1. What is the projected Compound Annual Growth Rate (CAGR) of the Feline Panleukopenia Virus Antigen Test Kit?
The projected CAGR is approximately 7.44%.
 Feline Panleukopenia Virus Antigen Test Kit
Feline Panleukopenia Virus Antigen Test KitFeline Panleukopenia Virus Antigen Test Kit by Type (1 Box of 10 Strips, 1 Box of 40 Strips, Others), by Application (Pet Hospital, Pet Clinic, Domestic, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Feline Panleukopenia Virus (FPV) Antigen Test Kit market is poised for significant expansion, projected to reach an estimated market size of $185 million by 2025, with a robust Compound Annual Growth Rate (CAGR) of 9.5% anticipated through 2033. This growth is primarily fueled by the increasing global pet population and a heightened awareness among pet owners and veterinary professionals regarding the severity and contagiousness of Feline Panleukopenia. The virus, often referred to as feline distemper, can be highly fatal, especially in kittens, driving the demand for rapid and accurate diagnostic tools. Technological advancements in lateral flow immunoassay technology have led to the development of more sensitive and specific FPV antigen test kits, offering quicker results compared to traditional laboratory methods. This speed is crucial in veterinary settings for timely diagnosis and initiation of treatment, thereby improving patient outcomes and reducing disease transmission within animal populations. The increasing prevalence of veterinary hospitals and clinics, coupled with a growing trend of pet humanization, where pets are increasingly treated as family members, further bolsters market growth.
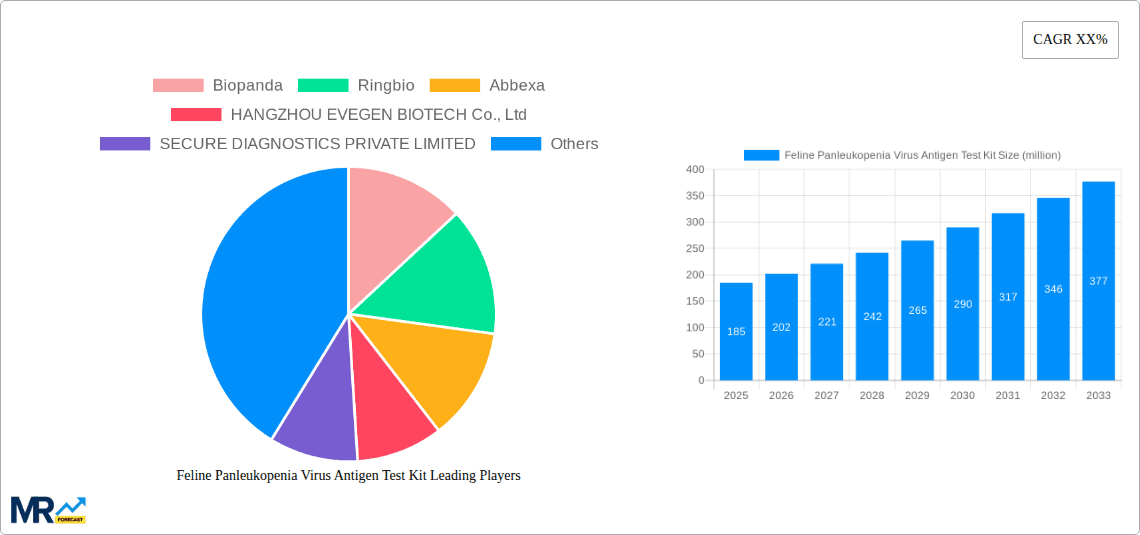

The market segmentation reveals a strong demand for convenient and user-friendly test formats, with "1 Box of 10 Strips" and "1 Box of 40 Strips" likely to dominate sales due to their suitability for both high-volume clinics and individual pet owners. Pet hospitals and clinics represent the largest application segments, owing to their consistent need for diagnostic tools. However, the "Domestic" application segment is expected to witness substantial growth as pet owners seek at-home testing solutions for early detection and peace of mind. Geographically, the Asia Pacific region, particularly China and India, is emerging as a key growth driver due to a rapidly expanding pet ownership base and increasing disposable incomes. North America and Europe continue to be mature markets with a well-established demand for veterinary diagnostics. Key players like Biopanda, Ringbio, and Abbexa are actively investing in research and development to enhance product accuracy, reduce testing time, and develop more cost-effective solutions, all contributing to the overall positive market trajectory. Despite the optimistic outlook, the primary restraint remains the relatively high cost of some advanced test kits, which may limit adoption in budget-conscious regions or among owners with fewer financial resources.
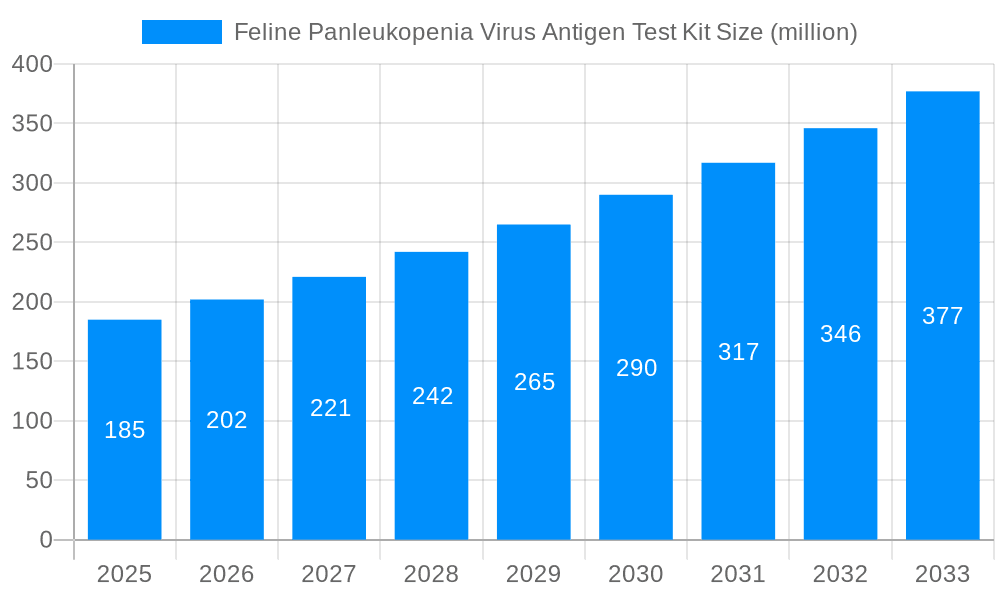

The global Feline Panleukopenia Virus (FPV) Antigen Test Kit market is poised for significant expansion, driven by an increasing awareness of feline health and the urgent need for rapid, on-site diagnostics. The study period of 2019-2033 encapsulates a dynamic evolution, with the base year of 2025 serving as a pivotal point for forecasting. The historical period of 2019-2024 has witnessed steady growth, fueled by a rising pet population and a greater emphasis on preventative care for domestic animals. Looking ahead, the forecast period of 2025-2033 is expected to see an accelerated trajectory, with market valuations reaching hundreds of millions of US dollars. This upward trend is intrinsically linked to the escalating prevalence of FPV infections, particularly in unvaccinated or immunocompromised feline populations, necessitating swift and accurate diagnostic solutions. The market is characterized by a growing demand for kits offering high sensitivity and specificity, minimizing false positives and negatives, thereby enabling timely and effective treatment. Furthermore, advancements in lateral flow immunoassay technology have paved the way for user-friendly, cost-effective, and rapid testing capabilities, making these kits indispensable tools in veterinary settings. The increasing adoption of these kits in both advanced veterinary facilities and developing regions underscores their growing importance in safeguarding feline health globally. The market’s future growth will also be influenced by evolving regulatory landscapes and the continuous drive for innovation in diagnostic technologies, aiming for even faster turnaround times and more integrated diagnostic platforms. The market is projected to surpass 250 million USD by the end of the forecast period, a substantial leap from its historical performance.
Key market insights:
The robust growth of the Feline Panleukopenia Virus Antigen Test Kit market is underpinned by a confluence of powerful driving forces, each contributing to its upward trajectory. Paramount among these is the escalating global pet population. The increasing humanization of pets has led to owners investing more in their companions' well-being, translating into a greater demand for advanced veterinary care, including sophisticated diagnostic tools. This trend is particularly pronounced in developed nations but is steadily gaining momentum in emerging economies as well. Secondly, the inherent virulence and significant mortality rate associated with FPV itself serve as a potent driver. The rapid onset and devastating impact of this disease necessitate immediate diagnostic confirmation, making quick and reliable test kits indispensable for veterinarians aiming to initiate timely treatment and improve patient outcomes. Furthermore, heightened awareness among pet owners and veterinary professionals regarding feline health and infectious diseases plays a crucial role. Educational campaigns and increased access to veterinary information empower owners to seek prompt care, thereby boosting the demand for diagnostic tests. The development and widespread adoption of point-of-care diagnostics have also been instrumental. These test kits, offering rapid results at the veterinarian's office or even at home, eliminate the delays associated with laboratory testing, facilitating immediate decision-making. This convenience and speed are highly valued in time-sensitive medical situations. Finally, continuous research and development efforts by diagnostic companies are introducing more sensitive, specific, and user-friendly test kits, further stimulating market expansion by offering superior diagnostic capabilities and addressing unmet needs. The market is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 7.5% during the forecast period.
Despite the promising growth trajectory, the Feline Panleukopenia Virus Antigen Test Kit market is not without its inherent challenges and restraints that could potentially impede its full realization. One of the primary hurdles is the cost of these diagnostic kits, particularly for widespread implementation in resource-limited regions or for individuals with limited disposable income. While prices have decreased with technological advancements, affordability remains a significant concern for broader market penetration. Secondly, the issue of false positives and false negatives, though diminishing with improved technologies, continues to pose a challenge. Inaccurate test results can lead to delayed or incorrect treatment, impacting animal welfare and eroding trust in diagnostic tools. This necessitates rigorous quality control and validation processes for all kits entering the market. Another significant restraint is the limited access to veterinary services and diagnostics in certain developing countries. Infrastructure limitations, a shortage of trained veterinary personnel, and a lack of awareness about the importance of FPV testing can hinder market growth in these areas. Furthermore, the availability of alternative diagnostic methods, such as PCR testing, while more accurate in some instances, can be more expensive and time-consuming, creating a complex competitive landscape. The stringent regulatory approval processes for veterinary diagnostic products in different regions can also act as a bottleneck, delaying the introduction of new and improved test kits. Finally, fluctuations in raw material costs and supply chain disruptions can impact the production and availability of test kits, potentially leading to price volatility and supply shortages. The market's growth could be limited by 5-10% due to these factors.
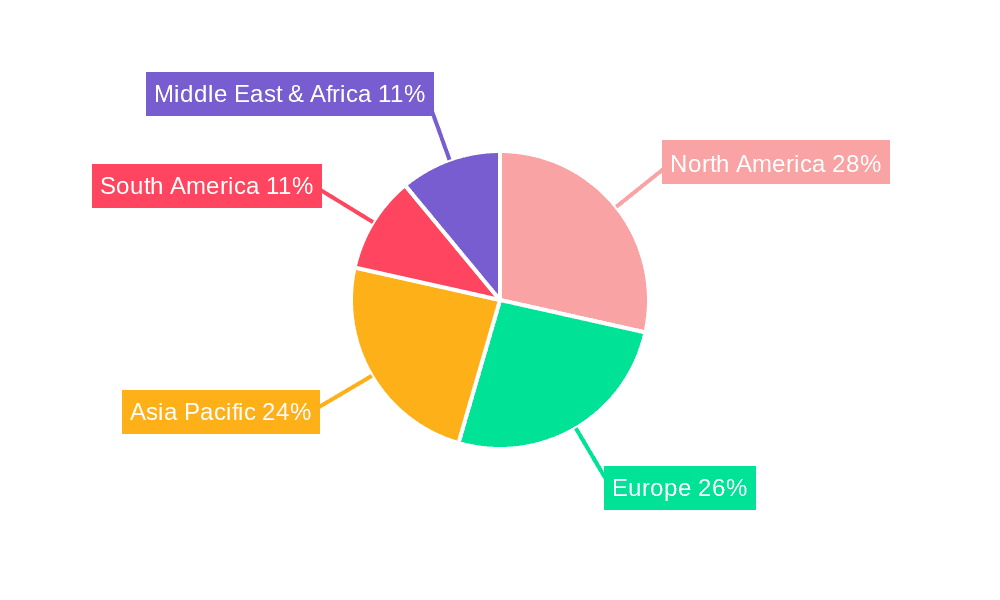
The global Feline Panleukopenia Virus Antigen Test Kit market is anticipated to witness significant dominance by both specific regions and certain market segments during the study period of 2019-2033, with the forecast period of 2025-2033 showing a continued trend.
Key Dominating Regions/Countries:
Key Dominating Segments:
The synergy between these dominating regions and segments will shape the market landscape, with North America and Europe leading in overall adoption, while the Asia-Pacific region presents the most significant growth potential. The 40-strip packaging and pet hospital application will represent the core demand drivers, reflecting the practical needs and economic considerations of veterinary practices worldwide. The market for FPV Antigen Test Kits is projected to reach over 250 million USD by 2033, with North America and Europe accounting for approximately 60% of this valuation.
Several key factors are accelerating the growth of the Feline Panleukopenia Virus Antigen Test Kit industry. The rising global pet population, coupled with increasing pet humanization, is a primary catalyst, leading to greater investment in feline healthcare. Furthermore, the inherent severity and rapid progression of FPV infections necessitate prompt diagnosis, thereby fueling the demand for quick and reliable on-site testing solutions. Advancements in diagnostic technologies, leading to improved sensitivity, specificity, and user-friendliness of these kits, are also significant growth drivers. Finally, increasing awareness among veterinarians and pet owners about the importance of early disease detection and preventative care is contributing to broader adoption and market expansion.
This comprehensive report delves into the intricate dynamics of the Feline Panleukopenia Virus Antigen Test Kit market, offering a forward-looking analysis from 2019 to 2033. It meticulously examines market trends, identifies the core driving forces behind growth, and scrutinizes the challenges that may impede expansion. The report provides an in-depth understanding of the key regions and market segments poised for dominance, offering valuable insights for strategic decision-making. Furthermore, it highlights the crucial growth catalysts shaping the industry's future and profiles the leading companies actively contributing to market innovation. With a detailed look at significant developments and a robust forecast, this report equips stakeholders with the necessary intelligence to navigate and capitalize on the evolving FPV Antigen Test Kit landscape. The market is anticipated to witness a significant valuation by 2025, projected to exceed 180 million USD.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.44% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7.44%.
Key companies in the market include Biopanda, Ringbio, Abbexa, HANGZHOU EVEGEN BIOTECH Co., Ltd, SECURE DIAGNOSTICS PRIVATE LIMITED, Nexgen Innovations, BioSupply Ltd., BIONOTE, J&G Biotech Ltd, Hubei Jinjian Biology, Ubio Biotechnology Systems Pvt Ltd, Creative Diagnostics, Ank Cares Private Ltd, Elabscience, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Feline Panleukopenia Virus Antigen Test Kit," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Feline Panleukopenia Virus Antigen Test Kit, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.