1. What is the projected Compound Annual Growth Rate (CAGR) of the Diabetic Macular Edema Treatment?
The projected CAGR is approximately 5.1%.
 Diabetic Macular Edema Treatment
Diabetic Macular Edema TreatmentDiabetic Macular Edema Treatment by Type (Laser Photocoagulation Therapy, Pharmacological Therapy, Bevacizumab, Ranibizumab, Triamcinolone), by Application (Hospitals, Ophthalmic Clinics, Research Institutes, Contract Research Organizations), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
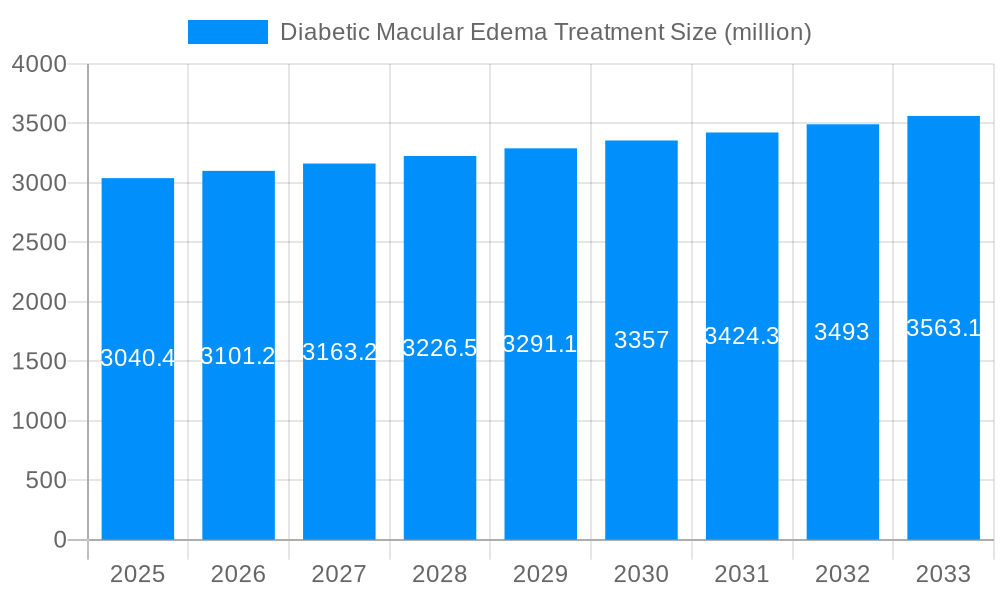
The global Diabetic Macular Edema (DME) treatment market is projected to reach USD 3040.4 million in 2025, exhibiting a Compound Annual Growth Rate (CAGR) of 2.0% throughout the forecast period of 2019-2033. This steady growth is fueled by the escalating prevalence of diabetes worldwide, a primary risk factor for DME. As the diabetic population continues to expand, so does the incidence of related ocular complications like DME, creating a sustained demand for effective treatment solutions. Technological advancements in treatment modalities, including the development of more targeted and less invasive therapies such as advanced laser photocoagulation and novel pharmacological agents, are further bolstering market expansion. The increasing focus on early diagnosis and proactive management of diabetes and its complications by healthcare providers and patients alike is also contributing significantly to market uptake.
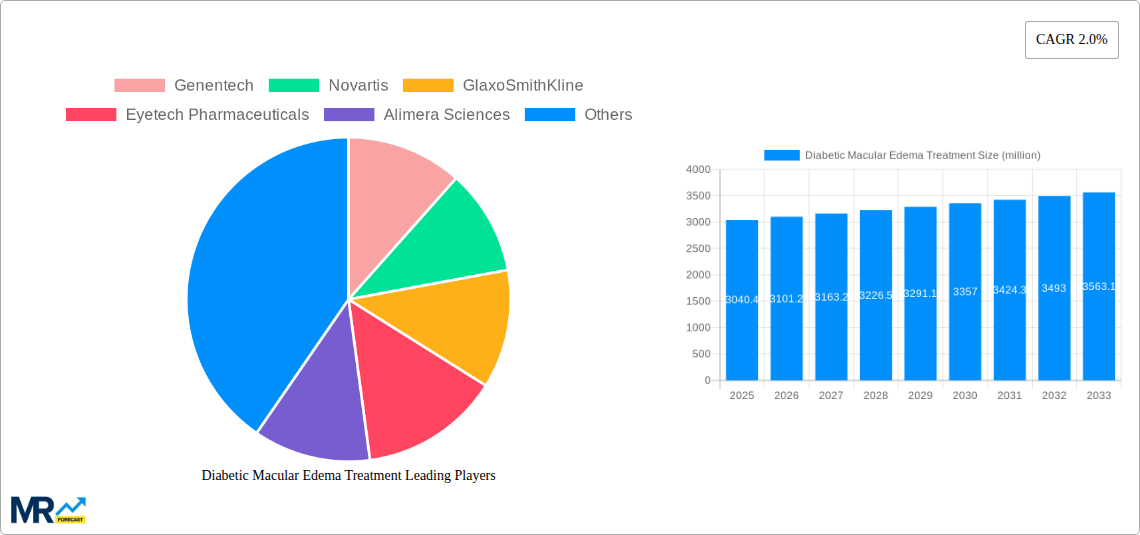

The market is characterized by a competitive landscape with key players like Genentech, Novartis, and GlaxoSmithKline driving innovation and market penetration. The development and commercialization of biologics like Bevacizumab and Ranibizumab have revolutionized DME management, offering improved visual outcomes for patients. However, challenges such as the high cost of certain advanced treatments and limited access to specialized care in developing regions present potential restraints. Nevertheless, the ongoing research into new drug delivery systems, combination therapies, and improved diagnostic tools, alongside the growing adoption of these treatments in hospitals and ophthalmic clinics, are expected to overcome these obstacles. The Asia Pacific region, with its burgeoning population and increasing awareness of diabetes management, is anticipated to emerge as a significant growth avenue for the DME treatment market in the coming years, driven by expanding healthcare infrastructure and rising disposable incomes.

The global Diabetic Macular Edema (DME) treatment market, valued at an estimated $4,500 million in the Base Year of 2025, is poised for robust expansion, projected to reach $9,200 million by the Estimated Year of 2025 and then surge to $18,500 million by 2033, reflecting a compound annual growth rate (CAGR) of approximately 8.5% during the Forecast Period (2025-2033). This significant growth trajectory is underpinned by a confluence of factors, most notably the escalating prevalence of diabetes globally, which directly correlates with the incidence of DME, a leading cause of vision loss among diabetic individuals. During the Historical Period (2019-2024), the market witnessed steady progress, fueled by advancements in therapeutic options and increasing awareness. However, the Study Period (2019-2033) encompasses a dynamic landscape where innovations are rapidly reshaping treatment paradigms.
The market is characterized by a discernible shift towards advanced pharmacological therapies, particularly anti-vascular endothelial growth factor (anti-VEGF) agents, which have largely superseded traditional treatments like laser photocoagulation in many clinical settings due to their superior efficacy and safety profiles in preserving and improving visual acuity. This evolution is further propelled by ongoing research and development efforts aimed at identifying novel therapeutic targets and optimizing existing treatment regimens. Consequently, pharmaceutical companies are investing heavily in R&D, leading to a pipeline of promising new drugs and drug delivery systems. Furthermore, the increasing demand for minimally invasive procedures and the growing adoption of biosimilars are also contributing to market dynamics. The expanding healthcare infrastructure in emerging economies, coupled with favorable reimbursement policies for advanced DME treatments, is also expected to play a crucial role in driving market growth in the coming years. The personalized medicine approach, tailoring treatments based on individual patient responses and genetic predispositions, is also gaining traction, promising more effective and efficient management of DME.
The escalating global burden of diabetes mellitus, a chronic metabolic disorder affecting millions worldwide, stands as the primary engine driving the growth of the Diabetic Macular Edema (DME) treatment market. As the diabetic population continues to swell, so does the incidence of its microvascular complications, with DME being a particularly insidious one that can lead to irreversible vision impairment. This demographic shift, coupled with an aging global population, further exacerbates the situation, as older individuals are more susceptible to both diabetes and its associated complications. Moreover, significant advancements in diagnostic tools and screening programs have led to earlier and more accurate detection of DME, enabling timely intervention and treatment. This increased diagnostic capability directly translates into a larger patient pool seeking effective management solutions. The continuous innovation within the pharmaceutical and biotechnology sectors, resulting in the development of more potent and targeted therapies such as novel anti-VEGF agents and sustained-release drug delivery systems, is another crucial catalyst. These advancements offer improved patient outcomes, greater convenience, and reduced treatment burden, thereby boosting market demand. The growing awareness among healthcare professionals and patients about the available treatment options and the potential for vision preservation also plays a pivotal role in driving market expansion.
Despite the promising growth trajectory, the Diabetic Macular Edema (DME) treatment market faces several hurdles that can potentially restrain its expansion. The high cost associated with advanced pharmacological therapies, particularly novel anti-VEGF agents, presents a significant challenge, especially in resource-limited regions or for patients with inadequate insurance coverage. This cost factor can limit patient access to optimal treatment, leading to delayed or suboptimal management of the condition. Furthermore, the complexity of DME management, often requiring multiple injections and regular follow-up visits, can lead to poor patient compliance, impacting treatment efficacy. The occurrence of adverse events associated with certain treatments, although generally well-tolerated, can also cause apprehension among patients and physicians, leading to a preference for alternative, albeit less effective, interventions. The presence of well-established but less effective treatment modalities like laser photocoagulation also creates a competitive landscape where transitioning patients to newer, more expensive therapies can be a slow process. Regulatory hurdles and the lengthy approval processes for new drugs and delivery systems can also impede market growth. Finally, the need for specialized ophthalmic expertise and advanced infrastructure for administering these treatments can be a limiting factor in certain geographical areas, hindering widespread adoption.
The Pharmacological Therapy segment, specifically driven by the dominance of anti-VEGF agents like Ranibizumab and Bevacizumab, is projected to be the leading segment in the global Diabetic Macular Edema (DME) treatment market. These treatments have revolutionized DME management, offering superior visual acuity outcomes compared to traditional methods. The Ophthalmic Clinics application segment is also expected to dominate, as these specialized centers are equipped with the necessary expertise, technology, and infrastructure to administer intravitreal injections and manage patients with complex ophthalmic conditions like DME.
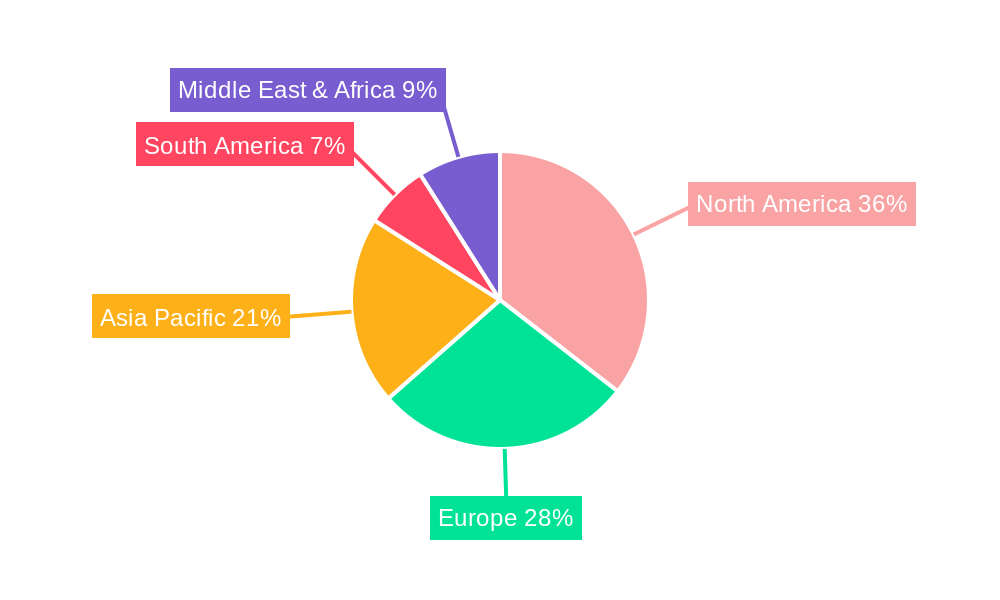
Within the regional landscape, North America is anticipated to maintain its leading position in the DME treatment market. This dominance is attributed to several key factors. Firstly, the region boasts a high prevalence of diabetes and a rapidly aging population, both of which are significant drivers for DME. Secondly, North America has a highly developed healthcare system with robust reimbursement policies that support the adoption of advanced and expensive therapies. The presence of leading pharmaceutical companies like Genentech and Eyetech Pharmaceuticals, heavily invested in R&D and commercialization of DME treatments, further bolsters the market. The high level of awareness among both healthcare providers and patients regarding the importance of timely and effective DME management also contributes to this leadership. Furthermore, the established network of specialized ophthalmic clinics and hospitals ensures access to cutting-edge treatments and expert care.
Following North America, Europe is also a significant contributor to the global DME treatment market. Factors such as increasing diabetes rates, growing awareness of retinal diseases, and a well-established healthcare infrastructure drive demand in this region. Countries like Germany, the UK, and France, with their advanced healthcare systems and strong presence of key market players like Novartis and GlaxoSmithKline, are major contributors.
Emerging markets, particularly in Asia Pacific, are expected to witness the fastest growth rate during the forecast period. This rapid expansion is driven by the escalating prevalence of diabetes, improving healthcare infrastructure, increasing disposable incomes, and a growing focus on chronic disease management. Countries like China and India, with their massive populations and rising rates of diabetes, present substantial untapped potential for DME treatment. The expanding network of ophthalmic clinics and the increasing adoption of advanced treatment modalities in these regions are key indicators of future market dominance.
The Type segment will be overwhelmingly dominated by Pharmacological Therapy. Within this, Bevacizumab and Ranibizumab will continue to command a significant market share due to their established efficacy and widespread use, despite the emergence of newer agents. However, the development of new anti-VEGF molecules and alternative therapeutic approaches like gene therapy and sustained-release implants is expected to diversify the pharmacological landscape, albeit with Bevacizumab and Ranibizumab maintaining strong positions due to cost-effectiveness and physician familiarity. Laser Photocoagulation Therapy, while historically significant, will see its market share diminish as pharmacological treatments demonstrate superior outcomes, relegated primarily to specific niche applications or as an adjunct therapy. Triamcinolone, as a corticosteroid, will continue to play a role, particularly in specific patient populations or when anti-VEGF therapies are insufficient or contraindicated, but it will not overshadow the dominance of anti-VEGF agents.
In terms of Application, Ophthalmic Clinics will remain the dominant channel for DME treatment delivery. Their specialized focus on eye care, coupled with the expertise of ophthalmologists and the availability of necessary equipment for intravitreal injections and diagnostic imaging, makes them the preferred setting for managing DME. Hospitals will also represent a significant application segment, particularly for patients with more severe cases or those requiring inpatient care. Research Institutes and Contract Research Organizations (CROs) will play a crucial role in driving innovation through clinical trials and R&D activities, contributing to the development of future treatments and market expansion indirectly.
The Diabetic Macular Edema (DME) treatment industry is fueled by several key growth catalysts. The escalating global prevalence of diabetes, a primary risk factor for DME, directly translates into a larger patient pool seeking treatment. Continued innovation in drug discovery and development, leading to the introduction of more effective and sustained-release therapeutic agents like novel anti-VEGF drugs and combination therapies, is a major driver. Advancements in diagnostic technologies enabling earlier and more accurate detection of DME also contribute to market expansion. Furthermore, increasing healthcare expenditure in emerging economies, coupled with improved access to specialized eye care facilities, is opening up new markets for DME treatments. The growing awareness among patients and healthcare providers about the importance of preserving vision and managing DME effectively also propels market growth.
This comprehensive report delves into the intricate landscape of Diabetic Macular Edema (DME) treatment, providing invaluable insights for stakeholders. It meticulously analyzes market trends, projecting a robust growth from an estimated $4,500 million in 2025 to $18,500 million by 2033. The report highlights the driving forces behind this expansion, primarily the escalating global diabetes epidemic and continuous therapeutic innovations. It also critically examines the challenges and restraints, such as high treatment costs and patient compliance issues, that influence market dynamics. Furthermore, the report identifies key regions and segments poised for dominance, emphasizing the leading roles of pharmacological therapies, particularly anti-VEGF agents, and specialized ophthalmic clinics. Leading market players and significant recent and upcoming developments are also detailed. This extensive coverage offers a strategic roadmap for understanding the current market, anticipating future trajectories, and identifying lucrative opportunities within the Diabetic Macular Edema treatment sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.1% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.1%.
Key companies in the market include Genentech, Novartis, GlaxoSmithKline, Eyetech Pharmaceuticals, Alimera Sciences, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Diabetic Macular Edema Treatment," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Diabetic Macular Edema Treatment, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.