1. What is the projected Compound Annual Growth Rate (CAGR) of the Household Wearable Drug-Induced Antiemetic Device?
The projected CAGR is approximately 9%.
 Household Wearable Drug-Induced Antiemetic Device
Household Wearable Drug-Induced Antiemetic DeviceHousehold Wearable Drug-Induced Antiemetic Device by Type (Single Use, Multiple Use, World Household Wearable Drug-Induced Antiemetic Device Production ), by Application (Online Sales, Offline Sales, World Household Wearable Drug-Induced Antiemetic Device Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
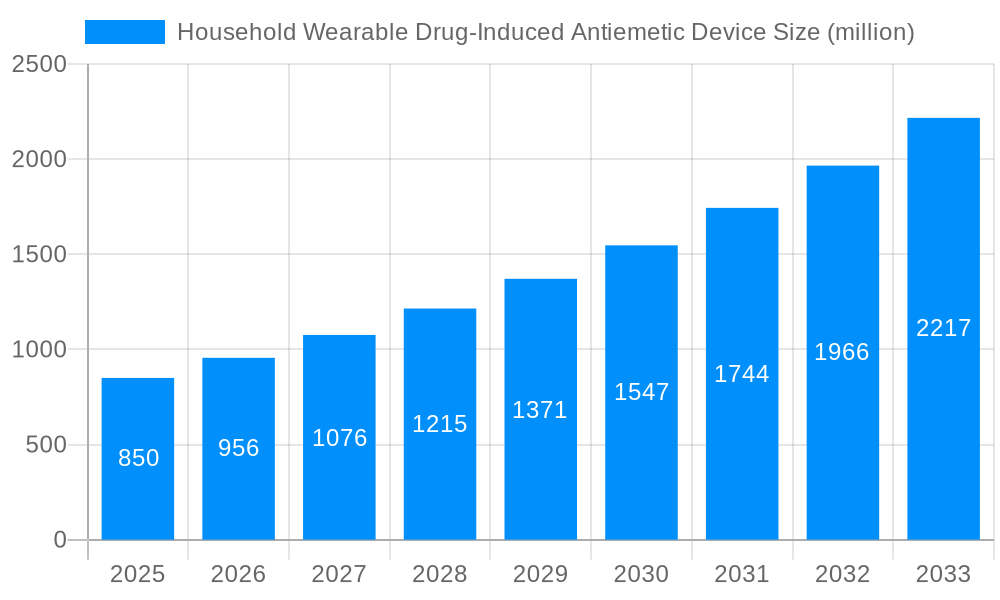
The global market for Household Wearable Drug-Induced Antiemetic Devices is experiencing robust growth, projected to reach approximately $850 million by 2025, with a compound annual growth rate (CAGR) of around 12.5% expected to continue through 2033. This expansion is primarily fueled by a growing awareness of non-invasive methods for managing nausea and vomiting, particularly those induced by medications such as chemotherapy, post-operative treatments, and motion sickness. The increasing prevalence of chronic diseases requiring long-term medication, coupled with a rising elderly population susceptible to drug-induced side effects, further propels market demand. Technological advancements leading to more comfortable, effective, and user-friendly wearable devices are also key drivers, making them an attractive alternative to traditional antiemetic drugs with their potential side effects. The market is also benefiting from a shift towards personalized healthcare solutions and a greater emphasis on patient comfort and quality of life during treatment.
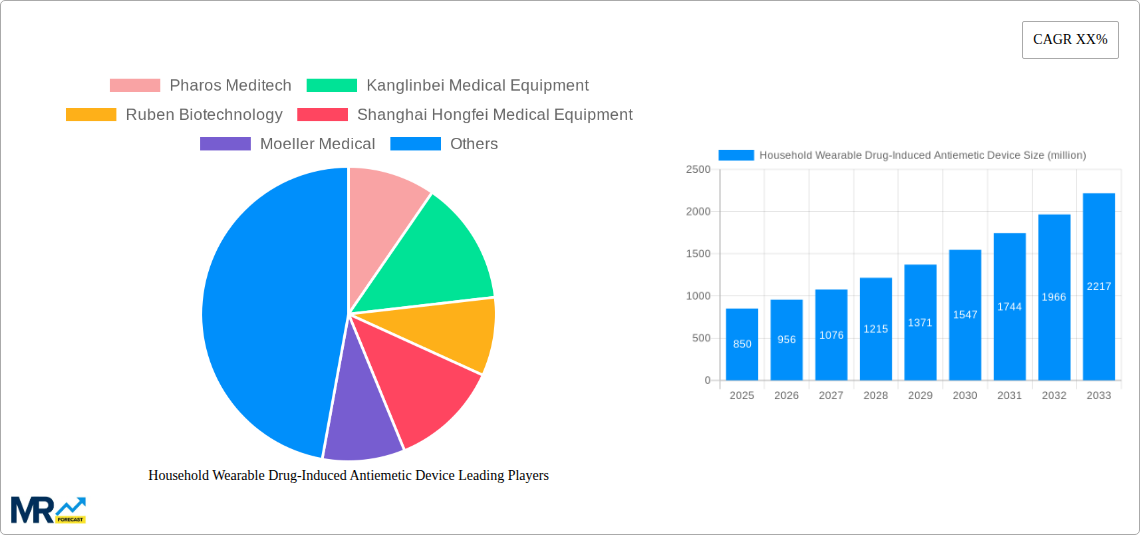

The competitive landscape is characterized by a mix of established medical device manufacturers and emerging biotech companies, all striving to innovate and capture market share. Key trends include the development of smart wearable devices with integrated tracking and personalized therapy adjustments, as well as the increasing adoption of online sales channels for wider accessibility. However, the market faces certain restraints, including the initial cost of some advanced devices and the need for greater physician and patient education regarding their efficacy and appropriate use. Geographic analysis reveals a strong presence in North America and Europe due to high healthcare spending and advanced technological adoption, while the Asia Pacific region presents significant growth potential driven by a rapidly expanding middle class and increasing healthcare infrastructure development. The development of single-use and multiple-use devices caters to diverse consumer needs and price points, further diversifying the market.

Here's a report description on Household Wearable Drug-Induced Antiemetic Devices, incorporating your specified elements:
The global market for Household Wearable Drug-Induced Antiemetic Devices is poised for significant expansion, driven by a growing awareness of motion sickness and chemotherapy-induced nausea, coupled with a desire for non-invasive and convenient relief. XXX, a key market insight, reveals a projected production volume of 55 million units in the base year of 2025, with an anticipated surge to 90 million units by 2033. This upward trajectory is underpinned by several converging trends. The increasing prevalence of conditions leading to nausea and vomiting, from routine travel to chronic illness management, is creating a sustained demand for effective antiemetic solutions. Furthermore, the technological advancements in wearable devices, focusing on miniaturization, enhanced efficacy, and user comfort, are making these devices more accessible and appealing to a broader consumer base. The shift towards personalized healthcare solutions also plays a crucial role, as individuals seek proactive and self-managed approaches to their well-being.
The study period from 2019 to 2033, with a base year of 2025, highlights a robust growth curve. The historical period (2019-2024) laid the groundwork for current market dynamics, characterized by initial adoption and product development. The forecast period (2025-2033) will witness accelerated market penetration. The market is segmenting into distinct categories, with Multiple Use devices expected to command a larger share due to their economic viability and reduced environmental impact, projected to reach 35 million units in 2025 and 60 million units by 2033. Conversely, Single Use devices will cater to specific acute needs and situations where convenience and hygiene are paramount, with production estimated at 20 million units in 2025, growing to 30 million units by 2033. The strategic importance of Online Sales is undeniable, expected to account for 65% of the total market in 2025, demonstrating a strong consumer preference for direct-to-consumer purchasing and accessibility, while Offline Sales will remain significant, particularly through pharmacies and medical supply stores, representing 35% of the market in 2025. This evolving landscape is characterized by an increasing emphasis on user experience, discreet design, and effective drug delivery mechanisms, promising a future where managing nausea and vomiting is seamlessly integrated into daily life.
Several potent forces are driving the impressive growth of the Household Wearable Drug-Induced Antiemetic Device market. A primary catalyst is the escalating global burden of conditions that induce nausea and vomiting. This includes, but is not limited to, the widespread experience of motion sickness during travel, a significant segment of the population undergoing chemotherapy, and individuals suffering from post-operative nausea and vomiting. As awareness regarding these debilitating symptoms grows, so does the demand for effective, accessible, and non-invasive relief options. Furthermore, the overarching trend towards proactive and personalized healthcare is empowering individuals to take greater control of their well-being. Wearable antiemetic devices align perfectly with this paradigm, offering a convenient, discreet, and often drug-free or minimally invasive approach to managing these symptoms without the systemic side effects associated with traditional oral medications. The increasing disposable income in developing economies, coupled with a rising health consciousness, is also contributing to the expanding consumer base for such advanced healthcare solutions.
Moreover, technological advancements are playing a pivotal role in shaping this market. Innovations in drug delivery systems, miniaturization of electronic components, and the development of ergonomic and comfortable designs are making these wearable devices more effective, user-friendly, and aesthetically appealing. The ability to deliver targeted and controlled doses of medication, or to utilize non-pharmacological mechanisms like transcutaneous electrical nerve stimulation (TENS), is enhancing the therapeutic outcomes and patient satisfaction. The growing acceptance of wearable technology across various health and wellness applications further normalizes the use of these specialized devices, paving the way for broader adoption. The increasing emphasis on a patient-centric approach in healthcare also encourages the development and commercialization of products that offer convenience and empower patients in their treatment journey.
Despite the promising growth trajectory, the Household Wearable Drug-Induced Antiemetic Device market is not without its hurdles. One significant challenge lies in the regulatory landscape. Obtaining approvals for new medical devices, especially those involving drug delivery, can be a complex, time-consuming, and expensive process. The stringent requirements for safety, efficacy, and quality control necessitate substantial investment in research, development, and clinical trials, which can deter smaller players and slow down market entry for innovative products. Another restraint is the consumer perception and education. While awareness is growing, a segment of the population may still be hesitant to adopt wearable medical devices, preferring traditional methods or lacking understanding of the technology's benefits and mechanisms. Educating consumers about the efficacy, safety, and convenience of these devices is crucial for widespread adoption.
Furthermore, cost and affordability can act as a barrier for some consumers, particularly in markets with lower disposable incomes or for individuals who require multiple devices or ongoing use. While Multiple Use devices offer long-term cost savings, the initial investment might still be prohibitive. The development of effective and targeted drug formulations or stimulation patterns specifically for wearable devices requires ongoing research and innovation. Ensuring the longevity and durability of the devices themselves, especially for Multiple Use models, also presents manufacturing challenges. Finally, the competition from established antiemetic therapies, such as oral medications, is a persistent challenge. These traditional treatments have a long history of use and are often readily available, requiring wearable devices to demonstrate a clear and significant advantage in terms of efficacy, side effect profile, or user experience to gain market share.
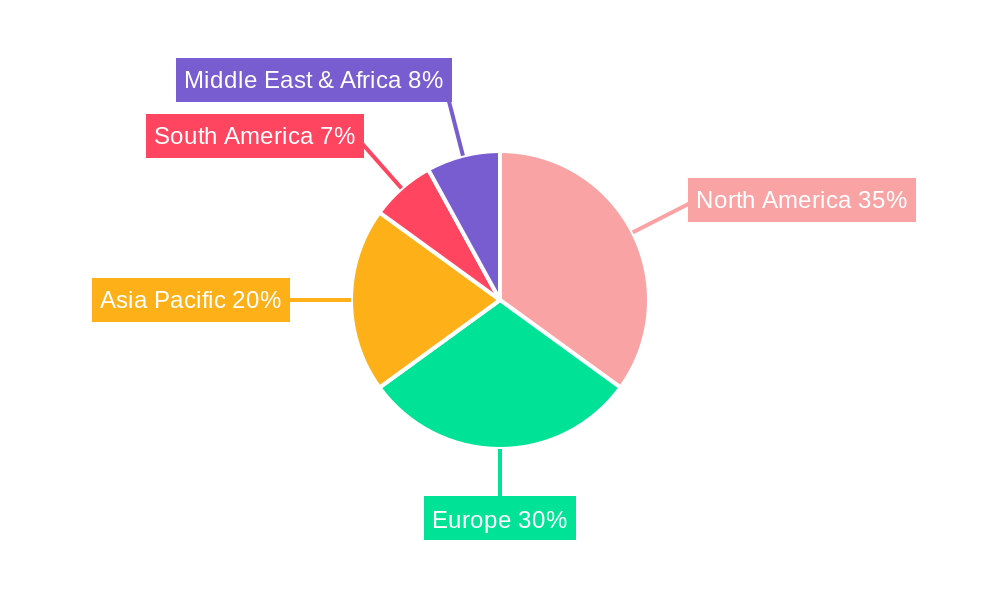
The global market for Household Wearable Drug-Induced Antiemetic Devices is projected to witness dominance by specific regions and segments, driven by a confluence of factors including population demographics, healthcare infrastructure, technological adoption rates, and prevailing health trends.
North America is anticipated to emerge as a leading region in this market. This dominance is attributed to several key drivers:
Within the Segments, the Multiple Use category is expected to lead the market's volume and value growth.
Furthermore, the Online Sales application segment is poised for significant dominance.
In terms of World Household Wearable Drug-Induced Antiemetic Device Production, the combined force of these factors points towards a robust market. In the base year of 2025, North America is projected to account for approximately 30% of global production, driven by strong demand and an advanced market ecosystem. The Multiple Use segment is expected to represent around 60% of the total production volume in 2025, while Online Sales will constitute approximately 65% of the distribution channels. By the forecast period ending 2033, these figures are expected to grow, with North America solidifying its leadership and the Multiple Use and Online Sales segments further expanding their market share due to increasing consumer adoption and technological advancements. The continuous innovation in drug delivery mechanisms and the growing emphasis on patient-centric care will further solidify the dominance of these regions and segments.
Several key growth catalysts are fueling the expansion of the Household Wearable Drug-Induced Antiemetic Device industry. A primary driver is the increasing prevalence of nausea and vomiting associated with motion sickness, chemotherapy, and post-operative recovery, creating a sustained demand for effective solutions. The escalating global adoption of wearable technology for health and wellness applications is normalizing the use of such devices. Furthermore, advancements in drug delivery systems and non-pharmacological therapeutic modalities are enhancing device efficacy and user experience. The growing trend towards personalized healthcare and self-management of health conditions also empowers consumers to seek convenient and discreet solutions like wearable antiemetics.
This comprehensive report delves deep into the multifaceted landscape of the Household Wearable Drug-Induced Antiemetic Device market. It provides an in-depth analysis of market trends, growth drivers, and potential challenges, offering strategic insights for stakeholders. The study meticulously examines the global production volume, projected to reach 55 million units in 2025 and expand significantly by 2033, with a detailed breakdown of Single Use and Multiple Use segments. Furthermore, the report highlights the critical role of Online Sales and Offline Sales channels in market penetration. With a study period spanning from 2019 to 2033, the report offers a robust historical perspective and a reliable forecast, enabling businesses to make informed decisions regarding product development, market entry, and investment strategies. It also provides a thorough overview of key industry developments and leading players, offering a holistic understanding of this dynamic sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 9%.
Key companies in the market include Pharos Meditech, Kanglinbei Medical Equipment, Ruben Biotechnology, Shanghai Hongfei Medical Equipment, Moeller Medical, WAT Med, B Braun, ReliefBand, EmeTerm.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Household Wearable Drug-Induced Antiemetic Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Household Wearable Drug-Induced Antiemetic Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.