1. What is the projected Compound Annual Growth Rate (CAGR) of the HIV Self-Test Kits?
The projected CAGR is approximately 7.5%.
 HIV Self-Test Kits
HIV Self-Test KitsHIV Self-Test Kits by Type (Blood, Urine, Oral Fluid), by Application (Hospitals, Diagnostic Laboratories, Specialty Clinics, Research Institutes, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global HIV self-test kit market is poised for substantial growth, projected to reach an estimated USD 232.9 million by 2025, expanding at a robust Compound Annual Growth Rate (CAGR) of 7.5% from 2019 to 2033. This upward trajectory is primarily driven by increasing global awareness surrounding HIV prevention and early detection, coupled with the growing demand for accessible and discreet testing solutions. Governments and non-governmental organizations are actively promoting self-testing as a crucial component of HIV management strategies, particularly in regions with limited healthcare infrastructure or stigma associated with traditional testing centers. The convenience and privacy offered by these kits empower individuals to take proactive control of their sexual health, leading to increased adoption rates. Advancements in diagnostic technology are also contributing to the market's expansion, with newer kits offering improved accuracy and faster results, further enhancing user confidence and market appeal.
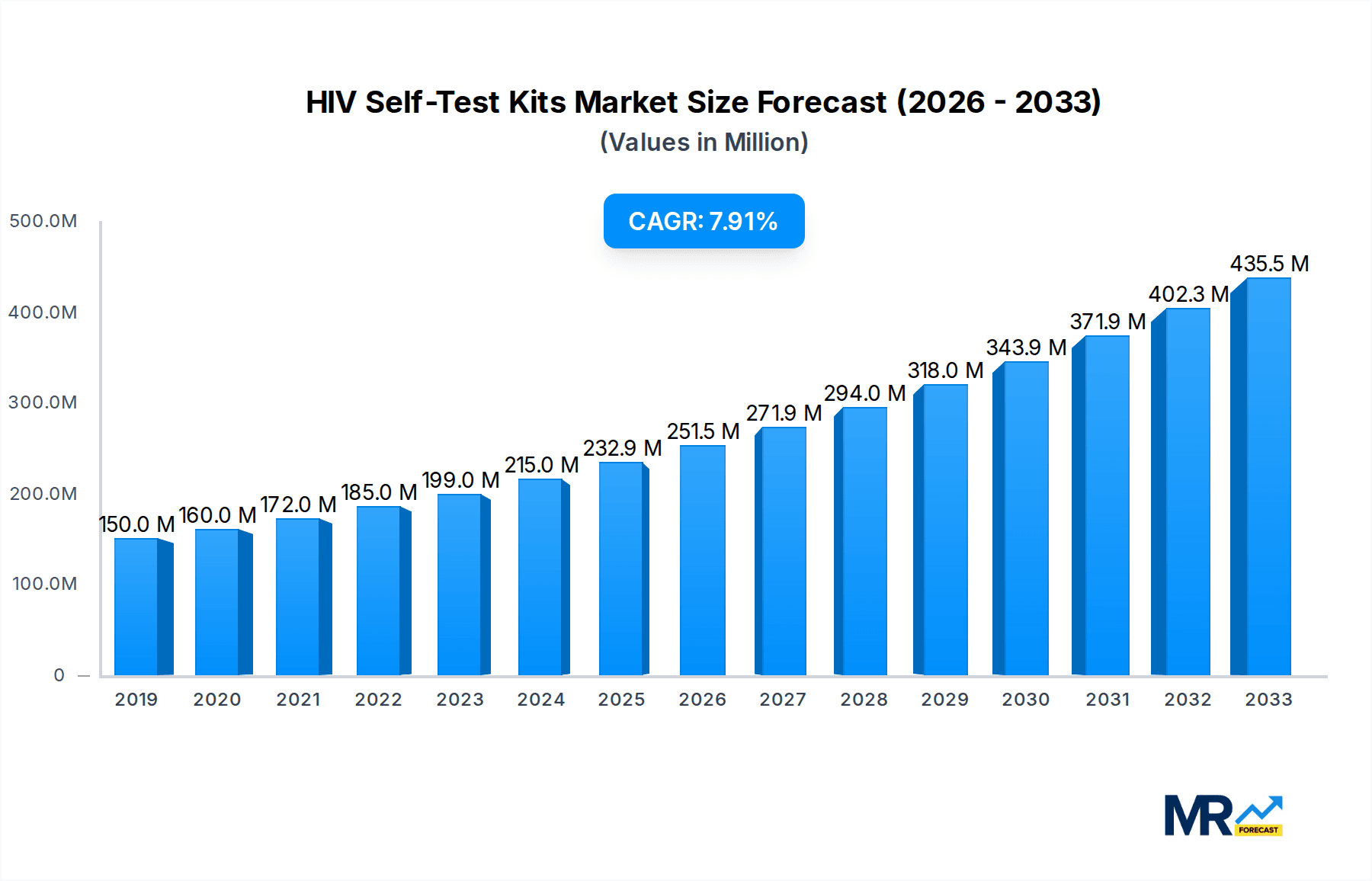

The market's growth is further supported by evolving consumer preferences and a rising comfort level with at-home diagnostic tools. The COVID-19 pandemic also played a significant role in accelerating the acceptance and adoption of telehealth and remote diagnostic solutions, including self-testing kits. Key segments driving this growth include blood and oral fluid-based tests, with hospitals, diagnostic laboratories, and specialty clinics being major application areas. While the market faces some restraints such as varying regulatory landscapes and the need for continuous public education to combat misinformation, the overwhelming positive trends and strategic initiatives in place are expected to propel the HIV self-test kit market to new heights. The competitive landscape features established players and emerging innovators, all vying to capture market share through product innovation, strategic partnerships, and expanding distribution networks to reach diverse populations globally.

This comprehensive report delves into the dynamic global market for HIV self-test kits, offering an in-depth analysis from the historical period of 2019-2024 to an estimated future outlook extending to 2033. With a base year set for 2025, the report meticulously examines market trends, driving forces, challenges, and key growth catalysts. It provides granular insights into regional dominance and segment-specific performance, supported by a detailed review of industry developments and leading market players. The study employs a rigorous methodology to project market values in the millions, offering a robust understanding of the sector's trajectory.
The global HIV self-test kit market is on an unprecedented growth trajectory, driven by a confluence of increasing awareness, advancements in testing technologies, and a growing emphasis on personal health management. From a market size of approximately \$750 million in 2019, the sector experienced steady expansion, culminating in an estimated \$1,200 million by the end of the historical period in 2024. The base year of 2025 is projected to see this market reach a substantial \$1,500 million, setting the stage for continued robust growth. The forecast period, from 2025 to 2033, anticipates a compound annual growth rate (CAGR) that will propel the market value to well over \$4,500 million by the end of the study. This surge is underpinned by several key trends. Firstly, the decentralization of HIV testing is a significant factor, as self-test kits empower individuals to take control of their health in the privacy and convenience of their homes, thereby reducing the stigma associated with traditional clinic-based testing. Secondly, technological innovations are continuously improving the accuracy, speed, and user-friendliness of these kits, making them more accessible to a wider population. The shift towards point-of-care diagnostics, where self-testing plays a pivotal role, is reshaping the healthcare landscape. Furthermore, government initiatives and non-governmental organization (NGO) efforts focused on increasing HIV testing rates, particularly among vulnerable populations, are creating sustained demand. The rising prevalence of HIV in certain demographics, coupled with the critical importance of early diagnosis for effective treatment and prevention, further fuels market expansion. The market is also witnessing a diversification in the types of self-test kits available, catering to different sample types and user preferences. This includes a growing interest in oral fluid-based tests, which offer a less invasive and simpler testing experience compared to blood-based methods. The increasing integration of digital health solutions, such as mobile applications for test result interpretation and follow-up care, is also enhancing the overall utility and adoption of HIV self-test kits. The market's evolution reflects a broader societal move towards preventative healthcare and individual empowerment in managing chronic conditions.
Several powerful forces are actively propelling the global HIV self-test kit market forward. A primary driver is the escalating global awareness surrounding HIV/AIDS, coupled with concerted efforts to achieve the UNAIDS 95-95-95 targets (95% of people living with HIV knowing their status, 95% on antiretroviral therapy, and 95% virally suppressed). Self-test kits offer a critical pathway to reaching the first 95, especially individuals who might otherwise face barriers to traditional testing due to geographical distance, cost, stigma, or lack of healthcare infrastructure. The inherent privacy and convenience afforded by self-testing are paramount, allowing individuals to undergo screening discreetly and at their own pace. This is particularly crucial in regions where societal stigma surrounding HIV remains a significant concern, deterring many from seeking clinical diagnosis. Furthermore, the continuous innovation in diagnostic technology is a relentless propellant. Manufacturers are consistently developing more accurate, faster, and user-friendly test kits, often with improved sensitivity and specificity, thereby increasing confidence in the results. The accessibility of these kits is also expanding, moving beyond traditional healthcare channels to pharmacies, community centers, and online platforms, making them readily available to a broader demographic. Government and public health organizations worldwide are increasingly recognizing the value of self-testing as a complementary strategy to national HIV testing programs, often supporting their distribution and promotion through subsidies and public awareness campaigns. This strategic endorsement significantly bolsters market growth. Finally, the growing emphasis on personalized healthcare and proactive health management by individuals is a fundamental shift that favors the adoption of self-test kits, empowering individuals to take a more active role in their well-being.
Despite the burgeoning growth, the HIV self-test kit market faces several significant challenges and restraints that could temper its full potential. A primary concern revolves around the issue of accurate interpretation of results, especially for individuals with limited literacy or without professional guidance. While test kits are designed for ease of use, the nuances of result interpretation can sometimes lead to confusion, anxiety, or incorrect self-diagnosis. This can result in a lack of linkage to care for those who test positive, or undue stress for those who misinterpret a negative result. Another substantial barrier is the ongoing challenge of stigma, even with the advent of self-testing. While kits aim to reduce stigma, residual societal biases can still discourage individuals from purchasing or using them, or from seeking follow-up care. Inadequate linkage to confirmatory testing and continuum of care remains a critical challenge; a positive self-test result is only the first step, and ensuring individuals access timely and appropriate medical follow-up is crucial for effective treatment and prevention. The cost of some self-test kits can also be a restraint, particularly in low-income settings where affordability is a major concern. While prices are decreasing, they may still be prohibitive for a significant portion of the target population. Regulatory hurdles and the need for robust quality assurance across different markets can also slow down market penetration and adoption. The varying regulatory frameworks in different countries can create complexities for manufacturers aiming for global distribution. Lastly, there's the potential for misuse or improper disposal of test kits, raising concerns about environmental impact and the integrity of testing data, although these are generally minor concerns in the overall market context. Addressing these challenges through education, improved kit design, accessible follow-up services, and supportive policy frameworks will be vital for the sustained and equitable growth of the HIV self-test market.
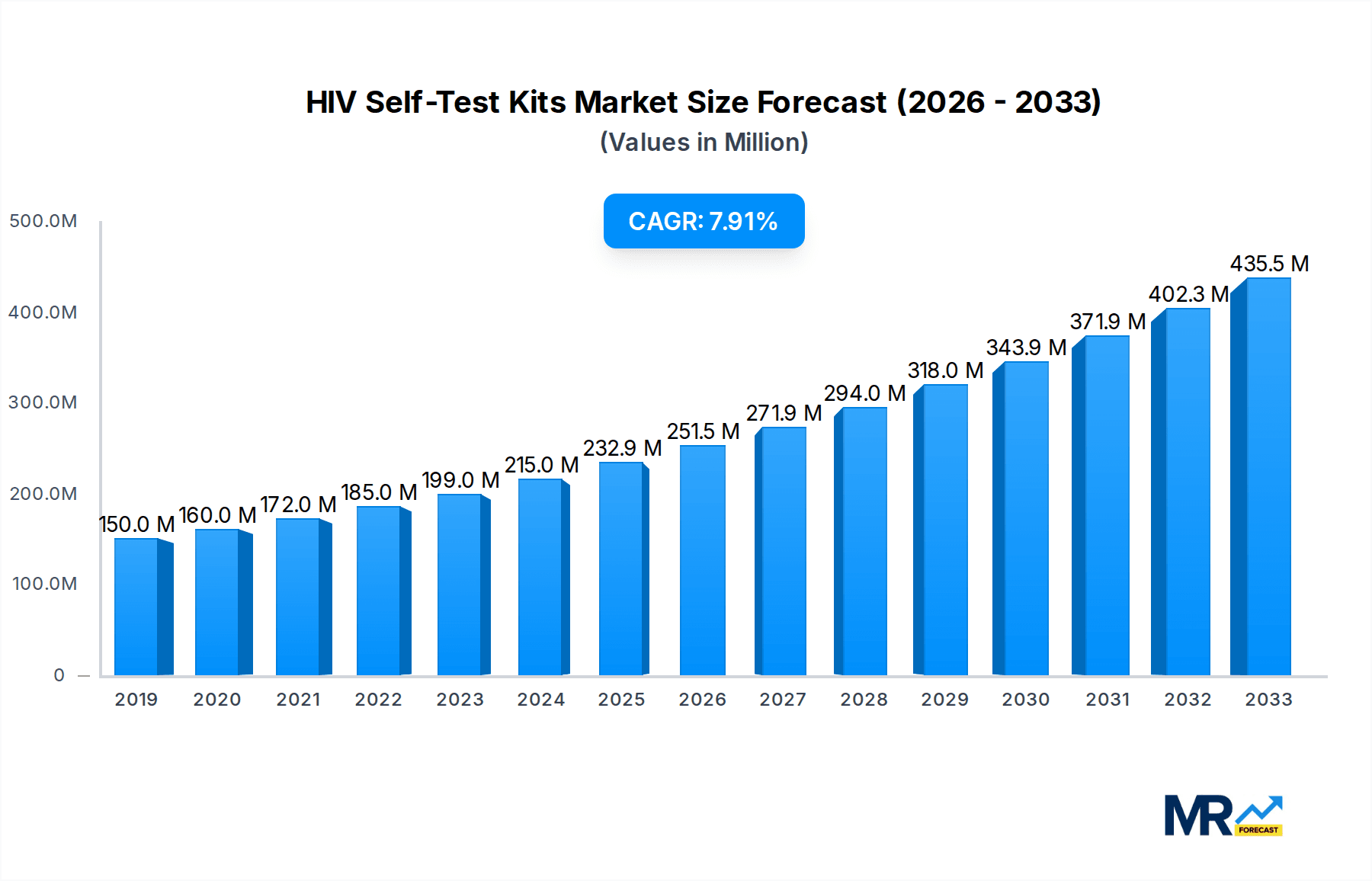
The global HIV self-test kit market is characterized by significant regional variations and segment-specific dominance. While North America and Europe have historically led in terms of market value and adoption due to established healthcare systems and high awareness levels, Asia Pacific is emerging as a key region poised for substantial growth and potential market domination in the coming years. This surge is driven by a combination of factors including a large and growing population, increasing prevalence of HIV in certain sub-regions, and a growing focus on public health initiatives aimed at early detection. Countries like India, with its vast population and ongoing efforts to control the epidemic, represent a massive untapped market for self-test kits. Similarly, Southeast Asian nations are witnessing a rise in HIV testing programs that are increasingly incorporating self-testing strategies. The affordability and accessibility of these kits are becoming paramount in these regions, leading to a greater demand for cost-effective solutions.
Within the Type segment, Oral Fluid-based HIV self-test kits are expected to increasingly dominate the market. While blood-based kits have been prevalent, oral fluid tests offer a less invasive, more user-friendly, and often faster alternative. This ease of use makes them particularly appealing for self-testing scenarios, reducing potential barriers related to needle phobia or the need for trained personnel to collect blood samples. As technology advances, the accuracy and reliability of oral fluid tests continue to improve, making them a preferred choice for a wider demographic seeking convenient and discreet HIV screening.
In terms of Application, while Hospitals and Diagnostic Laboratories will continue to be significant end-users, the Others segment, which broadly encompasses community-based organizations, pharmacies, and direct-to-consumer sales, is anticipated to be a major driver of volume and growth, especially in regions with developing healthcare infrastructures. The ability to distribute self-test kits through non-traditional channels allows for broader reach and accessibility, particularly to hard-to-reach populations and those who might not regularly engage with formal healthcare settings. This decentralized approach is crucial for increasing testing coverage and achieving public health goals.
The growth in Asia Pacific is further fueled by supportive government policies and increased funding for HIV prevention and control programs. The rising disposable incomes in some of these nations also contribute to the affordability of self-test kits. As awareness about the benefits of early diagnosis grows, and as the stigma associated with HIV gradually diminishes, the demand for accessible and user-friendly testing solutions like oral fluid self-test kits distributed through a wide network of channels, including the "Others" application segment, will undoubtedly solidify Asia Pacific's position as a dominant force in the global HIV self-test kit market.
Several key growth catalysts are propelling the HIV self-test kits industry forward. The increasing global focus on achieving UNAIDS targets, particularly increasing the number of people who know their HIV status, is a major driver. Technological advancements leading to more accurate, rapid, and user-friendly kits are enhancing adoption. Furthermore, supportive government policies and public health initiatives aimed at expanding HIV testing access are crucial. The growing awareness and acceptance of self-testing as a viable and stigma-reducing method are also significant. Finally, the expansion of distribution channels beyond traditional healthcare settings, including pharmacies and online platforms, is increasing accessibility for a broader population.
This comprehensive report provides an in-depth market analysis of HIV self-test kits, offering insights from 2019 to 2033. It examines market size in millions, covering historical trends, base year projections for 2025, and a detailed forecast period from 2025 to 2033. The report delves into driving forces, challenges, regional dominance, and key segments like blood, urine, and oral fluid tests, and applications in hospitals, diagnostic laboratories, specialty clinics, research institutes, and others. It also highlights significant industry developments and leading market players. The objective is to equip stakeholders with a robust understanding of the market dynamics, enabling informed strategic decision-making.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7.5%.
Key companies in the market include Atomo Diagnostics, BioSURE United Kingdom Ltd., Biosynex Group, bioLytical Laboratories, OraSure Technologies, Chembio Diagnostic Systems, Orangelife Comércio e Indústria, Bedford Biotech Nigeria, Sedia Biosciences Corporation, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "HIV Self-Test Kits," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the HIV Self-Test Kits, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.