1. What is the projected Compound Annual Growth Rate (CAGR) of the Generic Nifedipine Controlled-release Tablets?
The projected CAGR is approximately 8.4%.
 Generic Nifedipine Controlled-release Tablets
Generic Nifedipine Controlled-release TabletsGeneric Nifedipine Controlled-release Tablets by Type (30mg per Tablet, 60mg per Tablet, World Generic Nifedipine Controlled-release Tablets Production ), by Application (Hypertension, Exertional Angina, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Generic Nifedipine Controlled-release Tablets market is poised for substantial growth, projected to reach approximately $1282.1 million by 2025 and expand at a Compound Annual Growth Rate (CAGR) of 8.4% through 2033. This robust expansion is primarily fueled by the increasing prevalence of cardiovascular diseases like hypertension and exertional angina worldwide. The aging global population, coupled with lifestyle factors such as sedentary habits and unhealthy diets, contributes significantly to the rising incidence of these conditions, thereby driving demand for effective and accessible treatments like generic nifedipine. Furthermore, the growing awareness and adoption of generic medications, owing to their cost-effectiveness compared to branded alternatives, are playing a crucial role in market penetration. The market's trajectory is further bolstered by ongoing research and development in pharmaceutical formulations aimed at improving drug delivery and patient compliance, ensuring sustained market momentum.
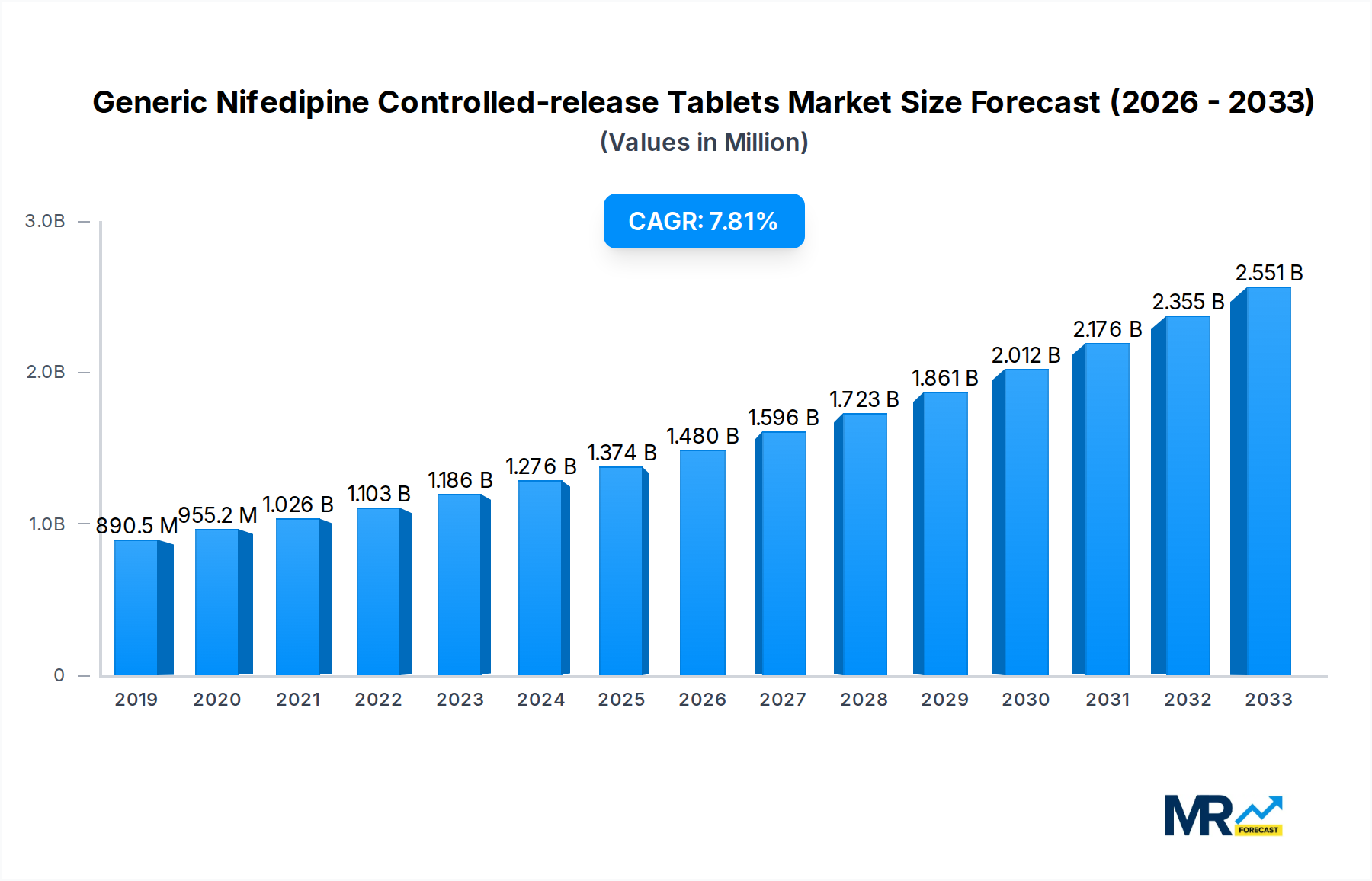

Key market drivers include the escalating burden of hypertension and angina, coupled with supportive government initiatives promoting the use of generic drugs and increasing healthcare expenditure in emerging economies. The market is segmented by tablet strength, with 30mg and 60mg per tablet being prominent, and by application, primarily hypertension and exertional angina. The production landscape is characterized by a competitive environment with established players like Shanghai Shyndec Pharma, Simcere, and CR DOUBLE-CRANE, alongside a host of regional manufacturers. Regionally, the Asia Pacific, led by China and India, is anticipated to exhibit the fastest growth due to a large patient pool, increasing disposable incomes, and improving healthcare infrastructure. North America and Europe represent mature markets with a steady demand driven by an aging population and a well-established healthcare system, while emerging markets in South America and the Middle East & Africa present significant untapped potential for future growth.

Here's a comprehensive report description for Generic Nifedipine Controlled-release Tablets, incorporating all your specified elements:
The global market for Generic Nifedipine Controlled-release Tablets is poised for a significant upswing, driven by a confluence of factors that underscore its enduring therapeutic importance and increasing accessibility. Our in-depth analysis, spanning the Study Period: 2019-2033, with a Base Year: 2025 and Estimated Year: 2025, reveals a dynamic landscape shaped by both established demand and emerging opportunities. The Historical Period: 2019-2024 laid the groundwork, demonstrating consistent growth in consumption, while the Forecast Period: 2025-2033 projects a more accelerated trajectory. Key market insights point towards a growing awareness among healthcare providers and patients regarding the cost-effectiveness and comparable efficacy of generic alternatives to branded Nifedipine formulations. The prevalence of cardiovascular diseases, particularly hypertension, continues to be a primary determinant of market demand, with aging global populations contributing to a sustained need for reliable antihypertensive therapies. Furthermore, advancements in controlled-release technologies have enhanced patient compliance and therapeutic outcomes, making these tablets a preferred choice for long-term management of conditions like hypertension and exertional angina. The market is also witnessing increased penetration in emerging economies, as healthcare infrastructure expands and access to essential medications improves. The World Generic Nifedipine Controlled-release Tablets Production is expected to witness substantial expansion, with manufacturers strategizing to meet burgeoning global demand. The dual-segmentation of 30mg per Tablet and 60mg per Tablet offerings caters to a broad spectrum of patient needs and physician prescribing habits, contributing to the overall market resilience. Innovations in manufacturing processes and a competitive pricing environment are further bolstering the market's upward trend, promising a robust growth phase in the coming years. The strategic focus on managing chronic conditions like hypertension, which affects millions globally, ensures a consistent and expanding market for these vital medications. The increasing emphasis on value-based healthcare also favors the adoption of cost-effective generics, further amplifying their market share.
The propulsion of the Generic Nifedipine Controlled-release Tablets market is intricately linked to a set of powerful drivers that are reshaping the pharmaceutical landscape. Foremost among these is the escalating global burden of cardiovascular diseases, with hypertension remaining a pervasive and growing public health concern. Millions of individuals worldwide require consistent and affordable treatment to manage their blood pressure, making Nifedipine-controlled release tablets a cornerstone therapy. The increasing preference for generic medications due to their cost-effectiveness and bioequivalence to branded counterparts is a significant propellant. As healthcare systems globally grapple with rising costs, physicians and patients alike are actively seeking out more economical yet therapeutically sound treatment options. This economic imperative, coupled with the established safety and efficacy profile of Nifedipine, creates a fertile ground for generic market expansion. Moreover, the aging global population is a crucial demographic driver, as older individuals are more susceptible to chronic conditions like hypertension and angina, thereby increasing the demand for long-term medication management. The advancements in pharmaceutical technology, particularly in controlled-release formulations, have significantly improved patient adherence and therapeutic outcomes. These sophisticated drug delivery systems ensure a steady release of the active ingredient, minimizing fluctuations in plasma concentration and reducing the frequency of dosing, which directly translates to better patient compliance and improved treatment efficacy.
Despite the robust growth prospects, the Generic Nifedipine Controlled-release Tablets market is not without its set of challenges and restraints that warrant careful consideration. A primary concern is the intense competition within the generic drug manufacturing sector. With numerous players vying for market share, price erosion can become a significant factor, impacting profit margins for manufacturers. This highly competitive environment often necessitates substantial investments in marketing and distribution to maintain a strong market presence. Another restraint arises from stringent regulatory hurdles and evolving compliance requirements across different geographies. Obtaining and maintaining regulatory approvals for generic drugs can be a complex and time-consuming process, requiring rigorous testing and documentation, which can delay market entry and increase operational costs. Furthermore, reimbursement policies and formulary restrictions imposed by healthcare payers can influence the prescribing patterns of physicians and the accessibility of these generic tablets to patients. In some instances, preferred drug lists might prioritize certain formulations or brands, indirectly affecting the market penetration of specific generic Nifedipine controlled-release tablets. The potential for counterfeit or substandard products entering the market is another critical issue that can erode patient trust and pose significant health risks. Ensuring the integrity of the supply chain and robust quality control measures are paramount to mitigate this threat. Finally, evolving clinical guidelines and the emergence of newer therapeutic classes for cardiovascular disease management could potentially introduce alternative treatment options, thereby posing a competitive challenge to established generic Nifedipine formulations over the long term.
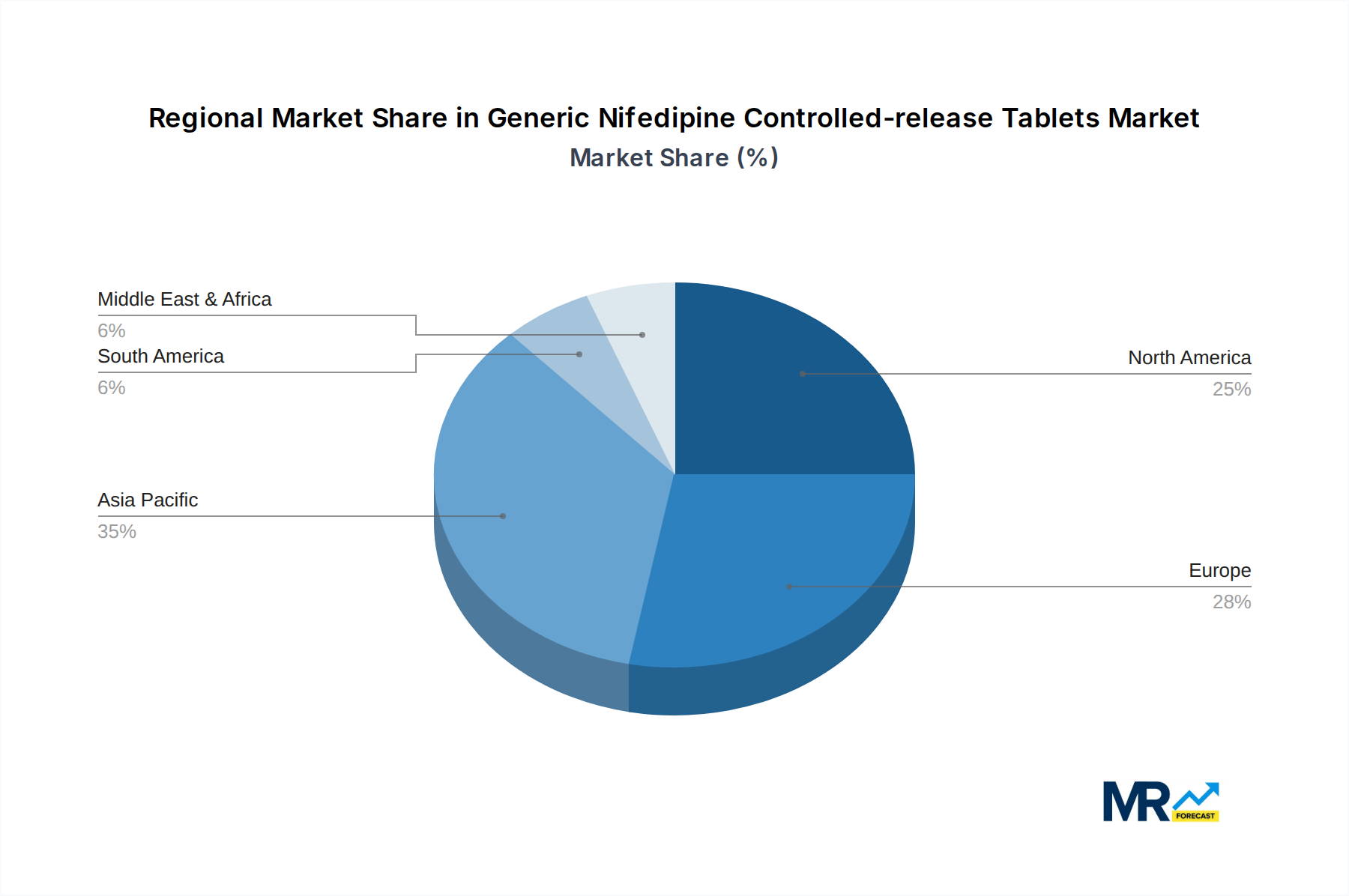
The global market for Generic Nifedipine Controlled-release Tablets is characterized by dynamic regional dominance and segment-specific growth. Examining the market through the lens of Application: Hypertension reveals its commanding position. Hypertension, a chronic condition affecting an estimated 1.28 billion adults globally, forms the bedrock of demand for Nifedipine controlled-release tablets. The consistent need for effective, affordable, and accessible treatments for this pervasive ailment ensures that the hypertension segment will continue to be the largest and most influential within the market. The 30mg per Tablet and 60mg per Tablet segments are both critical, with the 30mg dosage catering to initial titration and milder cases, while the 60mg dosage addresses more severe hypertension or when higher therapeutic levels are required. The choice between these two strengths often depends on individual patient needs, physician preference, and formulary guidelines.
Geographically, the Asia Pacific region is projected to emerge as a dominant force in the Generic Nifedipine Controlled-release Tablets market. This dominance is fueled by several interconnected factors:
While Hypertension is the primary application, Exertional Angina also represents a significant therapeutic area, contributing to sustained demand. Patients suffering from chronic stable angina often rely on Nifedipine for symptom relief and improved quality of life. The ability of controlled-release formulations to provide consistent anti-anginal effects makes them valuable in this segment.
In summary, the Application: Hypertension segment, driven by its widespread prevalence and the inherent need for long-term management, will undeniably lead the market. Geographically, the Asia Pacific region, with its massive patient base, expanding healthcare access, and strong manufacturing capabilities, is set to dominate the global Generic Nifedipine Controlled-release Tablets market in the coming years, with contributions from companies like Shanghai Shyndec Pharma, Simcere, and Qingdao Baiyang Pharmaceutical being particularly noteworthy.
Several key growth catalysts are actively propelling the Generic Nifedipine Controlled-release Tablets industry forward. The relentless rise in the global prevalence of hypertension, a primary indication for Nifedipine, continues to be a fundamental driver. Simultaneously, the increasing global awareness and acceptance of generic pharmaceuticals, attributed to their proven efficacy and significant cost savings compared to branded alternatives, are expanding market reach. Furthermore, strategic investments in research and development by manufacturers to enhance controlled-release technologies are leading to improved drug delivery profiles, better patient compliance, and ultimately, superior therapeutic outcomes, acting as significant catalysts for sustained growth.
This comprehensive report offers an in-depth analysis of the global Generic Nifedipine Controlled-release Tablets market, meticulously covering the Study Period: 2019-2033, with a Base Year: 2025 and a detailed Forecast Period: 2025-2033. It delves into the critical aspects influencing market dynamics, from the escalating prevalence of hypertension and exertional angina to the growing preference for cost-effective generic alternatives. The report meticulously examines the impact of various dosage strengths, including 30mg per Tablet and 60mg per Tablet, on market penetration and consumer choice. Furthermore, it provides a granular overview of World Generic Nifedipine Controlled-release Tablets Production capabilities, identifying key manufacturing hubs and their strategic importance. The report also highlights significant industry developments and the competitive landscape, featuring insights into the strategic moves of leading players like Shanghai Shyndec Pharma, Simcere, and Qingdao Baiyang Pharmaceutical. This holistic approach ensures a thorough understanding of the market's past performance, current state, and future trajectory.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.4% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 8.4%.
Key companies in the market include Shanghai Shyndec Pharma, Simcere, Qingdao Baiyang Pharmaceutical, CR DOUBLE-CRANE, Hefei Cube Pharma, Deyao Pharma, Apichope, Nanjing Easeheal Pharma, Beijing Honglin Pharma.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Generic Nifedipine Controlled-release Tablets," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Generic Nifedipine Controlled-release Tablets, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.