1. What is the projected Compound Annual Growth Rate (CAGR) of the Labeled And Unlabeled Secondary Antibodies?
The projected CAGR is approximately 7%.
 Labeled And Unlabeled Secondary Antibodies
Labeled And Unlabeled Secondary AntibodiesLabeled And Unlabeled Secondary Antibodies by Type (Goat Anti-Rabbit Secondary Antibody, Goat Anti-Mouse Secondary Antibody, Mouse Anti-Rabbit Secondary Antibody, Donkey Anti-Rabbit Secondary Antibody, Other), by Application (ELISA, WB, IHC, ICC, Flow Cytometry, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global market for labeled and unlabeled secondary antibodies is set for substantial expansion, propelled by escalating demand in life sciences research, diagnostics, and drug discovery. With an estimated market size of USD 750 million in the base year 2025, the market is projected to grow at a robust Compound Annual Growth Rate (CAGR) of 7% through 2033. This growth is underpinned by advancements in immunoassay techniques such as ELISA, Western Blot (WB), and Immunohistochemistry (IHC), which are critical for protein detection and analysis. Key accelerators include the increasing prevalence of chronic diseases, the burgeoning biotechnology sector, and significant R&D investments by pharmaceutical and academic institutions. Furthermore, the development of novel labeling technologies offering enhanced sensitivity and specificity is driving market dynamism.
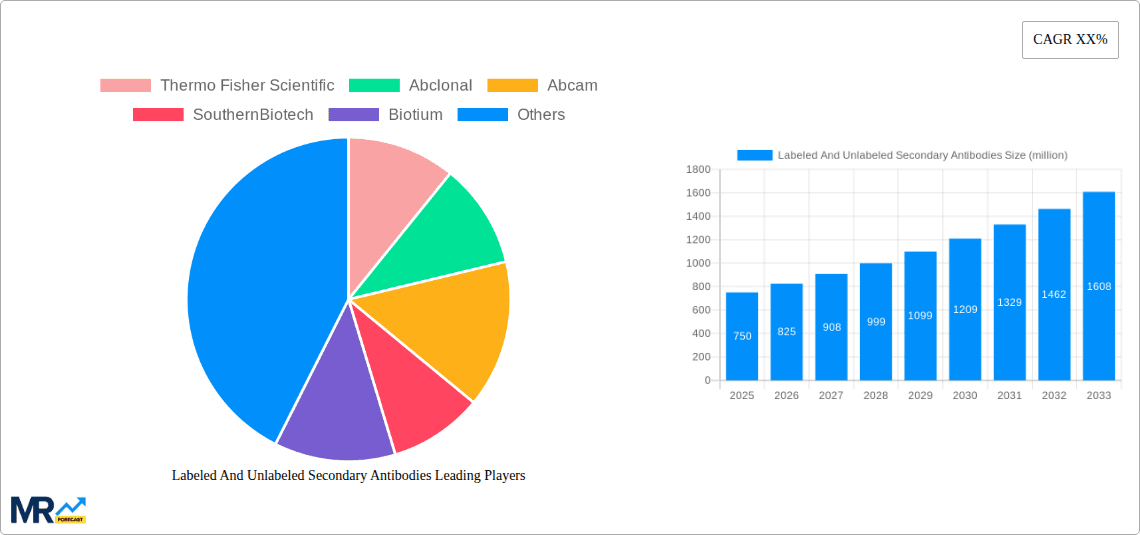

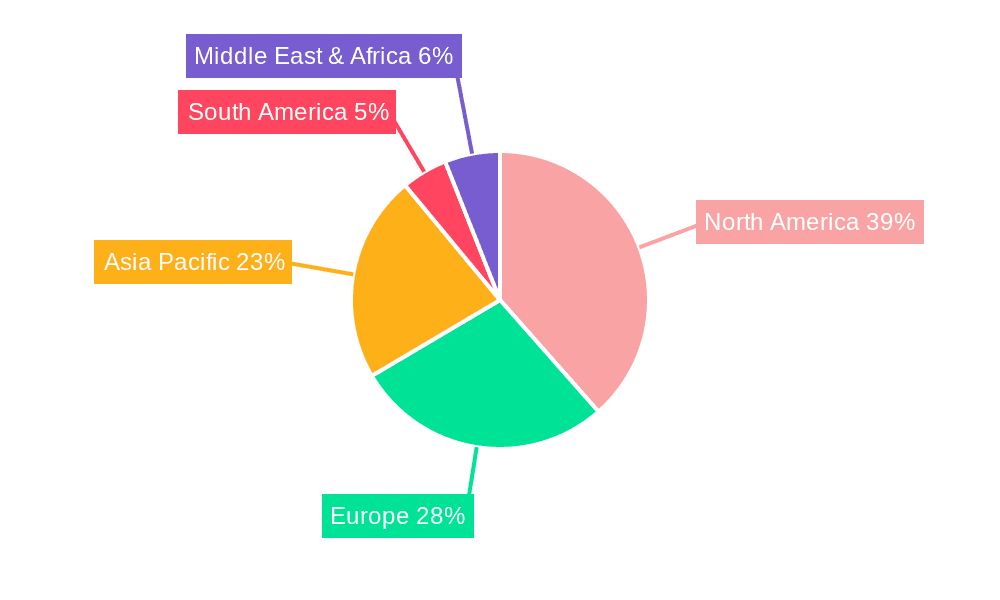
The secondary antibody market encompasses a diverse range of applications, with ELISA and WB leading demand due to their widespread use in research and clinical settings. Immunohistochemistry and Immunocytochemistry (ICC) are also experiencing steady growth, particularly driven by precision medicine and cancer research. While the market is segmented by antibody type, including high-demand Goat Anti-Rabbit and Goat Anti-Mouse secondary antibodies, the "Other" category is also expanding with specialized antibody conjugates. Geographically, North America currently leads the market, supported by a strong research infrastructure and high R&D expenditure. However, the Asia Pacific region is anticipated to exhibit the fastest growth, fueled by increasing government support for biotechnology, a rising number of research institutes, and a growing pool of skilled scientists. Market restraints, such as stringent regulatory policies and high antibody production costs, are being addressed through technological innovations and strategic collaborations.
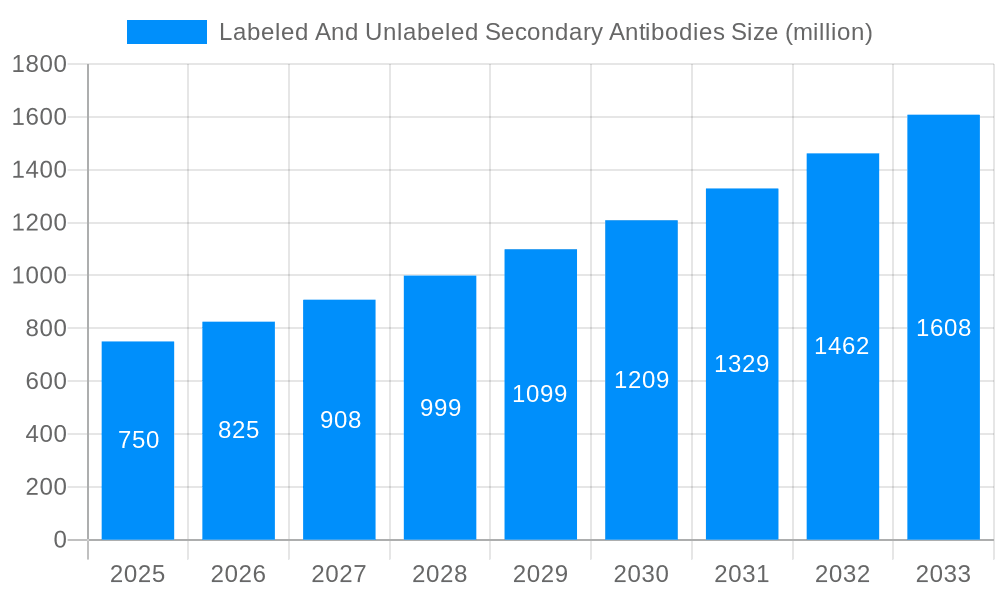

The global labeled and unlabeled secondary antibodies market is projected for substantial growth, driven by the escalating demand for advanced diagnostic and research tools. XXX estimates that the market, valued in the hundreds of millions of US dollars in the base year of 2025, is poised to witness a Compound Annual Growth Rate (CAGR) of over 10% during the forecast period of 2025-2033. This expansion is underpinned by the increasing prevalence of chronic diseases, the growing emphasis on personalized medicine, and the continuous advancements in life science research. The historical period of 2019-2024 has already laid a strong foundation for this growth, with research institutions and pharmaceutical companies heavily investing in antibody-based assays for a wide range of applications, including Western Blotting (WB), Enzyme-Linked Immunosorbent Assay (ELISA), Immunohistochemistry (IHC), and Immunocytochemistry (ICC). The need for highly specific and sensitive detection methods in these applications directly fuels the demand for both labeled and unlabeled secondary antibodies.
Furthermore, the increasing adoption of high-throughput screening and multiplexing techniques in drug discovery and development is also a significant trend. Labeled secondary antibodies, particularly those conjugated with fluorophores or enzymes, are crucial for such applications, enabling the simultaneous detection of multiple targets with remarkable precision. Unlabeled secondary antibodies, on the other hand, remain indispensable for applications requiring downstream conjugation or where signal amplification is achieved through other means, such as biotin-streptavidin systems. The study period of 2019-2033 will likely see a continued surge in demand, with the market value reaching into the billions of US dollars by the end of the forecast period. This sustained growth trajectory is a testament to the fundamental role secondary antibodies play across the entire spectrum of biological research and clinical diagnostics.
The rapid expansion of the labeled and unlabeled secondary antibodies market is primarily propelled by the surging global investments in life science research and development. Governments, academic institutions, and private companies are allocating significant resources towards understanding complex biological pathways, identifying novel drug targets, and developing innovative therapeutic interventions. This research-intensive environment necessitates highly sensitive and specific detection methods, where secondary antibodies play a pivotal role. The increasing prevalence of various diseases, including cancer, infectious diseases, and autoimmune disorders, is also a major driver. Accurate diagnosis and effective treatment strategies for these conditions heavily rely on antibody-based detection techniques, thereby boosting the demand for secondary antibodies.
Moreover, the advancements in biotechnology, particularly in areas like monoclonal antibody production and conjugation technologies, are continuously enhancing the quality and utility of secondary antibodies. The development of novel labeling chemistries and the availability of a wider array of secondary antibody specificities are further broadening their application scope. The growing adoption of personalized medicine approaches, which require detailed molecular profiling of individual patients, is also contributing to market growth. This allows for the development of targeted therapies, and secondary antibodies are essential tools for identifying the molecular biomarkers that guide these personalized treatments.
Despite the robust growth, the labeled and unlabeled secondary antibodies market faces certain challenges and restraints that could temper its expansion. One significant restraint is the high cost associated with research and development activities, which can impact the purchasing power of smaller research institutions and emerging biotech companies. The development and validation of highly specific and reliable secondary antibodies, especially for niche targets, require substantial investment, which can translate into higher product prices. Furthermore, the market is characterized by intense competition, leading to price pressures and potentially impacting the profit margins of manufacturers.
Another challenge lies in the stringent regulatory requirements associated with diagnostic applications. Ensuring the consistency, purity, and performance of secondary antibodies used in clinical diagnostics requires adherence to rigorous quality control measures and regulatory approvals, which can be time-consuming and expensive. The emergence of alternative detection technologies, although currently not as widely adopted as antibody-based methods, could also pose a long-term challenge. Finally, the ongoing need for highly specialized secondary antibodies for emerging research areas and rapidly evolving diagnostic needs requires constant innovation and adaptation from manufacturers, which can be a demanding and resource-intensive process. The complexity of antibody-based assays and the potential for non-specific binding can also lead to challenges in data interpretation, requiring careful assay design and optimization.
The North America region is poised to dominate the global labeled and unlabeled secondary antibodies market, driven by a confluence of factors including substantial investments in biotechnology research, a high concentration of leading pharmaceutical and diagnostic companies, and a robust academic research infrastructure. The United States, in particular, stands out with its extensive funding for life sciences, numerous research institutions, and a strong ecosystem for innovation in antibody development and application. The significant prevalence of chronic diseases and the increasing adoption of advanced diagnostic techniques further bolster the demand for secondary antibodies in this region.
Within this dominant region, and indeed globally, the Goat Anti-Mouse Secondary Antibody segment is expected to hold a significant market share. This is primarily due to the widespread use of mouse models in preclinical research and the extensive development of mouse-derived monoclonal antibodies, which necessitate the use of goat anti-mouse secondary antibodies for detection. The versatility of Goat Anti-Mouse Secondary Antibodies across various applications such as WB, ELISA, IHC, and Flow Cytometry solidifies its leading position.
The robust presence of key players like Thermo Fisher Scientific, Abcam, and Jackson ImmunoResearch Laboratories in North America further consolidates its leading position. Their extensive product portfolios, strong distribution networks, and commitment to innovation cater to the diverse needs of researchers and diagnostic developers in the region.
The labeled and unlabeled secondary antibodies industry is experiencing significant growth catalysts, primarily driven by the accelerating pace of advancements in life science research and diagnostics. The increasing global burden of diseases, coupled with a growing emphasis on early and accurate disease detection, is spurring substantial investment in R&D. This, in turn, fuels the demand for highly sensitive and specific immunoassay tools. Furthermore, the expanding applications of antibody-based techniques in areas like proteomics, genomics, and personalized medicine are opening up new avenues for market expansion. The continuous innovation in antibody conjugation and labeling technologies is also a crucial growth catalyst, leading to the development of more efficient and multiplexed detection systems.
This report offers a comprehensive analysis of the global labeled and unlabeled secondary antibodies market, providing in-depth insights into market dynamics, trends, and future projections. It covers a detailed examination of the market segmentation by type, application, and end-user, with a particular focus on the dominance of segments like Goat Anti-Mouse Secondary Antibodies and applications such as ELISA and WB. The report delves into the key driving forces, challenges, and restraints shaping the market landscape throughout the study period of 2019-2033. It also highlights the leading players and their significant developments, offering a holistic view of the competitive environment. The report aims to equip stakeholders with actionable intelligence to navigate this rapidly evolving market and identify lucrative growth opportunities within the billions of US dollars valuation projected by the end of the forecast period.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7%.
Key companies in the market include Thermo Fisher Scientific, Abclonal, Abcam, SouthernBiotech, Biotium, Jackson Nutrition Laboratories, LI-COR Biosciences, BD Biosciences, Santa Cruz Biotechnology, Biotyscience, Proteintech, Origene, SuZhou KeChuang Biotechnology, ACROBiosystems, Bio-Rad Antibodies.
The market segments include Type, Application.
The market size is estimated to be USD 1.5 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Labeled And Unlabeled Secondary Antibodies," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Labeled And Unlabeled Secondary Antibodies, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.