1. What is the projected Compound Annual Growth Rate (CAGR) of the Mechanical Thrombectomy Devices?
The projected CAGR is approximately 11.18%.
 Mechanical Thrombectomy Devices
Mechanical Thrombectomy DevicesMechanical Thrombectomy Devices by Type (6-8 F, Others, World Mechanical Thrombectomy Devices Production ), by Application (Suction, Others, World Mechanical Thrombectomy Devices Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
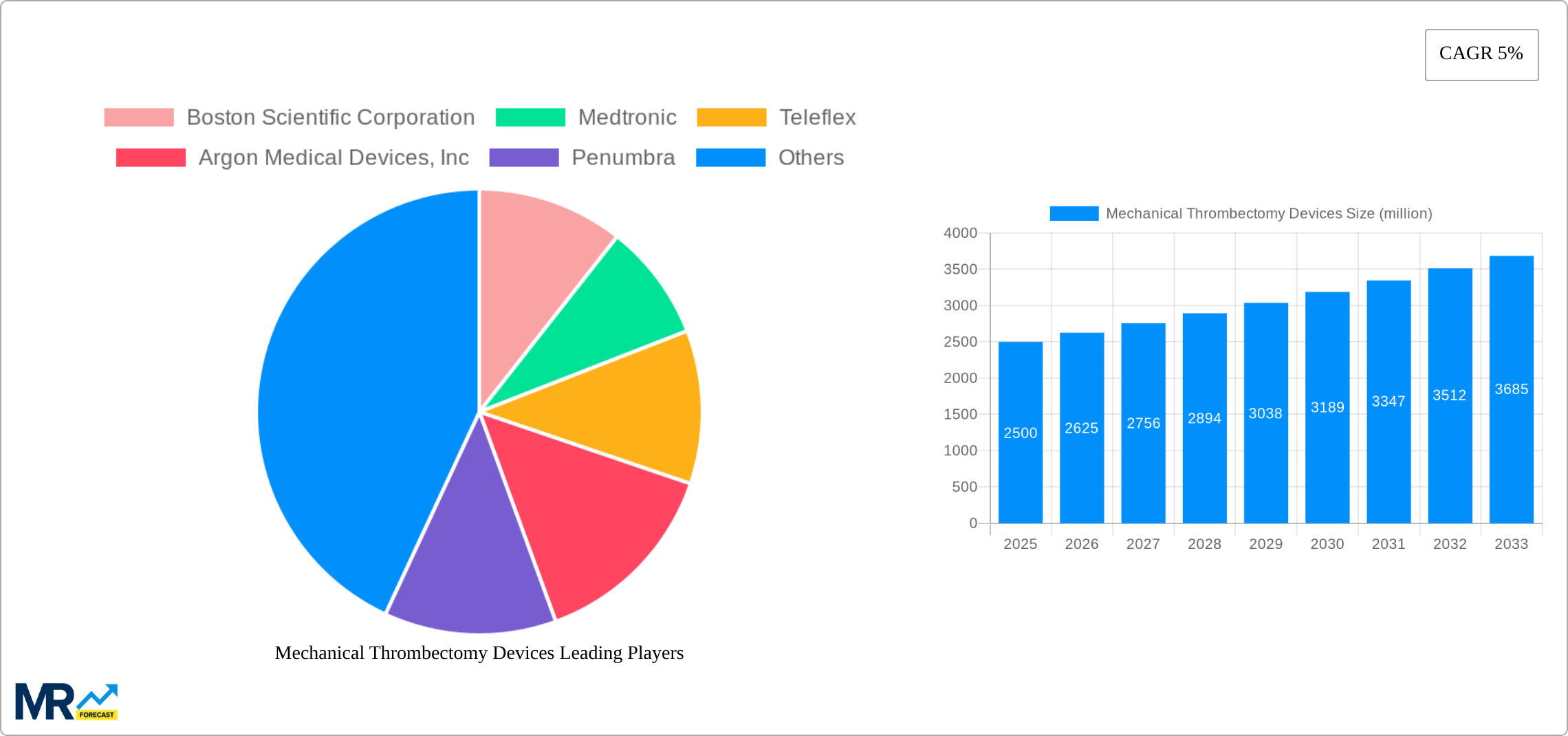
The global mechanical thrombectomy devices market is experiencing robust growth, driven by increasing prevalence of stroke and other thromboembolic diseases, advancements in device technology leading to improved treatment outcomes, and a rising geriatric population susceptible to these conditions. The market's compound annual growth rate (CAGR) of 5% reflects a steady expansion, projected to continue through 2033. Key market drivers include the increasing adoption of minimally invasive procedures, a growing awareness among healthcare professionals regarding the benefits of mechanical thrombectomy, and favorable reimbursement policies in several regions. Technological advancements, such as the development of more efficient and precise devices with improved clot retrieval capabilities, are further fueling market expansion. Segmentation by device type (e.g., aspiration catheters, stent retrievers) and application (e.g., ischemic stroke, peripheral artery disease) reveals significant variations in market share, with aspiration catheters currently dominating due to their widespread use and established efficacy. Major players in the market are engaged in intensive research and development to introduce innovative products and expand their geographical reach. Competitive pressures are driving innovation and improvements in device design, efficacy, and cost-effectiveness. Regional variations in market growth are expected, with North America and Europe maintaining a significant share due to advanced healthcare infrastructure and higher adoption rates. However, emerging markets in Asia Pacific are poised for substantial growth driven by increasing healthcare spending and rising awareness. Geographic expansion and strategic partnerships are key strategies employed by market players to enhance their market position.

Despite the positive outlook, certain restraints could moderate market growth. These include the high cost of devices, the need for specialized training and expertise for effective device usage, and potential complications associated with the procedure. However, ongoing technological advancements and a growing emphasis on cost-effectiveness are expected to mitigate these challenges. The market is likely to witness further consolidation as larger companies acquire smaller players to strengthen their product portfolios and market presence. Long-term market growth will continue to be shaped by factors such as technological innovation, regulatory approvals, and increasing accessibility of advanced medical care globally. The focus will remain on developing safer and more effective devices, optimizing treatment protocols, and ensuring broader patient access to these life-saving technologies.
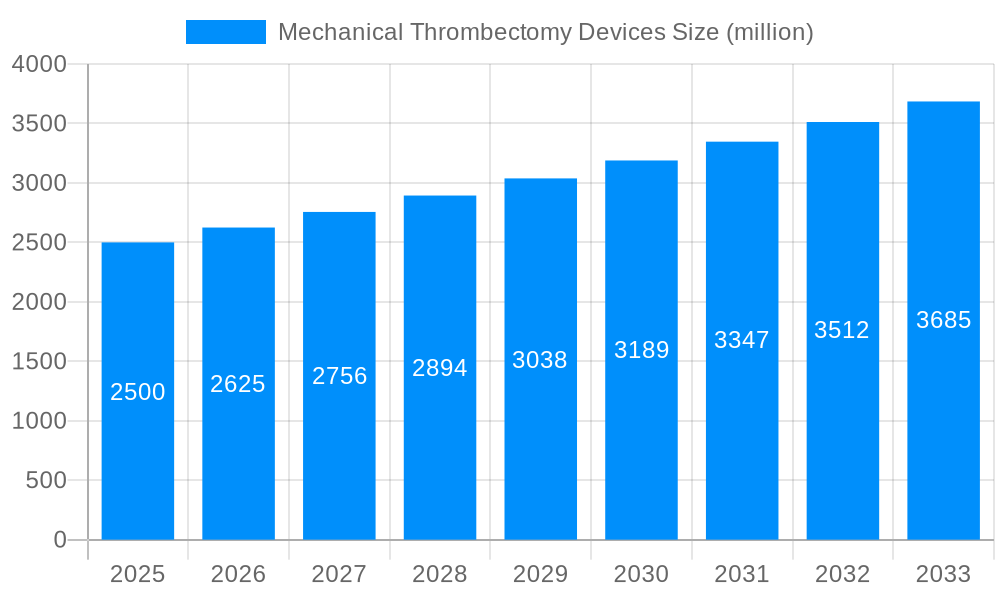

The global mechanical thrombectomy devices market is experiencing robust growth, driven by a confluence of factors. The study period from 2019 to 2033 reveals a consistent upward trajectory, with the estimated market value in 2025 exceeding several billion units. This expansion is largely attributed to the increasing prevalence of ischemic stroke, a leading cause of disability and mortality worldwide. The rising geriatric population, a key demographic susceptible to stroke, further fuels market demand. Technological advancements in device design, leading to improved efficacy and reduced procedural complications, are also significant contributors to market growth. Furthermore, the increasing adoption of minimally invasive procedures and a growing awareness among healthcare professionals regarding the benefits of mechanical thrombectomy are propelling market expansion. The forecast period (2025-2033) anticipates continued growth, with projections indicating a substantial increase in market size. This positive outlook is reinforced by ongoing research and development efforts focused on enhancing device performance and expanding therapeutic applications. Competition among key players like Boston Scientific, Medtronic, and Penumbra is driving innovation and ensuring the availability of advanced devices at competitive prices, further benefiting market expansion. The historical period (2019-2024) serves as a solid foundation for understanding the current market dynamics and predicting future trends. The base year of 2025 provides a crucial benchmark for evaluating the market’s current state and projecting future growth trajectories. Overall, the market demonstrates a promising outlook with substantial opportunities for growth in the coming years.
Several key factors are propelling the growth of the mechanical thrombectomy devices market. The escalating incidence of ischemic stroke globally is a primary driver, necessitating effective and timely treatment options. The aging global population significantly contributes to this rise in stroke cases, creating a substantial demand for advanced thrombectomy devices. Technological advancements, including the development of more efficient and minimally invasive devices, have improved treatment outcomes and reduced complications, further boosting market adoption. Increased awareness among healthcare professionals and patients regarding the benefits of mechanical thrombectomy compared to traditional treatments is also a strong driving force. Favorable reimbursement policies and increasing healthcare expenditure in several regions are enabling wider accessibility to these devices. Finally, a continuous stream of research and development efforts focused on improving device design, materials, and functionality are sustaining market growth and expanding its therapeutic applications. These combined factors ensure that the mechanical thrombectomy devices market will continue its impressive growth trajectory in the foreseeable future.
Despite the considerable growth potential, several challenges and restraints impact the mechanical thrombectomy devices market. High device costs can hinder accessibility, particularly in low- and middle-income countries. The complexity of the procedures and the need for specialized training among healthcare professionals can pose barriers to widespread adoption. Strict regulatory approvals and the lengthy timeframes involved in obtaining necessary certifications can delay market entry for new devices. Potential complications associated with mechanical thrombectomy, although rare, can create hesitancy among healthcare providers. Furthermore, the emergence of alternative treatment modalities for ischemic stroke could potentially reduce the demand for mechanical thrombectomy devices. Lastly, the reimbursement landscape can be variable across different regions, impacting market penetration and access. Addressing these challenges requires collaborative efforts among stakeholders, including regulatory bodies, healthcare providers, and device manufacturers, to ensure equitable access to this life-saving technology.
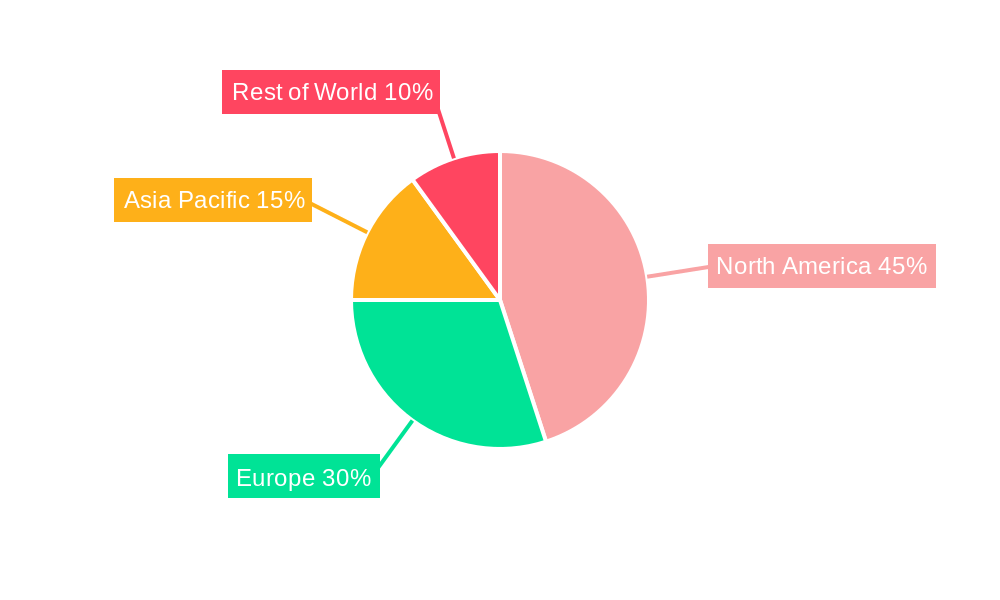
The North American market, specifically the United States, is currently leading the global mechanical thrombectomy devices market. This dominance is attributed to several factors including:
High Prevalence of Ischemic Stroke: The US has a high incidence of ischemic stroke, driving demand for effective treatment options.
Advanced Healthcare Infrastructure: A well-established healthcare infrastructure and high healthcare expenditure facilitate the adoption of advanced technologies like mechanical thrombectomy devices.
Strong Regulatory Framework: A robust regulatory framework ensures product safety and quality, fostering investor confidence and promoting market growth.
High Research & Development Activity: Significant investments in research and development within the US contribute to innovation and the development of advanced devices.
Technological Advancement: Early adoption of innovative devices in the US drives market growth.
Beyond North America, Europe is also a significant market, although slightly behind in terms of market share. Asia-Pacific is exhibiting rapid growth potential due to its increasing elderly population and improving healthcare infrastructure. The 6-8 F segment within the “Type” category dominates the market due to its compatibility with a wider range of patients and its versatility in various clinical scenarios. Similarly, the "Suction" application segment holds the largest market share within the "Application" category, reflecting the efficacy and widespread acceptance of suction-based thrombectomy techniques. The substantial market share of these segments points towards a continued focus on these key areas for both manufacturers and healthcare providers. The growth in these dominant segments is expected to be fueled by ongoing technological advancements and increasing awareness of mechanical thrombectomy’s efficacy.
The mechanical thrombectomy devices industry is experiencing significant growth fueled by several key catalysts. The rising prevalence of stroke, particularly in aging populations, creates substantial unmet needs. Technological innovations continue to improve device design and efficacy, leading to better patient outcomes. Increasing healthcare spending and favorable reimbursement policies facilitate greater access to these life-saving devices. Finally, growing awareness among both healthcare professionals and the public about the benefits of mechanical thrombectomy plays a vital role in driving market expansion.
(Note: This is a sample of potential developments. Actual developments will need to be researched based on industry publications and news sources.)
This report provides a comprehensive analysis of the mechanical thrombectomy devices market, encompassing market size, growth drivers, challenges, key players, and future outlook. The report offers valuable insights for stakeholders including manufacturers, healthcare providers, investors, and regulatory bodies, providing a strategic understanding of this rapidly evolving market. It leverages rigorous market research methodologies, including both primary and secondary data sources, to provide accurate and reliable market intelligence. The report incorporates detailed forecasts and analyses of key market segments and geographies, providing a holistic perspective on the future trajectory of this critical medical device sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.18% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 11.18%.
Key companies in the market include Boston Scientific Corporation, Medtronic, Teleflex, Argon Medical Devices, Inc, Penumbra, Zeus, Peijia Medical Limited, Koninklijke Philips, BD, Abbott, TZ Medical, Surmodics, Mermaid Medical, Inari Medical, Imperative Care.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Mechanical Thrombectomy Devices," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Mechanical Thrombectomy Devices, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.