1. What is the projected Compound Annual Growth Rate (CAGR) of the Disposable Embolic Protection Device?
The projected CAGR is approximately 8.3%.
 Disposable Embolic Protection Device
Disposable Embolic Protection DeviceDisposable Embolic Protection Device by Application (Cardiovascular Diseases, Neurovascular Diseases, Peripheral Diseases), by Type (Nitinol Embolic Protection Device Material, Polyurethane Embolic Protection Device Material), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Disposable Embolic Protection Device market is poised for significant expansion, projected to reach approximately $5.5 billion by 2033, with a robust Compound Annual Growth Rate (CAGR) of around 8.5% from 2025 to 2033. This growth is primarily fueled by the increasing prevalence of cardiovascular and neurovascular diseases globally, necessitating advanced interventional procedures. The rising adoption of minimally invasive techniques, driven by their lower risk profiles and faster recovery times, directly boosts demand for these devices. Key market drivers include an aging global population, a surge in lifestyle-related diseases like hypertension and diabetes that contribute to vascular conditions, and continuous technological advancements leading to more effective and safer embolic protection solutions. The market's value, estimated at around $2.5 billion in the base year of 2025, reflects a dynamic landscape where innovation and patient outcomes are paramount.
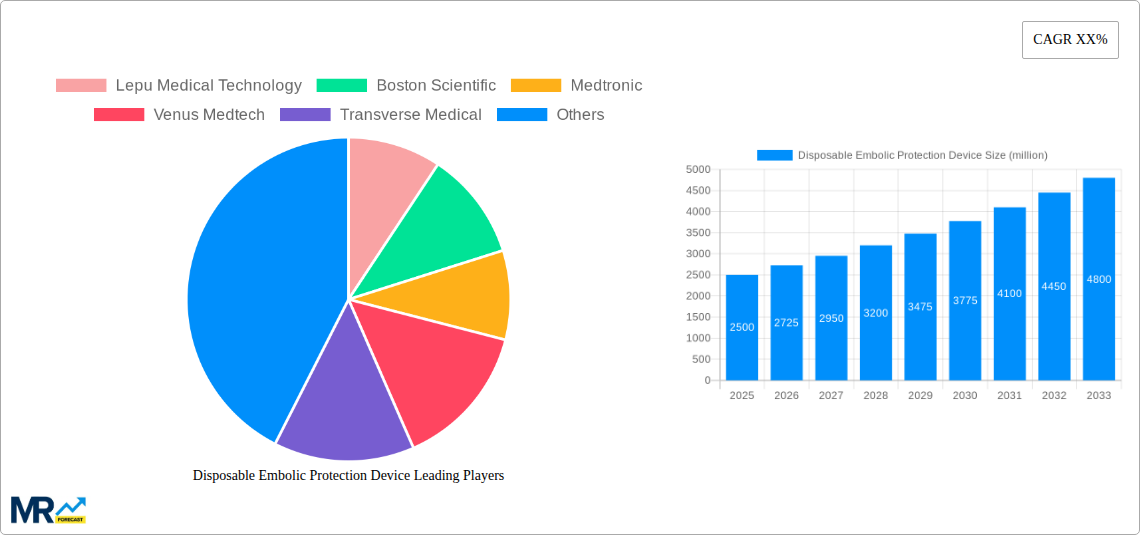

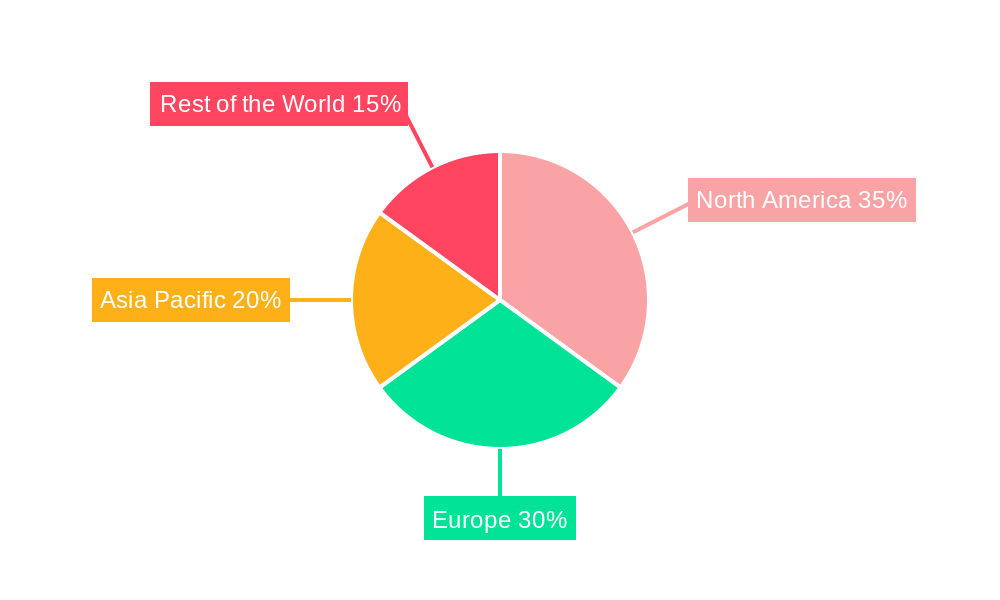
The market is segmented by application into Cardiovascular Diseases, Neurovascular Diseases, and Peripheral Diseases, with Cardiovascular Diseases currently holding the largest share due to the high incidence of conditions requiring such devices. Geographically, North America, particularly the United States, is anticipated to lead the market, followed closely by Europe, owing to advanced healthcare infrastructure, high disposable incomes, and early adoption of new medical technologies. The Asia Pacific region is expected to exhibit the fastest growth, driven by an expanding healthcare sector, increasing medical tourism, and growing awareness of advanced treatment options. While the market presents substantial opportunities, potential restraints include stringent regulatory approvals, high manufacturing costs for sophisticated devices, and the development of alternative treatment modalities. However, the ongoing efforts by leading companies to enhance device efficacy and expand their product portfolios are expected to mitigate these challenges and ensure sustained market growth.
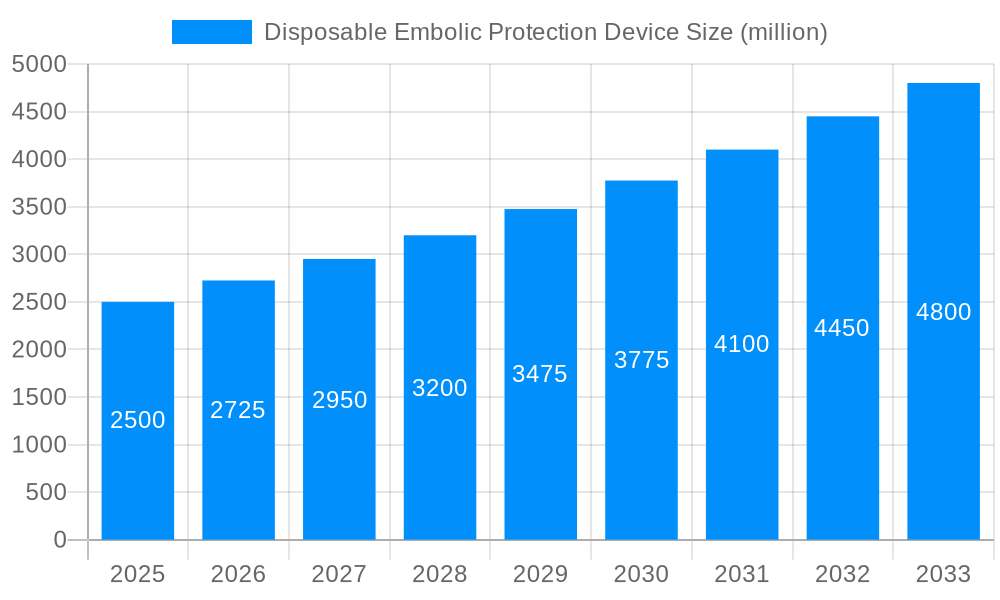

The global Disposable Embolic Protection Device market is poised for significant expansion, driven by an increasing awareness of embolic complication prevention and advancements in minimally invasive procedures. XXX projects a robust trajectory for this market, anticipating a surge from an estimated XXX million units in 2025 to an impressive XXX million units by 2033, reflecting a compound annual growth rate (CAGR) of XXX% during the forecast period. This growth is underpinned by the rising prevalence of cardiovascular and neurovascular diseases, which necessitate sophisticated interventional treatments where embolic protection devices play a crucial role in mitigating stroke and other emboli-related complications.
The historical period from 2019 to 2024 laid a strong foundation for this market, marked by increasing adoption rates and product innovations. The base year of 2025 serves as a critical pivot point, with substantial investments in research and development expected to fuel further market penetration. The demand is particularly strong in developed economies due to higher healthcare expenditure and greater access to advanced medical technologies. However, emerging economies are also witnessing a growing interest, fueled by improving healthcare infrastructure and increasing diagnosis rates of critical diseases. The product landscape is evolving, with a clear trend towards devices offering enhanced filtration efficiency, ease of deployment, and compatibility with a wider range of interventional procedures. The focus on patient safety and improved clinical outcomes remains paramount, pushing manufacturers to continuously innovate and refine their offerings. The integration of advanced materials, such as nitinol, further enhances device performance, offering superior flexibility and biocompatibility, which are key determinants in the success of these disposable devices. The market's dynamism is also influenced by regulatory frameworks and reimbursement policies, which can either accelerate or temper adoption rates across different geographies.
The escalating global burden of cardiovascular and neurovascular diseases is the primary engine driving the demand for disposable embolic protection devices. As interventional cardiology and neurointerventional procedures become more prevalent for treating conditions like atrial fibrillation, stroke, and peripheral artery disease, the need for devices that prevent embolic events during these procedures intensifies. Minimally invasive techniques, while offering significant patient benefits, inherently carry a risk of dislodging atherosclerotic plaque or thrombus, leading to potentially devastating embolic complications. Embolic protection devices act as a critical safeguard, capturing these emboli before they can reach vital organs. Furthermore, a growing emphasis on patient safety and the desire to reduce hospital stays and recovery times associated with stroke further bolster the adoption of these devices. The increasing awareness among healthcare professionals and patients about the benefits of embolic protection during high-risk interventions is a significant catalyst for market growth.
Despite the promising growth trajectory, the disposable embolic protection device market faces several challenges that can impede its full potential. One of the significant restraints is the cost associated with these devices, which can add a substantial economic burden to healthcare systems, particularly in resource-limited settings. The high price point can lead to hesitancy in adoption, especially when reimbursement policies are not fully established or are inadequate. Another hurdle is the potential for procedural complications, although rare, associated with the deployment and retrieval of these devices, such as vessel trauma or device embolization. Continuous training and expertise are required for optimal utilization, and a lack of skilled interventionalists can limit widespread adoption. Regulatory approvals for new devices, while necessary for ensuring safety and efficacy, can be a lengthy and complex process, delaying market entry for innovative products. Moreover, the market is highly competitive, with established players and new entrants vying for market share, leading to price pressures that can affect profit margins for manufacturers.
The Cardiovascular Diseases segment is projected to be the dominant application area for disposable embolic protection devices, largely due to the pervasive nature of these conditions and the increasing volume of interventional cardiovascular procedures performed globally. XXX estimates that this segment alone will account for a substantial portion of the market share, driven by the widespread use of these devices in Percutaneous Coronary Intervention (PCI) and Transcatheter Aortic Valve Replacement (TAVR) procedures.
Within the Cardiovascular Diseases application, specific procedures are expected to drive the most significant demand:
Geographically, while North America is expected to hold a commanding position, Europe is also a significant market, driven by a similar demographic of aging populations and a growing incidence of cardiovascular diseases, coupled with advanced healthcare systems. The Asia-Pacific region, particularly countries like China and India, represents a rapidly growing market due to increasing healthcare expenditure, improving access to medical care, and a rising awareness of stroke prevention.
Several factors are acting as powerful growth catalysts for the disposable embolic protection device industry. The continuous innovation in device design, leading to enhanced filtration efficiency, improved navigability, and simpler deployment mechanisms, is a key driver. The expanding indications for minimally invasive cardiovascular and neurovascular interventions, coupled with a greater emphasis on patient safety and the reduction of procedure-related complications like stroke, are significantly boosting demand. Furthermore, an increasing global aging population, which is more susceptible to cardiovascular and neurovascular diseases, fuels the need for effective preventative measures during interventional procedures.
This comprehensive report provides an in-depth analysis of the global Disposable Embolic Protection Device market, offering valuable insights for stakeholders. It delves into market dynamics, including market size and forecast, segmentation by application, type, and region, and identifies key growth drivers, restraints, and opportunities. The report highlights the competitive landscape, profiling leading companies and their strategic initiatives. Detailed historical data from 2019-2024 and future projections from 2025-2033, with a base year of 2025, ensure a thorough understanding of market trends. The report also examines significant industry developments and regional market potential, empowering businesses to make informed strategic decisions.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.3% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 8.3%.
Key companies in the market include Lepu Medical Technology, Boston Scientific, Medtronic, Venus Medtech, Transverse Medical, Innovative Cardiovascular Solutions, Protembis GmbH, Filterlex Medical, Emboline, Abbott Laboratories, Cordis, Contego Medical, W. L. Gore & Associates, Silk Road Medical, Claret Medical, Allium Medical Solutions, Angioslide, .
The market segments include Application, Type.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Disposable Embolic Protection Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Disposable Embolic Protection Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.