1. What is the projected Compound Annual Growth Rate (CAGR) of the Endoscope Transport System?
The projected CAGR is approximately 7.6%.
 Endoscope Transport System
Endoscope Transport SystemEndoscope Transport System by Type (4 Floors, 6 Floors, 8 Floors, Others), by Application (Hospital, Clinic), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Endoscope Transport System market is projected for substantial growth, estimated to reach USD 37.08 billion by 2025, with a Compound Annual Growth Rate (CAGR) of 7.6% anticipated from 2025 to 2033. This expansion is driven by increased demand for advanced reprocessing solutions in healthcare, fueled by the rising incidence of gastrointestinal and respiratory diseases. Growing emphasis on infection control and patient safety necessitates efficient endoscope transport systems to prevent cross-contamination. Technological advancements in automated and integrated systems, improving workflow efficiency and reducing manual handling, are key growth factors. The "4 Floors" and "6 Floors" segments are expected to lead, serving diverse healthcare facility needs.

Market growth is further supported by the rising number of endoscopic procedures worldwide, increasing the demand for safe transport and storage of medical devices. Key industry players are investing in research and development for innovative solutions. However, high initial investment costs and stringent regulatory compliance present challenges. Despite these, escalating awareness of healthcare-associated infections and healthcare providers' efforts to upgrade infrastructure to meet international standards are expected to sustain demand across major regions, with North America and Europe leading adoption.
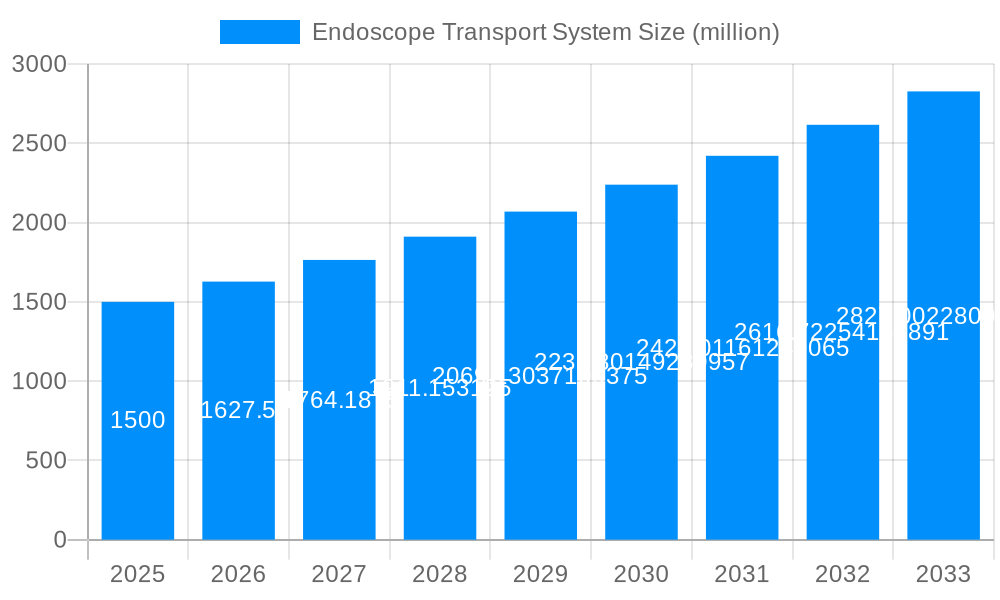

This report provides an in-depth analysis of the global Endoscope Transport System market from 2019-2024, with projections through 2033. The base year of 2025 is a key reference. The market dynamics are shaped by technological innovation, evolving regulations, and the demand for efficient endoscope reprocessing. With an estimated market size of hundreds of millions of USD in 2025, the market is set for significant expansion, driven by heightened infection control awareness and the need for streamlined healthcare workflows.
The Endoscope Transport System market is witnessing a profound transformation, moving beyond basic containment solutions to sophisticated integrated systems that prioritize patient safety, operational efficiency, and regulatory compliance. XXX reveals a notable trend towards automated and semi-automated transport solutions. These systems are designed to minimize manual handling of soiled endoscopes, thereby reducing the risk of cross-contamination and protecting healthcare personnel from potential exposure to biohazards. The market is also observing a surge in demand for modular and customizable transport carts that can be adapted to the specific spatial constraints and workflow requirements of different healthcare facilities, ranging from small clinics to large-scale hospitals. Furthermore, the integration of smart technologies, such as RFID tagging and real-time tracking, is gaining traction. This enables better inventory management, enhances traceability throughout the reprocessing cycle, and provides valuable data for process optimization. The growing emphasis on infection prevention and control, bolstered by stringent regulatory guidelines worldwide, is a primary driver for the adoption of advanced endoscope transport systems. As the prevalence of healthcare-associated infections (HAIs) remains a significant concern, the investment in robust transport solutions that ensure the integrity and sterility of endoscopes from point-of-use to reprocessing is escalating. The market is also seeing a shift towards eco-friendly materials and energy-efficient designs in the manufacturing of these systems. Companies are increasingly focusing on developing transport solutions that are not only functional but also sustainable, aligning with broader environmental consciousness within the healthcare industry. The evolving nature of endoscopic procedures, with an increasing complexity and diversity of instruments, is also influencing the design and functionality of transport systems, necessitating greater adaptability and specialized compartments. The increasing adoption of single-use endoscopes in certain applications, while potentially impacting the demand for reprocessing infrastructure in the long term, also necessitates efficient and segregated transport for both reusable and disposable scopes. The overall market trajectory points towards a future where endoscope transport is an integral, highly controlled, and technologically advanced component of the entire endoscopic workflow, contributing significantly to patient outcomes and operational excellence in healthcare.
The growth of the Endoscope Transport System market is propelled by several interconnected forces, primarily centered around the paramount importance of infection control and patient safety. The persistent threat of healthcare-associated infections (HAIs), coupled with increasingly stringent regulatory mandates from bodies like the FDA and ISO, compels healthcare facilities to invest in robust systems that minimize the risk of cross-contamination during endoscope handling. This includes the development and implementation of standardized protocols for the transport of soiled endoscopes from the patient procedure area to the decontamination and sterilization units. Furthermore, the growing volume of endoscopic procedures performed globally, driven by an aging population, rising prevalence of chronic diseases, and advancements in minimally invasive surgery, directly translates to a higher demand for efficient and reliable endoscope transport solutions. Hospitals and clinics are seeking to optimize their workflow and reduce turnaround times for endoscopes, leading to an increased adoption of automated and semi-automated transport systems that can streamline the movement of scopes and related accessories. The pursuit of operational efficiency also plays a crucial role. Facilities are looking to minimize manual handling, reduce the labor associated with transporting scopes, and improve the overall productivity of their endoscopy departments. This has spurred innovation in the design of transport carts and systems that are ergonomic, easily maneuverable, and capable of accommodating multiple scopes and their accessories, thereby reducing the number of trips required.
Despite the promising growth trajectory, the Endoscope Transport System market faces several challenges and restraints that can impede its full potential. One significant hurdle is the considerable initial investment required for advanced, automated, and technologically integrated transport systems. For smaller healthcare facilities or those with budget constraints, the upfront cost can be a deterrent, leading them to opt for less sophisticated or manual solutions. This creates a disparity in the adoption of best practices across the industry. Another challenge lies in the diverse range of existing infrastructure within healthcare facilities. Many older hospitals may not have the spatial layout or the necessary infrastructure to seamlessly integrate new, larger, or automated transport systems without significant modifications, which further increases costs and complexity. Furthermore, resistance to change and a lack of comprehensive training among healthcare personnel can also pose a restraint. Adopting new transport protocols and systems requires a cultural shift and consistent training to ensure proper utilization and adherence to safety standards. Without adequate training, the effectiveness of even the most advanced systems can be compromised. The complexity of certain endoscope designs and the accessories associated with them also presents a challenge in designing universal transport solutions. Systems must be versatile enough to accommodate a wide array of scopes, from delicate flexible instruments to rigid scopes and their associated equipment, demanding flexible and adaptable storage solutions. Finally, the ongoing debate and evolving understanding of best practices in decontamination and reprocessing, while driving innovation, can also create a degree of uncertainty for manufacturers and end-users regarding future-proof system designs.
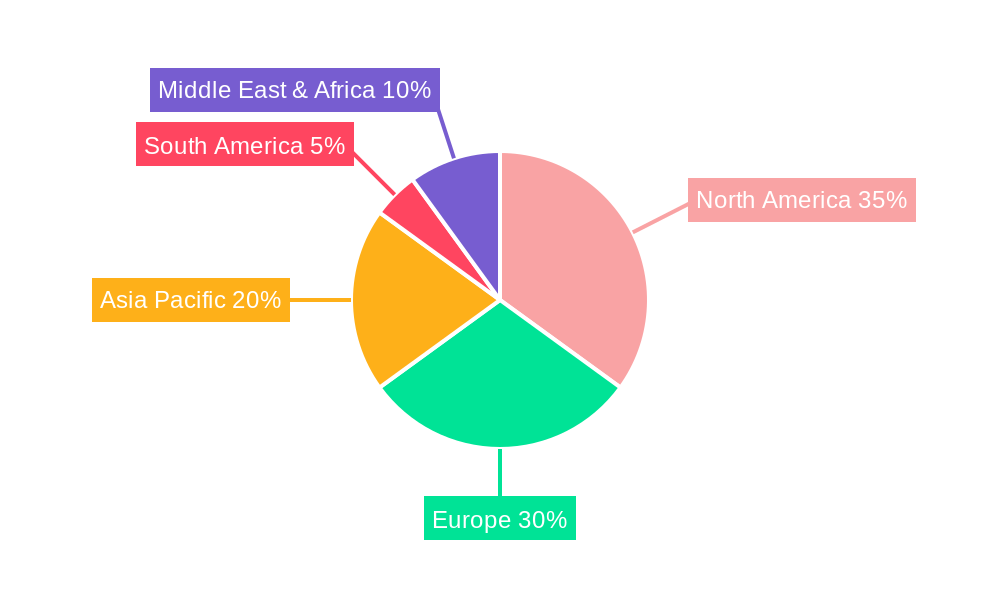
The global Endoscope Transport System market is characterized by distinct regional strengths and segment dominance, with North America poised to be a leading region in terms of market share and growth during the forecast period. This dominance is underpinned by several critical factors.
Robust Healthcare Infrastructure and High Adoption Rates: North America, particularly the United States, possesses a highly developed healthcare infrastructure with a significant number of advanced hospitals, clinics, and specialized endoscopy centers. This advanced infrastructure naturally facilitates a higher adoption rate of sophisticated medical equipment, including endoscope transport systems. The existing network of healthcare providers is well-equipped to invest in and integrate these technologies to enhance patient care and operational efficiency.
Stringent Regulatory Environment and Focus on Patient Safety: The stringent regulatory landscape in North America, driven by organizations like the FDA, places a strong emphasis on infection control and patient safety. This regulatory pressure directly translates into a heightened demand for reliable and compliant endoscope transport solutions that minimize the risk of healthcare-associated infections (HAIs). Facilities are compelled to adopt best practices, making advanced transport systems a necessity rather than a luxury.
High Incidence of Endoscopic Procedures: The region witnesses a substantial volume of endoscopic procedures, driven by factors such as an aging population, the increasing prevalence of gastrointestinal disorders, and the growing adoption of minimally invasive diagnostic and therapeutic techniques. This high procedural volume necessitates efficient and rapid turnaround times for endoscopes, thereby fueling the demand for effective transport systems that streamline the workflow from point-of-use to reprocessing.
Technological Advancements and Innovation: North America is at the forefront of technological innovation in the medical device sector. The presence of leading manufacturers and a strong R&D ecosystem fosters the development of advanced endoscope transport systems, including automated, semi-automated, and smart solutions with features like RFID tracking and real-time monitoring. This constant influx of innovative products attracts early adopters and drives market growth.
Within the broader market, the Hospital segment is expected to dominate in terms of revenue and adoption of endoscope transport systems. This dominance is attributed to several factors:
Volume and Complexity of Procedures: Hospitals, by their nature, handle a significantly higher volume and a wider variety of endoscopic procedures compared to standalone clinics or industrial applications. This includes complex surgeries and diagnostic interventions, which require a constant and reliable supply of sterilized endoscopes.
Dedicated Infection Control Departments and Resources: Hospitals typically have dedicated infection control departments and specialized reprocessing units with the necessary resources and expertise to implement and manage advanced endoscope transport protocols and systems. The financial capacity and organizational structure of hospitals are more conducive to investing in such comprehensive solutions.
Regulatory Compliance and Liability Concerns: Hospitals face greater scrutiny regarding regulatory compliance and are more susceptible to liability claims arising from HAIs. This heightened awareness of risks incentivizes them to adopt the most robust and secure transport systems available to mitigate these concerns.
Integration with Broader Hospital Workflow: Endoscope transport systems in hospitals often need to integrate seamlessly with other hospital logistics and workflow systems. This necessitates more sophisticated and interconnected solutions, driving the demand for feature-rich and adaptable transport systems.
While clinics and other segments are important, the sheer scale of operations, the complexity of procedures, and the stringent requirements for patient safety and regulatory adherence within the hospital setting position it as the primary driver for the Endoscope Transport System market. The 6 Floors type of transport system is also expected to witness significant adoption, particularly in medium to large hospitals, offering a balance between capacity, maneuverability, and efficiency for multi-level facilities.
The Endoscope Transport System industry is propelled by several key growth catalysts. The unrelenting focus on infection prevention and control is paramount, driven by increasing awareness of HAIs and stricter regulatory mandates globally. As healthcare facilities strive to meet these standards, the demand for reliable and safe endoscope transport solutions escalates. Furthermore, the rising volume of endoscopic procedures, fueled by an aging population and advancements in minimally invasive surgery, directly translates to a greater need for efficient scope handling and reprocessing. This necessitates improved workflow management and reduced turnaround times, a critical function of effective transport systems. Technological advancements, such as automation, smart tracking, and modular designs, are making these systems more efficient, user-friendly, and adaptable to diverse healthcare settings. These innovations are not only improving operational efficiency but also enhancing patient safety, thereby driving market adoption.
This report offers an unparalleled, comprehensive overview of the global Endoscope Transport System market, providing deep insights and actionable intelligence for stakeholders. Spanning the study period of 2019-2033, with a crucial base year of 2025, it meticulously analyzes historical trends, current market dynamics, and future projections. The report leverages detailed market segmentation, examining various types (4 Floors, 6 Floors, 8 Floors, Others) and applications (Hospital, Clinic, Industry) to offer granular perspectives. It identifies and scrutinizes the key market drivers, restraints, opportunities, and emerging trends, providing a holistic understanding of the competitive landscape. Furthermore, it profiles leading industry players and highlights significant technological developments and strategic initiatives shaping the market's evolution. This report is an indispensable resource for manufacturers, distributors, healthcare providers, and investors seeking to navigate and capitalize on the opportunities within the dynamic Endoscope Transport System market, estimating its value to be in the hundreds of millions of USD.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 7.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 7.6%.
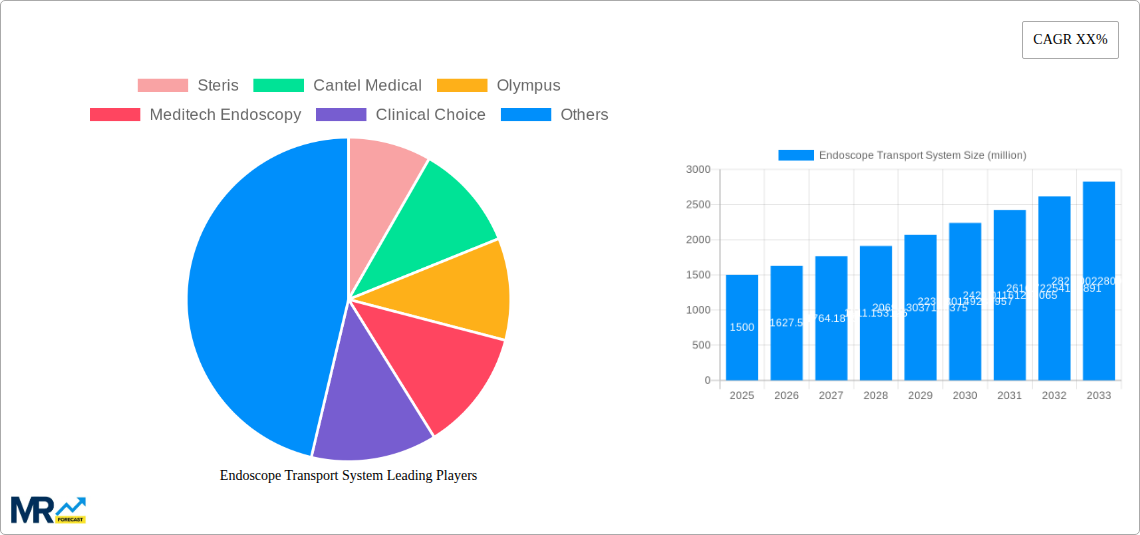
Key companies in the market include Steris, Cantel Medical, Olympus, Meditech Endoscopy, Clinical Choice, MASS Medical Storage, Cygnus Medical, Harloff Manufacturing, Wassenburg Medical, Creo Medical Group, Optim LLC, Ruhof, .
The market segments include Type, Application.
The market size is estimated to be USD 37.08 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion and volume, measured in K.
Yes, the market keyword associated with the report is "Endoscope Transport System," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Endoscope Transport System, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.