1. What is the projected Compound Annual Growth Rate (CAGR) of the Energy Based Ablation Devices?
The projected CAGR is approximately XX%.
 Energy Based Ablation Devices
Energy Based Ablation DevicesEnergy Based Ablation Devices by Type (Electrical, Light, Radiation, Radiofrequency, Ultrasound, Other), by Application (Ophthalmic Surgery, General Surgery, Cancer Therapy, Cardiovascular Disease, Other), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
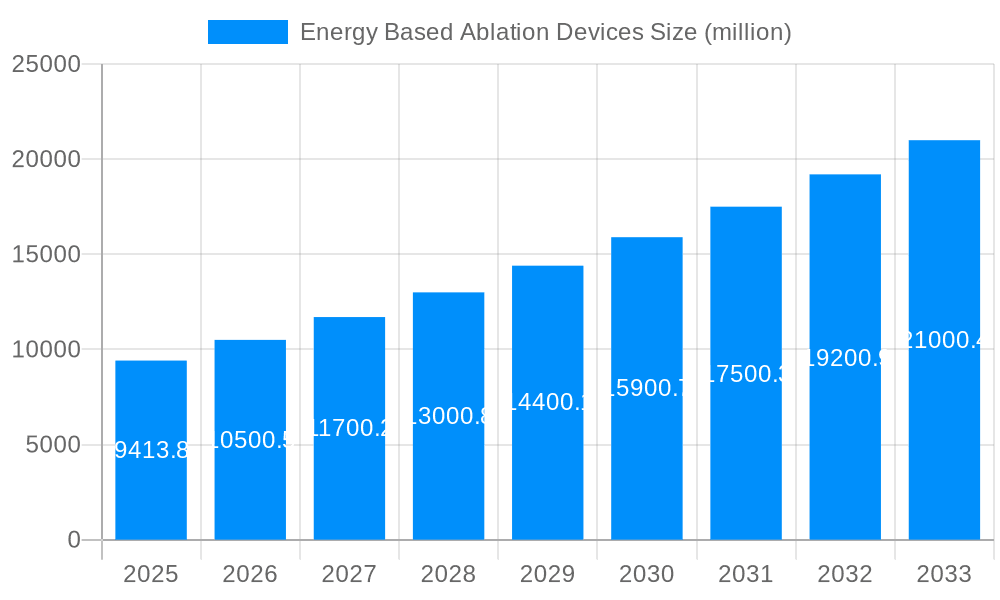
The global Energy Based Ablation Devices market is poised for significant expansion, projected to reach a substantial valuation of $9413.8 million by 2025. This growth is fueled by a confluence of technological advancements and an increasing demand for minimally invasive surgical procedures across a broad spectrum of medical applications. Notably, the market is driven by the rising prevalence of chronic diseases such as cardiovascular conditions and cancer, where energy-based ablation offers a more targeted and less invasive treatment alternative to traditional surgery. Furthermore, the burgeoning elderly population worldwide, susceptible to age-related health issues, further amplifies the demand for these advanced medical devices. The continuous innovation in ablation technologies, including advancements in radiofrequency, laser, and ultrasound-based devices, is expanding their therapeutic scope and improving patient outcomes, thereby propelling market growth.
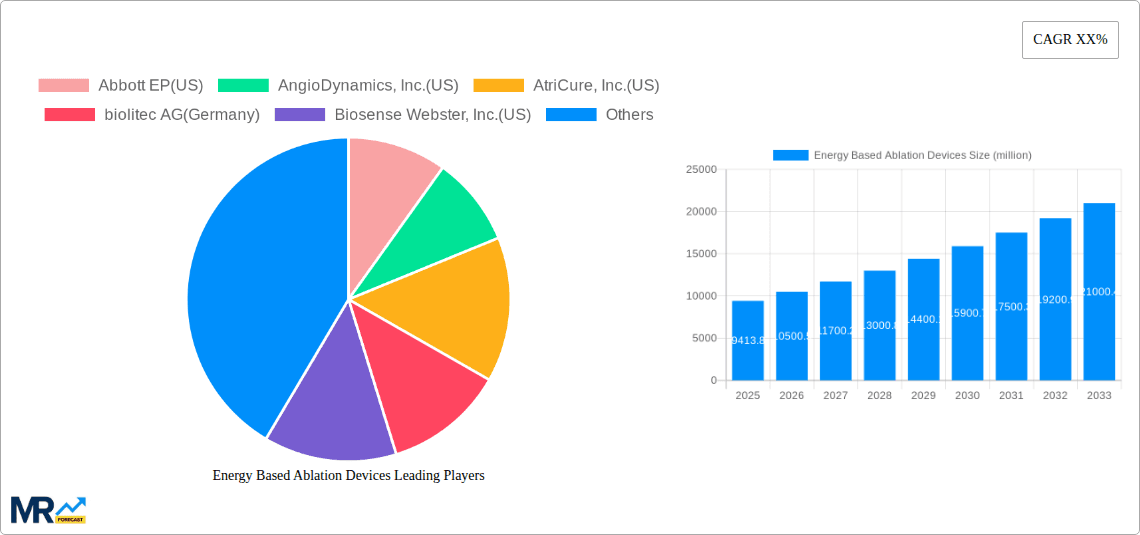

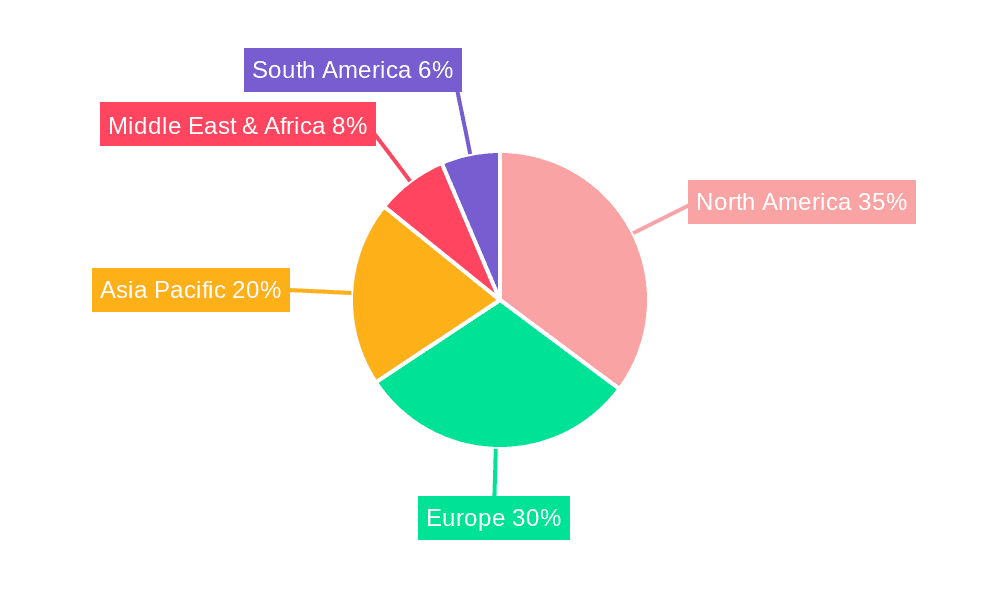
The market segmentation reveals a dynamic landscape. In terms of type, Electrical and Radiofrequency ablation devices are anticipated to dominate owing to their established efficacy and widespread adoption in critical applications like cardiovascular disease treatment and general surgery. The Application segment highlights the significant contributions of Ophthalmic Surgery and Cancer Therapy, where energy-based ablation is revolutionizing treatment protocols. Regions like North America and Europe are expected to lead the market share, driven by high healthcare expenditure, advanced healthcare infrastructure, and a strong presence of key market players. However, the Asia Pacific region is exhibiting rapid growth, attributed to increasing healthcare investments, a growing patient pool, and the expanding access to sophisticated medical technologies. While the market demonstrates robust growth, potential restraints such as high device costs and the need for specialized training for healthcare professionals warrant strategic consideration by market participants.

Here's a report description on Energy Based Ablation Devices, structured as requested:
The global Energy Based Ablation Devices market is poised for significant expansion, driven by an escalating prevalence of chronic diseases and the continuous pursuit of minimally invasive surgical techniques. With a projected market valuation of $15,250 million in 2025, this sector is set to witness robust growth throughout the forecast period of 2025-2033. The historical period of 2019-2024 laid the groundwork for this ascent, characterized by increasing adoption rates and technological refinements. Key trends shaping the market include the surging demand for advanced ablation technologies in cardiovascular interventions, particularly for treating arrhythmias like atrial fibrillation, where radiofrequency and cryoablation devices are gaining considerable traction. Furthermore, the oncological landscape is witnessing a paradigm shift with the increasing integration of image-guided ablation therapies for various solid tumors, offering targeted destruction of cancerous cells with reduced damage to surrounding healthy tissues. The ophthalmic surgery segment is also a significant contributor, with laser-based ablation technologies playing a crucial role in refractive error correction and the treatment of various ocular pathologies. The market's trajectory is also influenced by the growing emphasis on patient recovery times and reduced hospital stays, directly aligning with the benefits offered by minimally invasive ablation procedures. Innovations in energy delivery mechanisms, improved imaging integration, and the development of more sophisticated navigation systems are continuously enhancing the efficacy and safety of these devices, further fueling market penetration. The estimated market size for 2025 stands as a testament to the pivotal role these devices play in modern healthcare.
The propulsion of the Energy Based Ablation Devices market is largely attributed to a confluence of critical factors. Foremost among these is the rapidly aging global population, which inherently leads to a higher incidence of age-related conditions such as cardiovascular diseases, cancers, and degenerative eye conditions, all of which benefit from ablation therapies. Concurrently, the persistent and growing burden of chronic diseases, including atrial fibrillation, liver cancer, prostate cancer, and uterine fibroids, necessitates effective and less invasive treatment modalities. Energy-based ablation devices offer a compelling solution by providing targeted tissue destruction with minimal collateral damage compared to traditional open surgical procedures. The relentless innovation in medical technology, particularly in the realm of energy delivery systems and imaging guidance, has significantly improved the precision, safety, and efficacy of ablation procedures. Advancements in radiofrequency, laser, microwave, and ultrasound technologies, coupled with the integration of sophisticated navigation and visualization tools, are enabling clinicians to perform more complex ablations with greater confidence. Furthermore, the increasing healthcare expenditure across developed and developing economies, coupled with favorable reimbursement policies for minimally invasive procedures, directly contributes to the wider adoption and accessibility of these devices, thereby propelling market growth.
Despite the promising outlook, the Energy Based Ablation Devices market encounters several significant challenges and restraints that could temper its growth trajectory. A primary hurdle is the high cost associated with advanced ablation devices and associated technologies. This can be particularly prohibitive for healthcare facilities in low- and middle-income countries, limiting their widespread adoption. The need for specialized training and expertise for healthcare professionals to operate these sophisticated devices effectively also presents a bottleneck, requiring substantial investment in education and skill development. Moreover, while ablation therapies are generally considered safe, there remains a risk of complications and side effects associated with any invasive procedure, which can deter some patients and clinicians. The regulatory landscape, characterized by stringent approval processes for new medical devices, can also slow down market entry for innovative products, leading to extended development timelines and increased costs for manufacturers. Furthermore, reimbursement policies for ablation procedures, while improving, can still be inconsistent across different healthcare systems and geographical regions, potentially impacting the economic viability of adopting these technologies for certain indications. Finally, the growing competition from alternative treatment modalities, including pharmaceutical interventions and less invasive non-ablation surgical techniques, presents a continuous challenge for market dominance.
The Cardiovascular Disease segment, particularly in terms of applications, is anticipated to be a dominant force in the Energy Based Ablation Devices market, with its influence extending across key regions. The increasing global burden of arrhythmias, such as atrial fibrillation, is a primary driver for this dominance. Radiofrequency ablation (RFA) and cryoablation remain the cornerstone treatments for these conditions, offering a minimally invasive alternative to long-term pharmacological management. The United States is expected to lead in this segment, driven by a combination of factors:
Another highly influential segment set to contribute significantly to market dominance is Cancer Therapy, particularly in the application of ablating solid tumors. The development and refinement of image-guided ablation techniques, such as microwave ablation, laser ablation, and focused ultrasound, are revolutionizing cancer treatment. These techniques offer targeted destruction of cancerous cells with reduced invasiveness and improved patient outcomes compared to traditional surgical resection or systemic therapies.
Within the Type segment, Radiofrequency ablation devices are expected to continue their reign as a dominant technology. Their established efficacy, versatility across various applications (cardiovascular, oncology, gynecology), and ongoing technological advancements, including pulsed RFA and multipolar RFA, solidify their market leadership. The integration of advanced mapping systems further enhances their precision and safety profile.
The Energy Based Ablation Devices industry is propelled by potent growth catalysts. The escalating prevalence of chronic diseases like cardiovascular disorders and cancer directly fuels the demand for effective, minimally invasive treatments. Continuous innovation in ablation technologies, such as enhanced energy delivery mechanisms and improved imaging guidance, leads to safer and more precise procedures. The growing preference for minimally invasive surgeries, offering reduced recovery times and hospital stays, further bolsters adoption. Government initiatives supporting advanced healthcare technologies and favorable reimbursement policies also contribute significantly to market expansion.
This comprehensive report offers an in-depth analysis of the Energy Based Ablation Devices market, providing critical insights for stakeholders. It meticulously details market dynamics, including historical trends from 2019-2024, the current landscape in the Base Year 2025, and robust projections for the Forecast Period 2025-2033. The report delves into key growth catalysts, such as the rising prevalence of chronic diseases and technological advancements, alongside a thorough examination of challenges like high costs and regulatory hurdles. It highlights dominant market segments and regions, offering strategic guidance for market players. The comprehensive coverage ensures a clear understanding of the market's potential and the strategies required for success.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Abbott EP(US), AngioDynamics, Inc.(US), AtriCure, Inc.(US), biolitec AG(Germany), Biosense Webster, Inc.(US), Boston Scientific Corporation(US), BTG Plc(UK), Cardiogenesis Corporation(US), Cynosure, Inc.(US), Conmed Corporation(US), EDAP TMS S.A.(France), Ethicon Endo-Surgery, Inc.(US), Halyard Health, Inc.(US), InSightec® Ltd., Integra LifeSciences Corporation(US), Lumenis Ltd., Medtronic, Plc, Monteris Medical, Inc.(US), Olympus America, Inc.(US), Sichuan Jinjiang Electronic Science and Technology Co., Ltd.(China), SonaCare Medical LLC(US), Smith & Nephew Plc(UK), Stryker Corporation(US), Syneron & Candela(US), Varian Medical Systems, Inc.(US).
The market segments include Type, Application.
The market size is estimated to be USD 9413.8 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Energy Based Ablation Devices," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Energy Based Ablation Devices, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.