While Colorimetric Testing kits have historically dominated due to their cost-effectiveness and ease of use, the superior sensitivity and dynamic range offered by fluorometric assays are driving their ascendance, particularly in research settings and for detecting low levels of neuraminidase activity. This shift is supported by ongoing research into novel fluorogenic substrates that exhibit enhanced specificity and signal-to-noise ratios. The "World Neuraminidase Assay Kit Production" segment is also witnessing a surge, driven by increased demand for influenza diagnostics and antiviral drug development. The rising global incidence of viral respiratory infections, coupled with the emergence of novel influenza strains, has propelled the need for rapid and accurate diagnostic tools. This has directly translated into heightened demand for neuraminidase assay kits, which are crucial for identifying viral activity and assessing the efficacy of neuraminidase inhibitors. Furthermore, advancements in assay kit formulations, including improved stability, simplified protocols, and multiplexing capabilities, are contributing to market growth. The integration of automation in clinical laboratories and the growing emphasis on personalized medicine are also shaping the market landscape, demanding highly efficient and reliable assay solutions. The market is also seeing a trend towards kits with higher throughput and lower detection limits to cater to the increasing volume of samples and the need for early disease detection. This is particularly relevant in the context of pandemic preparedness and the continuous monitoring of circulating viral strains. The burgeoning field of drug discovery and development, specifically targeting neuraminidase as a therapeutic target for various viral diseases, is another significant driver. Researchers are actively developing and optimizing assay kits to screen potential drug candidates and to monitor their therapeutic effects in preclinical and clinical studies. The expanding geographical reach of healthcare infrastructure and the growing awareness about the importance of accurate viral diagnostics in developing economies are also contributing to the overall market expansion. The increasing focus on understanding the role of neuraminidase in various biological processes beyond viral infections, such as cancer and neurodegenerative diseases, is also opening up new avenues for assay kit utilization and market growth.
The global Neuraminidase Assay Kit market is experiencing robust growth, projected to reach 80 million units by 2033, propelled by a confluence of critical factors. A primary driver is the escalating global burden of viral infections, particularly influenza, which necessitates rapid and accurate diagnostic solutions. Neuraminidase's pivotal role in the viral replication cycle makes it a prime target for detection and therapeutic intervention. Consequently, the increasing prevalence of influenza outbreaks and the constant threat of novel viral pandemics have significantly amplified the demand for neuraminidase assay kits in both clinical diagnostics and public health surveillance. Furthermore, the burgeoning pharmaceutical industry's relentless pursuit of effective antiviral therapies is a substantial market stimulant. Neuraminidase inhibitors are a cornerstone in treating influenza, and the development of new and improved drugs necessitates a constant supply of reliable assay kits for drug screening, efficacy testing, and mechanism-of-action studies. The continuous innovation in assay kit technologies, leading to enhanced sensitivity, specificity, and user-friendliness, also plays a crucial role. Companies are investing in developing kits that offer faster turnaround times, require less sample volume, and can be integrated into automated laboratory workflows, thereby improving laboratory efficiency and reducing operational costs. The growing awareness among healthcare professionals and the general public about the importance of early disease detection and the availability of advanced diagnostic tools further contribute to market expansion. Additionally, the increasing application of neuraminidase assays in academic research, exploring its multifaceted roles in various biological processes beyond viral pathogenesis, such as in cancer and neurological disorders, is opening up new market segments and driving demand for specialized kits.
Despite the promising growth trajectory, the Neuraminidase Assay Kit market faces several challenges that could temper its expansion, with the market size anticipated to reach 85 million units by 2033. One significant restraint is the high cost of advanced assay technologies, particularly for sophisticated fluorometric kits. While offering superior performance, their higher price point can be a barrier for widespread adoption, especially in resource-limited settings or for routine diagnostic applications where cost-effectiveness is paramount. This can limit the market penetration of the most advanced technologies, leaving a larger share to more traditional and economical methods. Another challenge lies in the standardization of assay protocols and results across different kit manufacturers and laboratory settings. Variations in reagent formulations, assay conditions, and detection methodologies can lead to inconsistencies in results, complicating inter-laboratory comparisons and potentially impacting diagnostic accuracy. This lack of universal standardization necessitates rigorous internal validation by each laboratory, adding to operational complexity and cost. Furthermore, the emergence of alternative diagnostic methods, such as molecular diagnostic techniques like RT-PCR, which can detect viral genetic material directly, poses a competitive threat. While these methods may have different targets, their increasing accessibility and established reliability for viral detection could divert some market share from enzyme-based assays. The stringent regulatory approval processes for diagnostic kits can also lead to extended development timelines and increased costs for manufacturers, potentially slowing down the introduction of new and innovative products. Finally, the reliance on specialized reagents and raw materials, which may be subject to supply chain disruptions or price volatility, can impact the cost-effectiveness and consistent availability of neuraminidase assay kits.
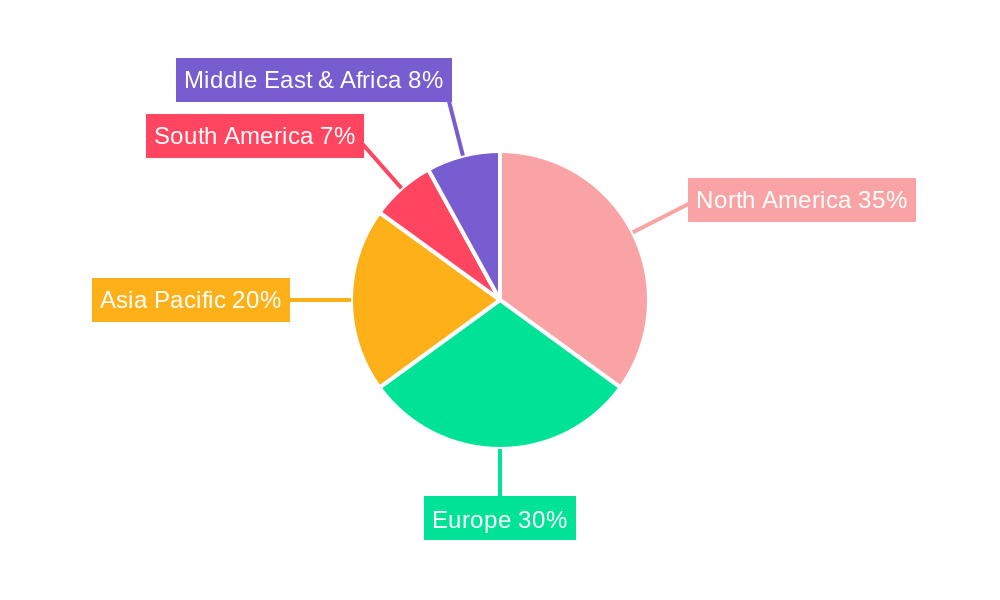
The global Neuraminidase Assay Kit market, projected to reach 90 million units by 2033, is poised for significant dominance by specific regions and market segments. North America is anticipated to lead the market, driven by its advanced healthcare infrastructure, substantial investment in research and development, and a high prevalence of influenza and other viral respiratory illnesses. The region’s strong presence of leading biotechnology companies, coupled with a proactive approach to public health surveillance and pandemic preparedness, further solidifies its dominant position. The United States, in particular, contributes significantly due to its well-established diagnostic laboratories, pharmaceutical research, and government funding for infectious disease control.
Within the market segments, Fluorometric Testing is expected to witness the most substantial growth and gain increasing market share. While Colorimetric Testing kits have historically been the workhorse due to their affordability and ease of use, the superior sensitivity, broader dynamic range, and potential for higher throughput offered by fluorometric assays are making them increasingly indispensable. This trend is fueled by:
While North America is projected to dominate, other regions like Europe are also significant contributors, supported by robust healthcare systems, strong pharmaceutical research sectors, and active public health initiatives. The Asia-Pacific region is expected to exhibit the fastest growth rate, driven by expanding healthcare infrastructure, increasing awareness of infectious diseases, and a growing population, leading to a higher demand for diagnostic solutions. The "World Neuraminidase Assay Kit Production" segment is also a critical indicator of market health, reflecting the overall capacity and output of manufacturers globally.
Several key factors are acting as significant growth catalysts for the Neuraminidase Assay Kit industry. The persistent and evolving threat of influenza and other viral respiratory pandemics remains a primary catalyst, driving sustained demand for rapid and accurate diagnostic tools. Furthermore, the burgeoning pharmaceutical pipeline focused on developing novel neuraminidase inhibitors for various viral diseases provides a continuous impetus for assay kit development and utilization. Ongoing research exploring neuraminidase's role in non-viral pathologies, such as cancer, opens up entirely new application areas and market opportunities. The increasing global healthcare expenditure and the expansion of laboratory infrastructure in emerging economies are also crucial catalysts, broadening the accessibility and adoption of these assay kits.
This report provides a holistic view of the Neuraminidase Assay Kit market. Beyond market sizing and segmentation, it offers an in-depth analysis of the competitive landscape, including company profiles, strategic initiatives, and market share estimations of key players like Abbexa, BioCat, BioVision, CLOUD CLONE, Elabscience Biotechnology, Intec Products, MyBiosource, and Sartorius. The report also meticulously examines the manufacturing landscape and supply chain dynamics, identifying potential bottlenecks and opportunities. Furthermore, it includes a detailed breakdown of industry-specific developments, regulatory frameworks, and emerging technologies that are shaping the future of neuraminidase detection and research. The comprehensive coverage ensures that stakeholders gain a deep understanding of the market's present state and future trajectory, enabling informed strategic decision-making.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Abbexa, BioCat, BioVision, CLOUD CLONE, Elabscience Biotechnology, Intec Products, MyBiosource, Sartorius.
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Neuraminidase Assay Kit," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Neuraminidase Assay Kit, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.