1. What is the projected Compound Annual Growth Rate (CAGR) of the Intrasaccular Device?
The projected CAGR is approximately 4.7%.
 Intrasaccular Device
Intrasaccular DeviceIntrasaccular Device by Type (Coil Devices, Flow Diversion Devices, Stent-assisted Coiling Devices, Liquid Embolic Agents), by Application (Hospital, Clinic, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
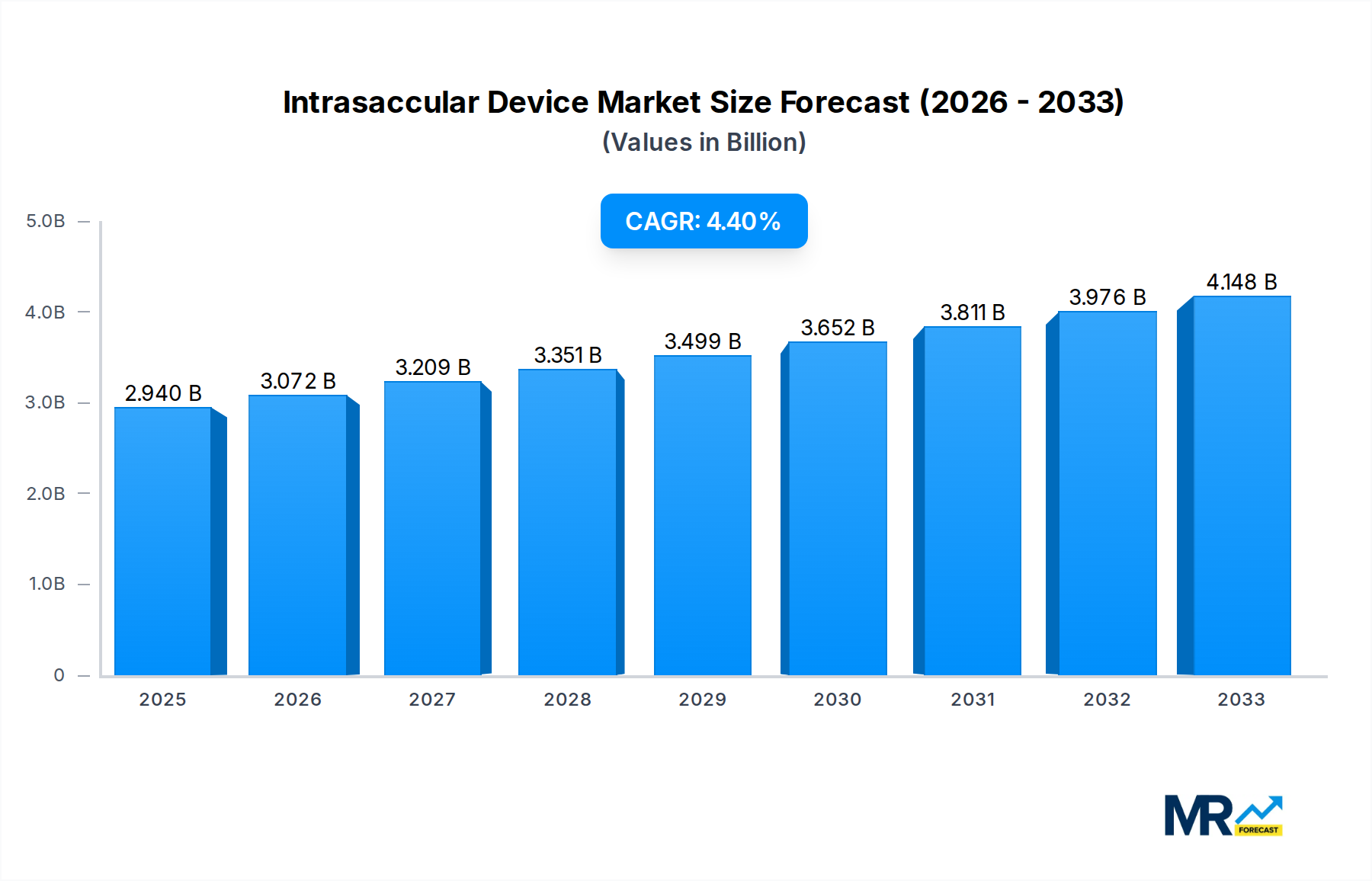
The global intrasaccular device market is poised for significant expansion, currently valued at an estimated USD 2.82 billion. This robust market is projected to grow at a compound annual growth rate (CAGR) of 4.7% through 2033, indicating sustained demand for these innovative neurovascular solutions. The increasing prevalence of cerebrovascular diseases, particularly aneurysms, coupled with advancements in minimally invasive surgical techniques, are the primary catalysts for this growth. Furthermore, a growing emphasis on patient-specific treatments and the development of novel, less invasive devices are contributing to market acceleration. The market is segmented by device type, with Coil Devices, Flow Diversion Devices, and Stent-Assisted Coiling Devices anticipated to witness substantial adoption. Liquid Embolic Agents are also emerging as a critical segment, offering alternative treatment modalities. Hospitals and clinics represent the dominant application segments, reflecting the primary settings for neurovascular interventions.

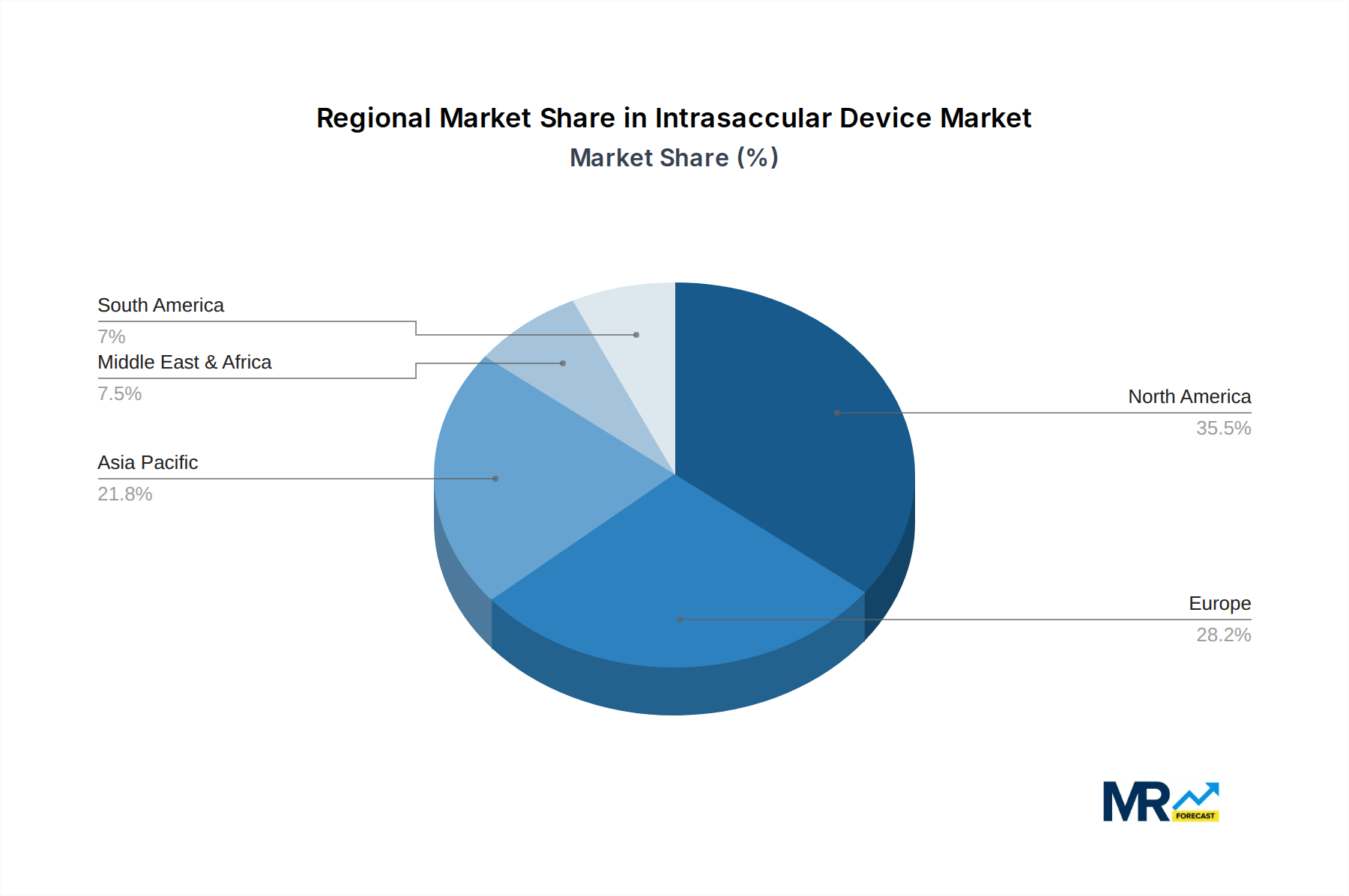
Geographically, North America currently leads the market, driven by high healthcare expenditure, advanced technological adoption, and a well-established healthcare infrastructure. Europe follows closely, with a growing awareness of minimally invasive procedures and increasing investment in neurovascular research and development. The Asia Pacific region is expected to exhibit the fastest growth, fueled by a large patient pool, improving healthcare access, and rising disposable incomes. However, restraints such as the high cost of these specialized devices and the need for specialized training for surgeons could temper growth in certain developing economies. Despite these challenges, the continuous innovation in intrasaccular device technology, focused on improved safety profiles and enhanced efficacy, will likely propel the market forward, offering new hope for patients suffering from complex neurovascular conditions.

Here is a unique report description for Intrasaccular Devices, incorporating your specified requirements:
The global intrasaccular device market is poised for remarkable expansion, projected to reach an impressive $15.2 billion by 2033. This significant growth is underscored by a compound annual growth rate (CAGR) of approximately 12.5% during the forecast period (2025-2033), building upon a solid foundation established during the historical period (2019-2024). The base year of 2025 serves as a critical pivot point, with the estimated market value for the same year already indicating a substantial $6.8 billion. Several interconnected trends are fueling this upward trajectory. The increasing prevalence of cerebrovascular diseases, particularly aneurysms, coupled with an aging global population, is a primary driver. Furthermore, advancements in minimally invasive surgical techniques and the development of more sophisticated, patient-specific intrasaccular devices are expanding treatment options and improving patient outcomes. The shift towards endovascular procedures over traditional open surgery for aneurysm treatment is also a significant contributor to market growth. Innovations in imaging technologies that enable precise visualization and navigation during deployment further enhance the efficacy and safety of these devices. The market is witnessing a dynamic interplay between established players and emerging innovators, each striving to capture market share through product differentiation and strategic collaborations. The burgeoning demand for minimally invasive treatments, driven by patient preference for reduced recovery times and lower complication rates, is creating fertile ground for the widespread adoption of intrasaccular devices across various healthcare settings. The continuous influx of research and development investments aimed at addressing unmet clinical needs, such as the treatment of complex or ruptured aneurysms, is expected to unlock new avenues for market expansion in the coming years. The evolving regulatory landscape, while presenting challenges, also encourages innovation and the development of safer, more effective therapeutic solutions.
The intrasaccular device market is experiencing a powerful surge driven by a confluence of compelling factors. Foremost among these is the escalating global burden of cerebrovascular diseases, with the incidence of brain aneurysms showing a persistent upward trend. This demographic shift, amplified by an aging population worldwide, directly translates into a larger patient pool requiring effective intervention. Concurrently, a paradigm shift in neurovascular treatment approaches is underway, favoring minimally invasive endovascular techniques over traditional open surgery. Patients and clinicians alike are increasingly recognizing the benefits of these less invasive procedures, including shorter hospital stays, reduced pain, and faster recovery times. This preference directly stimulates the demand for the sophisticated intrasaccular devices that facilitate these endovascular interventions. The relentless pace of technological innovation is another critical propellant. Manufacturers are continuously investing in research and development to create devices that offer enhanced precision, improved deliverability, and greater therapeutic efficacy. These advancements are not only expanding the range of treatable aneurysms but also improving the safety profile of the procedures. The growing awareness and diagnosis of aneurysms, partly due to improved neuroimaging capabilities, further contribute to the expanding market landscape. As more aneurysms are identified, the need for effective treatment solutions, like intrasaccular devices, escalates.
Despite the robust growth trajectory, the intrasaccular device market is not without its hurdles. A significant challenge stems from the inherent complexity and potential risks associated with endovascular procedures. While minimally invasive, these interventions still carry risks such as thromboembolism, hemorrhage, and device migration, which can lead to adverse patient outcomes. Such complications can temper the confidence of some healthcare providers and patients, potentially slowing down widespread adoption. The high cost of advanced intrasaccular devices and the associated procedural expenses can also present a restraint, particularly in resource-limited healthcare systems or for patients with inadequate insurance coverage. This cost factor can limit access to these innovative treatments for a broader segment of the population. Furthermore, the regulatory landscape, while crucial for ensuring safety and efficacy, can be a lengthy and rigorous process for new device approvals. Obtaining clearance from regulatory bodies like the FDA or EMA requires extensive clinical trials and documentation, which can delay market entry for innovative products and add to development costs. The need for specialized training and expertise among neurosurgeons and interventional radiologists to effectively deploy these complex devices also presents a practical barrier. Inadequate training infrastructure or limited access to such specialized training programs can restrict the number of qualified practitioners, thereby impacting market penetration. Lastly, ongoing research into alternative treatment modalities or the development of next-generation therapies could pose a competitive threat, potentially influencing market dynamics and investment decisions.
The global intrasaccular device market is characterized by a dynamic interplay of regional dominance and segment leadership, with certain areas and product categories poised to steer the market's future trajectory.
Key Dominant Region/Country:
North America is anticipated to emerge as a dominant force in the intrasaccular device market throughout the study period (2019-2033), with an estimated market value of $5.3 billion in 2025. Several factors contribute to this leadership:
Europe is another significant market, projected to reach $3.1 billion by 2025, driven by similar trends in aging populations, increasing disease awareness, and a commitment to advanced medical treatments.
Dominant Segment: Type of Device
Within the intrasaccular device market, Coil Devices are expected to maintain their leadership position, holding an estimated market share of 45% in 2025 and contributing approximately $3.1 billion to the overall market value. This dominance is attributed to:
Following closely are Flow Diversion Devices, projected to witness the fastest growth due to their effectiveness in treating wide-necked and complex aneurysms with a minimally invasive approach, capturing an estimated $1.9 billion by 2025. Stent-assisted Coiling Devices are also a rapidly growing segment, offering enhanced stability and coil retention for challenging aneurysms, with an estimated market of $1.2 billion in 2025. Liquid Embolic Agents, while a niche segment, are gaining traction for specific aneurysm types, contributing an estimated $0.6 billion in 2025.
Dominant Segment: Application
The Hospital segment is the primary application area for intrasaccular devices, accounting for an estimated $5.6 billion in 2025. This is due to:
The Clinic segment, while smaller, is expected to grow at a notable CAGR of 13.1%, driven by the increasing capacity of outpatient centers to handle elective procedures and a focus on cost-efficiency.
The intrasaccular device industry is experiencing robust growth fueled by several key catalysts. The escalating global incidence of cerebrovascular diseases, particularly brain aneurysms, presents a continuously expanding patient population. Technological advancements are consistently yielding safer, more effective, and minimally invasive devices, enhancing procedural success rates and patient outcomes. Furthermore, the growing preference for endovascular over open surgical interventions, driven by reduced recovery times and patient comfort, directly stimulates demand. Increased physician awareness and adoption of these advanced devices, alongside supportive reimbursement policies in key markets, are also crucial growth enablers, paving the way for wider accessibility and utilization.
This comprehensive report offers an in-depth analysis of the global intrasaccular device market, providing critical insights into market dynamics, trends, and future projections. It delves into the market's estimated value of $6.8 billion in the base year of 2025, with a robust forecast to reach $15.2 billion by 2033, demonstrating a substantial CAGR of 12.5%. The report meticulously examines the driving forces behind this growth, including the rising prevalence of cerebrovascular diseases and advancements in minimally invasive techniques. It also addresses the challenges and restraints, such as procedural risks and cost considerations. Furthermore, the report highlights key regional and segmental dominance, identifying North America as a leading market and Coil Devices as the dominant segment, while exploring the burgeoning potential of Flow Diversion Devices and Stent-assisted Coiling. Strategic analyses of growth catalysts, leading players, and significant industry developments provide a holistic view of the market landscape, making it an indispensable resource for stakeholders.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 4.7% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 4.7%.
Key companies in the market include Medtronic, MicroVention, Stryker, Terumo, Balt International, Penumbra, Acandis, Cerenovus, Phenox, Rapid Medical.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Intrasaccular Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Intrasaccular Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.