1. What is the projected Compound Annual Growth Rate (CAGR) of the Latent Tuberculosis Infection Screening Test?
The projected CAGR is approximately 7.1%.
 Latent Tuberculosis Infection Screening Test
Latent Tuberculosis Infection Screening TestLatent Tuberculosis Infection Screening Test by Type (/> Tuberculin Skin Test (TST), Interferon Gamma Released Assay (IGRA)), by Application (/> Hospital, Diagnostic Laboratories, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
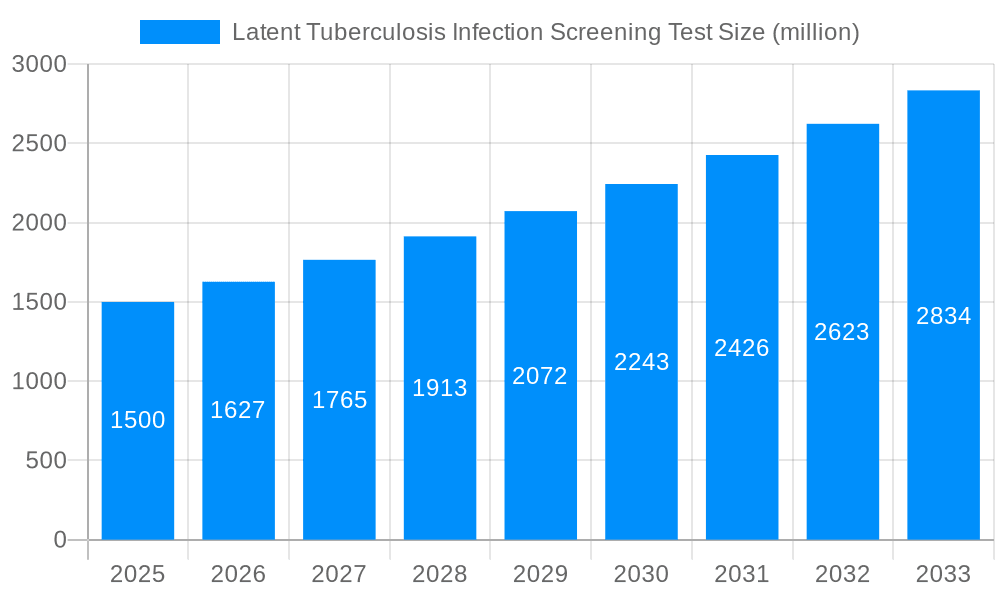
The global market for Latent Tuberculosis Infection (LTBI) Screening Tests is poised for significant expansion, driven by increasing awareness of tuberculosis (TB) prevention and diagnosis, coupled with robust government initiatives and healthcare investments worldwide. Estimated at approximately USD 1.5 billion in 2025, the market is projected to grow at a Compound Annual Growth Rate (CAGR) of around 8.5% through 2033. This growth is primarily fueled by the rising prevalence of latent TB, which affects an estimated one-quarter of the world's population, necessitating early detection and treatment to prevent progression to active disease. Advancements in diagnostic technologies, particularly the development of more accurate and accessible Interferon-Gamma Release Assays (IGRAs), are further stimulating market demand. The continuous focus on reducing TB incidence by organizations like the World Health Organization (WHO) and national public health agencies also plays a crucial role in driving the adoption of screening tests in both clinical settings and public health programs.
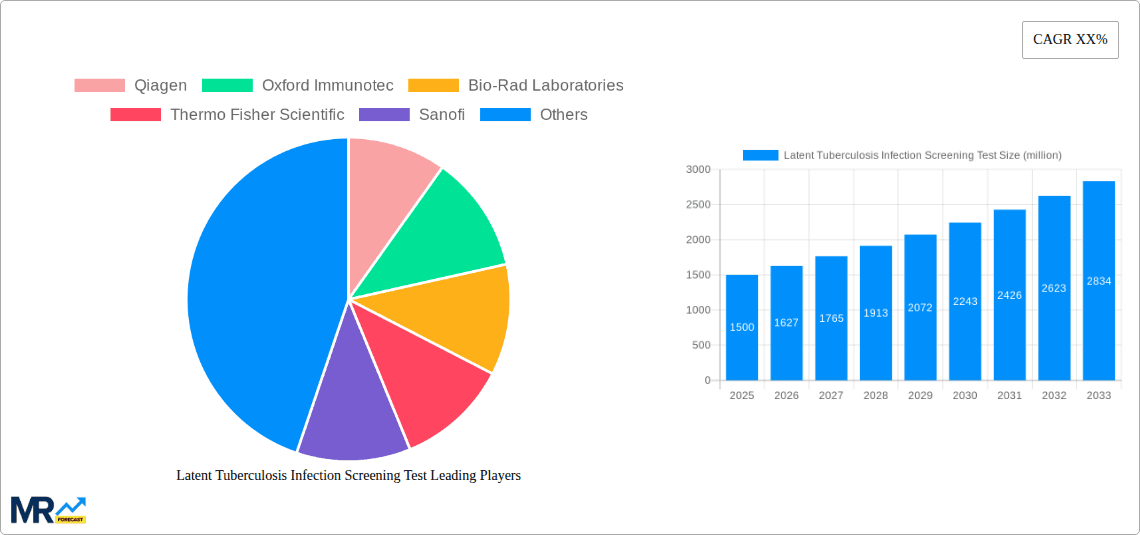

The market is characterized by a strong preference for IGRAs over traditional Tuberculin Skin Tests (TSTs) due to their higher specificity and reduced need for patient follow-up, especially in BCG-vaccinated populations. Hospitals and diagnostic laboratories represent the dominant application segments, leveraging these advanced screening tools for comprehensive patient care and public health monitoring. Geographically, North America and Europe currently lead the market, owing to well-established healthcare infrastructures and proactive TB control strategies. However, the Asia Pacific region is anticipated to witness the fastest growth, driven by a high TB burden and increasing healthcare expenditure. Despite this positive outlook, challenges such as the cost of advanced diagnostic tests and varying healthcare access in developing regions may present minor restraints. Nevertheless, the overarching global effort to eradicate TB is expected to propel the LTBI screening test market to reach an estimated USD 3.2 billion by 2033.

The Latent Tuberculosis Infection (LTBI) screening test market is poised for significant growth, driven by increasing global awareness of TB's persistent threat and advancements in diagnostic technologies. This report delves into the intricate dynamics of this vital sector, analyzing trends, drivers, challenges, regional landscapes, and key players from the historical period of 2019-2024 through to the projected forecast period of 2025-2033, with a base and estimated year of 2025.
The Latent Tuberculosis Infection (LTBI) screening test market is undergoing a transformative period, marked by a substantial expansion in demand and technological evolution. The global prevalence of latent TB, estimated to affect billions of individuals, underscores the immense need for effective screening and subsequent preventative treatment. This ongoing public health challenge is directly fueling market growth, with a projected market value reaching over 1,500 million USD by the end of the forecast period in 2033. The study period from 2019 to 2033 encompasses a comprehensive analysis, with the base and estimated year of 2025 serving as critical reference points for market valuation and projection. A significant driver within this trend is the increasing adoption of Interferon Gamma Release Assays (IGRAs) over the traditional Tuberculin Skin Test (TST). While TST remains a foundational screening method, its susceptibility to false positives due to BCG vaccination and its requirement for patient follow-up visits present limitations. Consequently, IGRAs, which offer greater specificity and convenience, are gaining substantial traction, particularly in regions with high BCG vaccination rates.
Furthermore, the report highlights a growing emphasis on proactive TB management strategies by healthcare systems worldwide. This includes expanded screening programs for high-risk populations such as immunocompromised individuals, healthcare workers, and close contacts of active TB cases. The integration of LTBI screening into routine health check-ups and pre-employment medical examinations is also contributing to the market's upward trajectory. The development of point-of-care (POC) diagnostic solutions and the expansion of laboratory infrastructure, especially in developing nations, are further augmenting market accessibility and penetration. The overall market sentiment points towards a sustained and robust expansion, driven by both the persistent burden of TB and the continuous innovation in diagnostic methodologies that improve accuracy, efficiency, and patient outcomes.
The Latent Tuberculosis Infection (LTBI) screening test market is being propelled by a confluence of critical factors that are actively shaping its trajectory. Foremost among these is the unabated global burden of tuberculosis (TB), which remains a significant public health concern, particularly in low and middle-income countries. The World Health Organization (WHO) estimates that approximately one-quarter of the world's population is infected with latent TB, presenting a vast reservoir for potential reactivation into active disease. This enormous latent population necessitates widespread and accurate screening to identify individuals at risk and initiate preventative therapy, thereby curbing the progression to active TB and reducing transmission.
Another potent driver is the increasing global commitment to TB eradication and control initiatives spearheaded by international organizations, national governments, and non-governmental agencies. These initiatives often include substantial funding for diagnostic research, procurement of screening tools, and the implementation of large-scale screening programs. This heightened focus translates directly into greater demand for LTBI screening tests. Moreover, advancements in diagnostic technologies are playing a pivotal role. The development and increasing adoption of Interferon Gamma Release Assays (IGRAs) offer distinct advantages over the traditional Tuberculin Skin Test (TST), such as improved specificity and reduced interference from prior BCG vaccination. This technological shift is making screening more accurate and reliable, thus encouraging wider use. The growing awareness among healthcare providers and the general public about the importance of LTBI management, coupled with evolving clinical guidelines that recommend routine screening for high-risk groups, are further solidifying the market's expansion.
Despite the promising growth trajectory, the Latent Tuberculosis Infection (LTBI) screening test market faces several formidable challenges and restraints that warrant careful consideration. A significant hurdle is the accessibility and affordability of advanced screening technologies, particularly in resource-limited settings where the burden of TB is often highest. While IGRAs offer superior performance, their higher cost compared to TST can be a prohibitive factor, limiting their widespread adoption in many developing regions. This economic disparity creates a bifurcated market, where advanced diagnostics are concentrated in well-funded healthcare systems, while reliance on older, less precise methods persists elsewhere.
Furthermore, the lack of standardized diagnostic protocols and follow-up procedures across different healthcare settings can lead to inconsistencies in screening and treatment initiation. This can result in underdiagnosis or delayed treatment, undermining the effectiveness of screening programs. The complex cascade of care, from screening to diagnosis, treatment initiation, and completion of preventative therapy, involves multiple touchpoints and requires robust patient management systems. Gaps in this cascade can lead to lost opportunities for intervention. Another restraint is the public perception and awareness surrounding LTBI. While awareness of active TB is generally high, understanding of latent infection and the importance of screening and preventative treatment is often lower. This can lead to patient hesitancy or a lack of perceived urgency. Finally, the ongoing global health crises and competing healthcare priorities can sometimes divert attention and resources away from TB control efforts, impacting the pace of market growth.
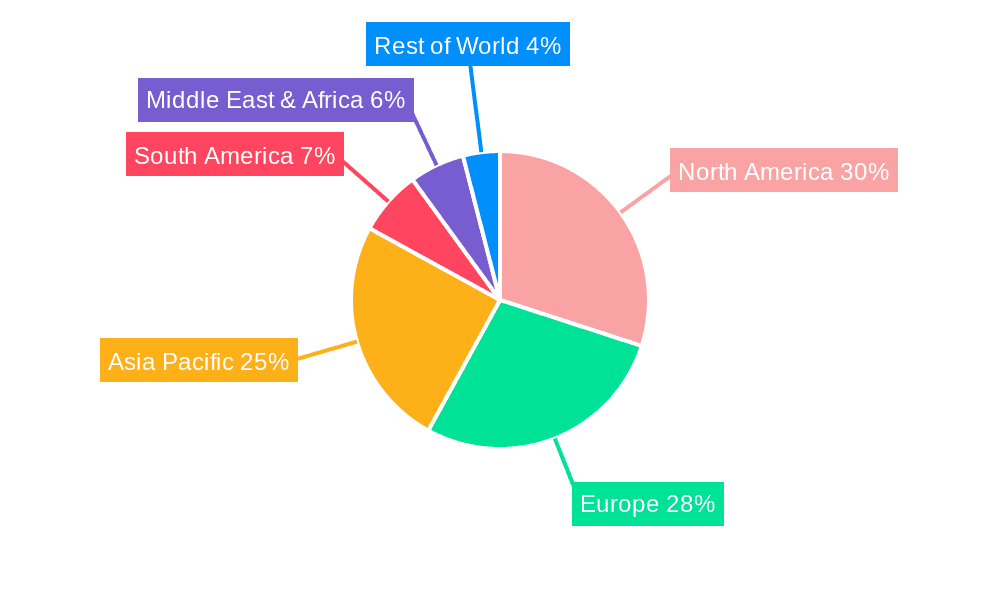
The Latent Tuberculosis Infection (LTBI) screening test market is projected to witness significant dominance by the Asia Pacific region, driven by a combination of a persistently high TB burden and a rapidly expanding healthcare infrastructure. Countries within this region, including India, China, Indonesia, and the Philippines, collectively account for a substantial proportion of the global TB cases. This immense patient population naturally translates into a high demand for screening and diagnostic solutions. Furthermore, these nations are experiencing robust economic growth, leading to increased healthcare expenditure and a greater capacity to invest in advanced medical technologies. The presence of a large, underserved population also presents a substantial market opportunity for both established and emerging diagnostic companies.
Within the segments, the Interferon Gamma Release Assay (IGRA) type is expected to emerge as the dominant force. While the Tuberculin Skin Test (TST) has historically been the cornerstone of LTBI screening due to its cost-effectiveness, its limitations, such as poor specificity in BCG-vaccinated populations and the need for repeat patient visits, are increasingly being recognized. IGRAs, such as those offered by companies like Qiagen and Oxford Immunotec, offer a more accurate and convenient alternative. Their ability to differentiate between TB infection and BCG vaccination, coupled with the single-visit convenience, makes them increasingly preferred by healthcare providers, especially in developed and increasingly in developing countries as their healthcare systems mature.
The Diagnostic Laboratories application segment is also anticipated to hold a significant share of the market. These specialized facilities are equipped with the necessary infrastructure and trained personnel to perform complex laboratory-based tests like IGRAs. As healthcare systems increasingly rely on accurate and standardized diagnostic procedures, diagnostic laboratories play a crucial role in providing these services. Their ability to handle a high volume of samples and offer a range of diagnostic tests makes them central to the LTBI screening ecosystem. While hospitals also play a vital role, particularly in initial patient contact and referral, dedicated diagnostic laboratories are becoming key hubs for performing and processing these specific tests.
The Latent Tuberculosis Infection (LTBI) screening test industry is poised for accelerated growth fueled by several key catalysts. A primary driver is the intensified global efforts and funding allocated towards TB elimination campaigns, significantly boosting the demand for effective screening tools. Furthermore, the ongoing development of more sensitive, specific, and user-friendly diagnostic technologies, including point-of-care tests and improved IGRA methodologies, is making screening more accessible and efficient. The increasing recognition of LTBI as a crucial step in preventing active TB, leading to expanded screening guidelines for high-risk populations, is also a significant growth accelerant.
This report provides an exhaustive examination of the Latent Tuberculosis Infection (LTBI) screening test market, encompassing a thorough analysis of market dynamics, segmentation, and regional landscapes. It offers critical insights into the historical performance from 2019-2024 and detailed projections through 2033, with a specific focus on the base and estimated year of 2025. The study meticulously outlines the driving forces, challenges, and emerging trends, leveraging quantitative data and expert qualitative analysis to deliver a robust market outlook. It is designed to equip stakeholders with the necessary information to understand the competitive environment, identify growth opportunities, and formulate effective business strategies in this vital public health sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.1% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7.1%.
Key companies in the market include Qiagen, Oxford Immunotec, Bio-Rad Laboratories, Thermo Fisher Scientific, Sanofi, Par Sterile, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Latent Tuberculosis Infection Screening Test," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Latent Tuberculosis Infection Screening Test, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.