1. What is the projected Compound Annual Growth Rate (CAGR) of the Medical Dry and Wet Separation Suspension Bridge Devices?
The projected CAGR is approximately 5%.
 Medical Dry and Wet Separation Suspension Bridge Devices
Medical Dry and Wet Separation Suspension Bridge DevicesMedical Dry and Wet Separation Suspension Bridge Devices by Type (Hanger Type, Siamese Type, Others, World Medical Dry and Wet Separation Suspension Bridge Devices Production ), by Application (Intensive Care Unit, Emergency Center, Others, World Medical Dry and Wet Separation Suspension Bridge Devices Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for Medical Dry and Wet Separation Suspension Bridge Devices is poised for substantial growth, projected to reach approximately $649.2 million by 2025, with a steady Compound Annual Growth Rate (CAGR) of 5% expected over the forecast period of 2025-2033. This robust expansion is primarily driven by an increasing demand for advanced healthcare infrastructure, particularly in intensive care units (ICUs) and emergency centers, where efficient patient monitoring and equipment management are paramount. The need for streamlined medical gas supply and integrated diagnostic capabilities within these critical care settings fuels the adoption of these sophisticated suspension bridge devices. Furthermore, a growing emphasis on infection control and the desire to optimize workflow for healthcare professionals are also significant contributors to market expansion. Innovations in device design, focusing on modularity, ease of installation, and enhanced safety features, are continuously shaping the market and encouraging wider adoption by healthcare facilities worldwide.
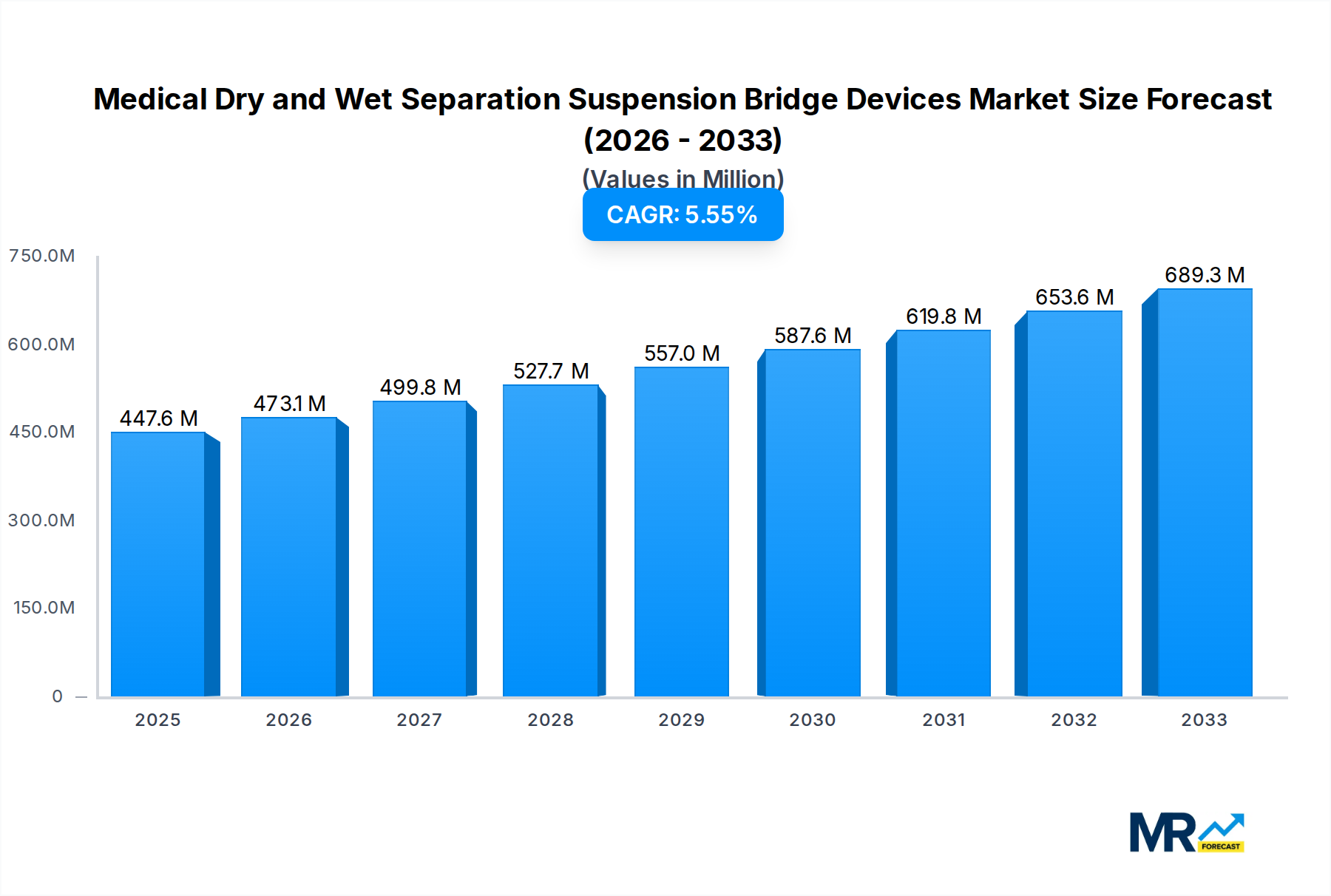

The market's trajectory is further influenced by evolving healthcare spending patterns and the continuous modernization of hospital facilities. While the market is experiencing healthy growth, certain factors could temper this expansion. The substantial initial investment required for these advanced systems and the need for specialized training for their operation and maintenance may present a challenge, particularly for smaller healthcare institutions or those in developing regions. However, the long-term benefits, including improved patient outcomes, reduced medical errors, and enhanced operational efficiency, are expected to outweigh these initial hurdles. Emerging economies are anticipated to witness a significant surge in demand as their healthcare sectors mature and adopt global best practices. The competitive landscape features a diverse range of established players and innovative newcomers, all vying to capture market share through product differentiation, strategic partnerships, and geographic expansion, all contributing to a dynamic and evolving market.
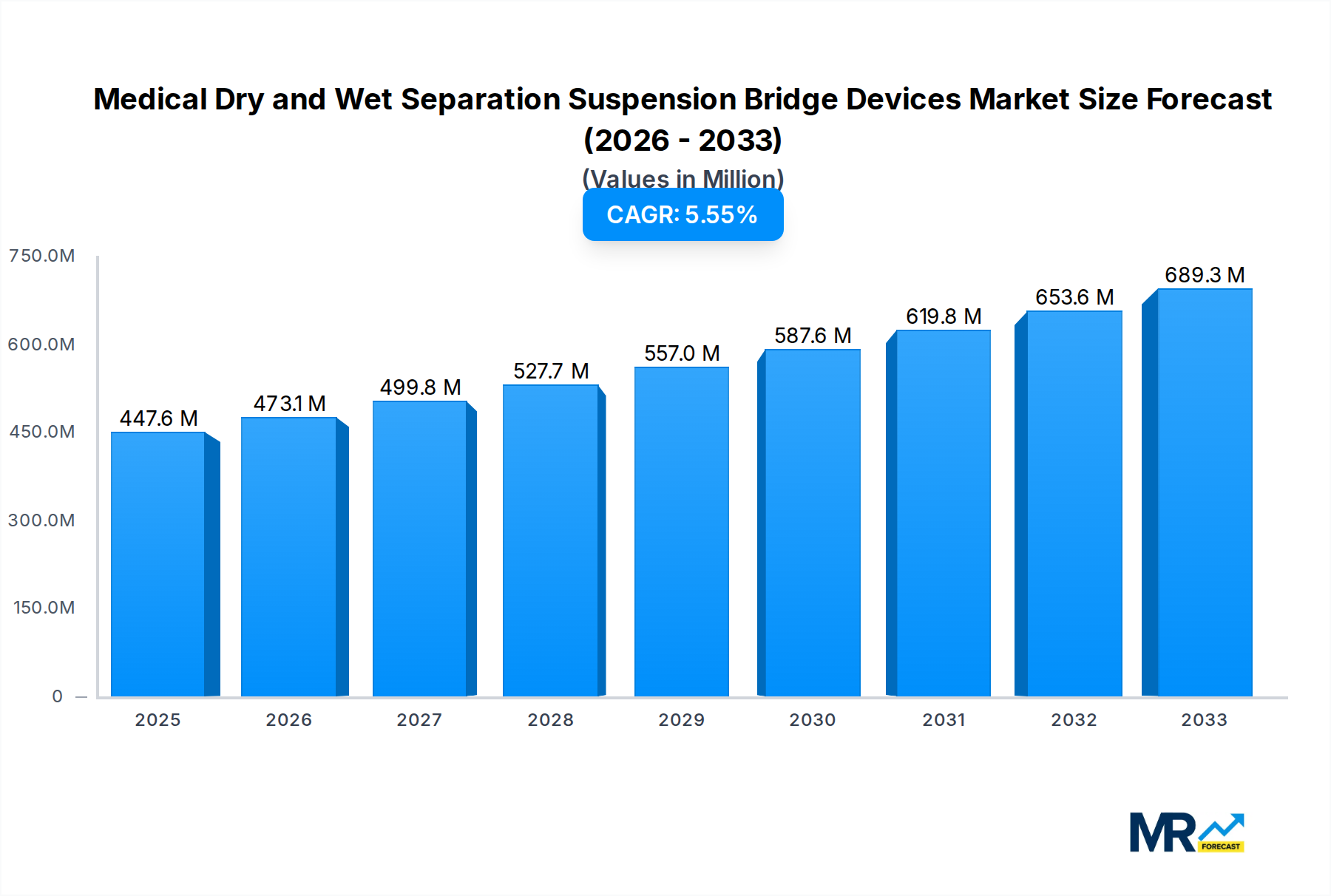

The global market for Medical Dry and Wet Separation Suspension Bridge Devices is poised for significant expansion, driven by an increasing emphasis on advanced healthcare infrastructure and the rising complexity of patient care. XXX projects a robust compound annual growth rate (CAGR) for this sector, indicating a substantial shift towards integrated medical gas supply systems that prioritize both efficiency and patient safety. The historical period from 2019 to 2024 has witnessed a steady adoption of these devices, fueled by the growing demand in critical care settings. The base year of 2025 serves as a pivotal point, with the estimated year also set at 2025, highlighting current market dynamics. Looking ahead to the forecast period of 2025-2033, the market is expected to surge, reflecting ongoing technological advancements and the increasing prevalence of acute and chronic conditions requiring sophisticated monitoring and life support. The integration of dry and wet separation capabilities is crucial, allowing for the distinct and safe management of different medical gases and fluids, thereby minimizing the risk of cross-contamination and ensuring optimal therapeutic delivery. This trend is particularly evident in the burgeoning Intensive Care Units (ICUs) and Emergency Centers, where rapid and reliable access to various medical gases is paramount. Furthermore, the global production of these devices is anticipated to scale significantly, with key players investing in enhanced manufacturing capabilities to meet the escalating demand. The market's trajectory is also influenced by evolving healthcare policies and the drive towards modernization of medical facilities worldwide. The study period from 2019 to 2033 encompasses the entire evolution and projected growth of this niche but critical segment within the medical equipment industry. The sheer volume of production, projected to be in the millions of units, underscores the widespread adoption and essential nature of these suspension bridge devices in contemporary medical practice. The report will delve into the nuances of this growth, examining the underlying drivers and the opportunities that lie ahead for manufacturers and healthcare providers alike.
The escalating demand for sophisticated patient care, particularly within critical environments like Intensive Care Units (ICUs) and Emergency Centers, stands as a primary driver for the Medical Dry and Wet Separation Suspension Bridge Devices market. As healthcare facilities worldwide grapple with an increasing burden of complex medical cases and a growing elderly population, the need for reliable, efficient, and safe medical gas and fluid delivery systems has never been more pronounced. These suspension bridge devices play a pivotal role in modern operating rooms and critical care units by centralizing essential medical utilities, thereby streamlining workflows, enhancing accessibility for medical professionals, and minimizing the risk of contamination. The inherent advantage of separating dry and wet medical utilities ensures that critical gases like oxygen and medical air are delivered with utmost purity, while simultaneously managing fluids like suction and irrigation in a segregated manner. This dual functionality is becoming indispensable for maintaining sterile environments and ensuring patient safety. Furthermore, the global push towards upgrading healthcare infrastructure, especially in emerging economies, is significantly contributing to the market's growth. Governments and private healthcare providers are investing heavily in state-of-the-art medical equipment, and these suspension bridge devices are an integral part of this modernization. The rising awareness among healthcare professionals and administrators about the benefits of integrated medical gas systems, including improved operational efficiency and reduced clutter, is also a key factor propelling the adoption of these advanced solutions. The projected production in the millions of units signifies a substantial commitment by the industry to meet this growing need for advanced medical infrastructure.
Despite the robust growth prospects, the Medical Dry and Wet Separation Suspension Bridge Devices market is not without its hurdles. One of the significant challenges lies in the substantial initial investment required for these sophisticated systems. The upfront cost of purchasing, installing, and integrating these advanced suspension bridges can be a deterrent for smaller healthcare facilities or those operating in resource-constrained regions. This can lead to a disparity in adoption rates between well-funded institutions and those with tighter budgets. Another considerable restraint is the complexity associated with the installation and maintenance of these devices. Proper integration requires specialized technical expertise and adherence to stringent medical standards and regulations. Any misstep during installation or maintenance can lead to operational disruptions, potential safety hazards, and significant repair costs, thus increasing the total cost of ownership. Furthermore, the market is susceptible to fluctuations in raw material prices, particularly for the specialized alloys and plastics used in the construction of these devices. These price volatilities can impact manufacturing costs and, consequently, the final product pricing, potentially affecting market accessibility. The rapid pace of technological evolution also presents a challenge. Manufacturers must continuously innovate and upgrade their product offerings to remain competitive, which necessitates substantial investment in research and development. Failure to keep pace with technological advancements could lead to obsolescence of existing product lines and a loss of market share. Lastly, stringent regulatory approvals and compliance requirements across different geographical regions can add to the time and cost associated with bringing new products to market, acting as a restraint on rapid expansion.
The Intensive Care Unit (ICU) segment is projected to be a dominant force in the global Medical Dry and Wet Separation Suspension Bridge Devices market. This dominance is underpinned by several critical factors that align with the core functionalities and benefits offered by these devices.
The dominance of the ICU segment underscores the essential role these suspension bridge devices play in modern, high-acuity patient care. As healthcare systems continue to prioritize patient safety, operational efficiency, and technological integration, the demand for these specialized units in ICUs will only intensify.
Several factors are acting as potent growth catalysts for the Medical Dry and Wet Separation Suspension Bridge Devices industry. The escalating global burden of chronic diseases and an aging population are driving an increased demand for advanced healthcare services, particularly in critical care settings like ICUs and emergency rooms. These environments necessitate the efficient and safe delivery of medical gases and fluids, a core function of these suspension bridges. Furthermore, the continuous technological advancements in medical equipment require integrated utility delivery systems, which these devices provide. Governments and private healthcare providers are actively investing in upgrading hospital infrastructure, leading to a surge in the adoption of modern medical gas supply solutions. The increasing focus on patient safety and infection control protocols also fuels the demand for devices that ensure the sterile separation of dry and wet utilities, directly contributing to market expansion.
This report offers a comprehensive analysis of the Medical Dry and Wet Separation Suspension Bridge Devices market, providing deep insights into its growth trajectory and future potential. The study meticulously examines key market trends, driving forces, and the challenges that shape the competitive landscape. It meticulously details the market segmentation by type, including "Hanger Type," "Siamese Type," and "Others," alongside an in-depth look at application areas such as Intensive Care Units, Emergency Centers, and other critical healthcare settings. The report quantifies market size and growth projections, utilizing data from the study period 2019-2033, with 2025 serving as both the base and estimated year. It further explores industry developments and identifies the leading market players, offering a holistic view of this vital segment of the medical equipment industry. The analysis presented will equip stakeholders with the necessary information to understand current market dynamics and capitalize on future opportunities.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5%.
Key companies in the market include Amcaremed Technology, Sostel, BiHealthcare, Oricare, Pegisdan, Kenswick, Megasan Medical Gas Systems, Pax Medical Instrument, Mediland Enterprise, Canopus, Medicop Medical Equipment, Modul Technik, Trilux Medical, Bicasa, Biolume, Wego, .
The market segments include Type, Application.
The market size is estimated to be USD 649.2 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Medical Dry and Wet Separation Suspension Bridge Devices," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Medical Dry and Wet Separation Suspension Bridge Devices, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.