1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Low Frequency Pulse Antiemetic Device?
The projected CAGR is approximately 5.4%.
 Medical Low Frequency Pulse Antiemetic Device
Medical Low Frequency Pulse Antiemetic DeviceMedical Low Frequency Pulse Antiemetic Device by Type (Single Use, Multiple Use), by Application (Online Sales, Offline Sales), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Medical Low Frequency Pulse Antiemetic Device market is projected for substantial growth, expected to reach $6.4 billion by 2024, with a CAGR of 5.4% through 2033. This expansion is driven by the rising incidence of chemotherapy-induced nausea and vomiting (CINV), post-operative nausea and vomiting (PONV), and motion sickness. Increasing preference for non-pharmacological, minimally invasive treatments fuels demand for these devices. Advances in wearable technology and a focus on patient comfort are also key growth catalysts. The efficacy and convenience of low-frequency pulse therapy, offering a side-effect-free alternative to traditional antiemetics, are promoting its adoption across healthcare settings.

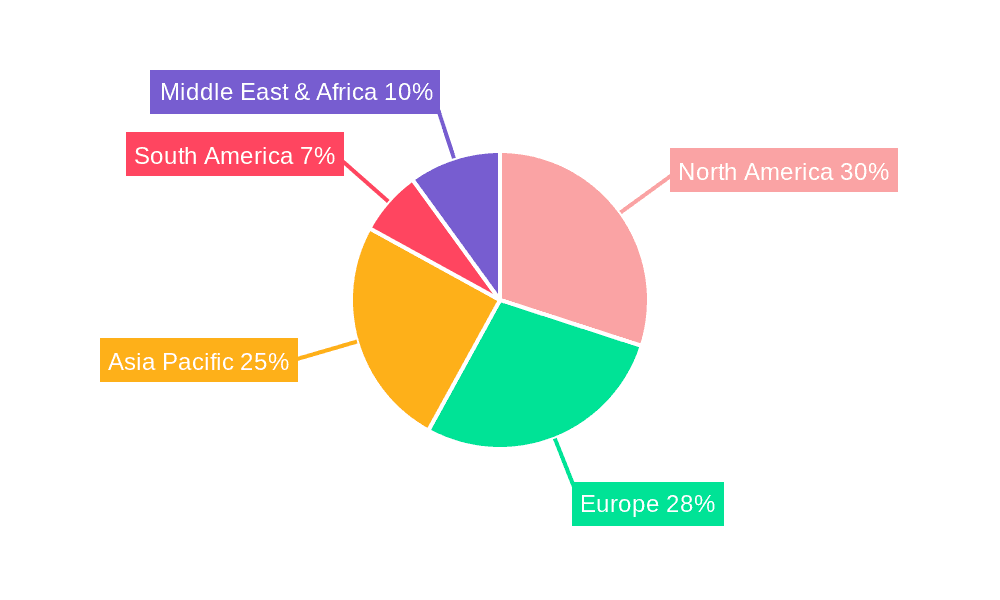
Market dynamics are further influenced by the growing importance of online sales channels for wider consumer reach, complementing traditional offline distribution via medical suppliers and healthcare facilities. Single-use devices are anticipated to dominate due to convenience and hygiene, while multi-use devices will serve recurring needs and homecare. Geographically, North America and Europe are expected to lead, owing to high healthcare spending and advanced technology adoption. The Asia Pacific region presents significant growth potential due to its large population, increasing health awareness, and rising disposable incomes. Key industry players are actively investing in R&D to improve device efficacy and user experience, further supporting market expansion.
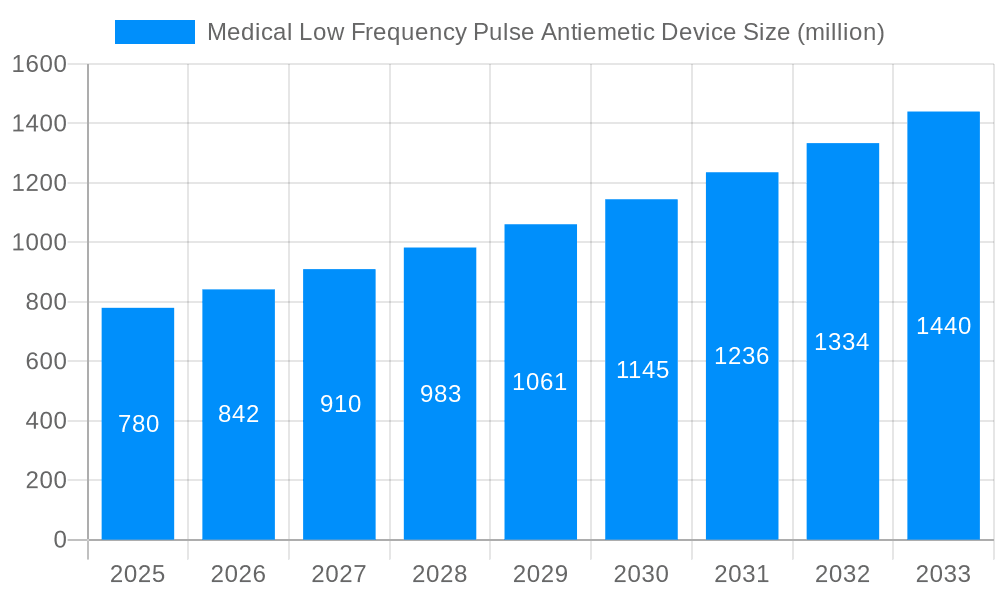

This report offers a comprehensive analysis of the Medical Low Frequency Pulse Antiemetic Device market, covering trends, drivers, challenges, and future opportunities. The study analyzes the 2019-2024 historical period, with 2024 as the base year and estimated year, and forecasts growth through 2033. This extensive research provides stakeholders with actionable insights into this expanding sector, projecting a market size of $6.4 billion.
XXX The Medical Low Frequency Pulse Antiemetic Device market is experiencing a significant surge in demand driven by increasing awareness of non-pharmacological alternatives for managing nausea and vomiting, particularly in the context of post-operative recovery, chemotherapy, and motion sickness. Consumers are actively seeking safer and more convenient methods to alleviate these debilitating symptoms, moving away from traditional medication with potential side effects. The growing prevalence of conditions that trigger nausea, such as cancer and pregnancy-related sickness, further fuels this trend. Technological advancements are also playing a pivotal role, with manufacturers focusing on developing more sophisticated, user-friendly, and personalized devices. Innovations in pulse wave modulation and biofeedback mechanisms are enhancing efficacy and patient comfort. The market is witnessing a shift towards wearable and discreet devices that can be easily integrated into daily life, offering continuous relief without disrupting activities. Furthermore, the rising adoption of telemedicine and remote patient monitoring solutions is creating new avenues for the distribution and application of these devices, allowing for greater accessibility and personalized care. The integration of smart technologies, enabling data tracking and personalized therapy adjustments, is another key trend shaping the market's trajectory. The market is also seeing a growing interest from the healthcare professional community, who are recognizing the therapeutic potential of these devices in complementing existing treatment protocols. This growing acceptance, coupled with increasing research and clinical trials validating their effectiveness, is poised to significantly expand the market in the coming years. The global push towards preventative healthcare and wellness is also contributing to the rising popularity of low-frequency pulse antiemetic devices as a proactive approach to managing potential nausea triggers.
Several key factors are propelling the growth of the Medical Low Frequency Pulse Antiemetic Device market. Primarily, the increasing global burden of diseases and conditions that cause nausea and vomiting, such as cancer, chemotherapy-induced nausea and vomiting (CINV), postoperative nausea and vomiting (PONV), and motion sickness, directly translates into a larger potential patient pool. This growing patient population necessitates effective and accessible antiemetic solutions. Secondly, there is a significant and growing preference among patients and healthcare providers for non-pharmacological and drug-free alternatives to traditional antiemetic medications. This shift is driven by concerns about the side effects, potential drug interactions, and the development of drug resistance associated with pharmaceutical treatments. Low-frequency pulse devices offer a perceived safer and more natural approach to symptom management. Furthermore, continuous technological advancements in the field are leading to the development of more sophisticated, user-friendly, and efficacious devices. These innovations include improved pulse wave algorithms, enhanced battery life, discreet and wearable designs, and features like adjustable intensity and personalized therapy programs. The increasing affordability and accessibility of these devices, coupled with growing consumer awareness fostered by digital marketing and online health platforms, are also significant drivers. The rise of the aging population globally, who are often more susceptible to conditions causing nausea, further contributes to the market's expansion.
Despite the promising growth trajectory, the Medical Low Frequency Pulse Antiemetic Device market faces several challenges and restraints. A significant hurdle is the lack of widespread awareness and understanding among both the general public and some healthcare professionals regarding the efficacy and safety of these devices. Many potential users may remain unaware of their existence or harbor skepticism about their therapeutic benefits compared to established pharmaceutical options. Another challenge lies in the regulatory landscape, which can be complex and vary across different regions, potentially slowing down product approvals and market penetration. Ensuring consistent and rigorous clinical validation for a broad range of applications is crucial but can be a time-consuming and costly process. The cost of some advanced devices can also be a barrier to adoption, particularly in price-sensitive markets or for individuals with limited disposable income. While the market is expanding, some segments might still find the initial investment prohibitive. Furthermore, the perception of medical devices as less effective than medication by a segment of the population, ingrained through years of pharmaceutical marketing, requires a significant shift in mindset. Overcoming this ingrained perception and building trust requires robust clinical evidence and effective patient education. Finally, the competition from established pharmaceutical antiemetic drugs, which have a long history of use and well-documented efficacy, remains a considerable restraint.
The Medical Low Frequency Pulse Antiemetic Device market is anticipated to witness robust growth across various regions and segments.
Dominant Regions:
North America: The United States and Canada are poised to lead the market. This dominance is attributed to several factors:
Europe: Western European countries, including Germany, the United Kingdom, France, and Italy, are expected to be significant contributors.
Dominant Segments:
Type: Multiple Use Devices: While Single Use devices offer convenience, Multiple Use devices are projected to dominate the market.
Application: Offline Sales: While Online Sales are growing rapidly, Offline Sales are expected to maintain a dominant position, especially in the near to mid-term.
The interplay of these regional and segmental factors will shape the overall market dynamics, with continuous innovation and evolving consumer preferences influencing the pace and extent of dominance.
The Medical Low Frequency Pulse Antiemetic Device industry is fueled by several key growth catalysts. The increasing global incidence of conditions leading to nausea, such as chemotherapy-induced nausea, postoperative recovery, and motion sickness, presents a vast patient pool. Coupled with this is the growing consumer demand for non-pharmacological, drug-free alternatives, driven by concerns over medication side effects and drug resistance. Continuous technological advancements in pulse modulation technology, leading to more effective, comfortable, and user-friendly wearable devices, are a significant catalyst. The rising awareness and acceptance of these devices, supported by an increasing body of clinical evidence and positive patient testimonials, further propel their adoption. Furthermore, the expansion of e-commerce platforms and telehealth services is broadening accessibility and reach, making these devices available to a wider demographic.
This comprehensive report on the Medical Low Frequency Pulse Antiemetic Device market provides an exhaustive analysis of the industry's current state and future outlook. It delves into key trends, exploring the increasing demand for non-pharmacological solutions, technological advancements in device design, and evolving consumer preferences. The report meticulously examines the driving forces behind market growth, including the rising prevalence of nausea-inducing conditions and the shift towards patient-centric care. Simultaneously, it addresses the challenges and restraints, such as regulatory hurdles, the need for greater awareness, and competition from established therapies. The analysis extends to identifying key regions and segments poised for dominance, offering valuable insights for strategic market entry and expansion. Furthermore, the report highlights significant developments and leading players, providing a holistic view of the competitive landscape and innovation pipeline. This report is an indispensable resource for manufacturers, investors, healthcare providers, and policymakers seeking to understand and capitalize on the opportunities within this rapidly evolving market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.4% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.4%.
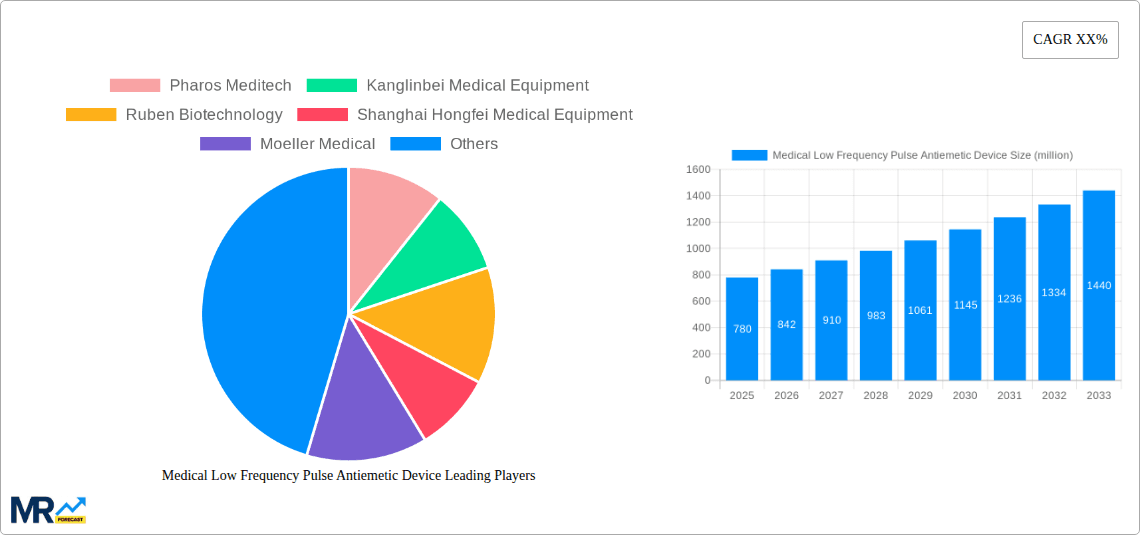
Key companies in the market include Pharos Meditech, Kanglinbei Medical Equipment, Ruben Biotechnology, Shanghai Hongfei Medical Equipment, Moeller Medical, WAT Med, B Braun, ReliefBand, EmeTerm.
The market segments include Type, Application.
The market size is estimated to be USD 6.4 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Medical Low Frequency Pulse Antiemetic Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Medical Low Frequency Pulse Antiemetic Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.