1. What is the projected Compound Annual Growth Rate (CAGR) of the Dendritic Cell Cancer Vaccines?
The projected CAGR is approximately 13.05%.
 Dendritic Cell Cancer Vaccines
Dendritic Cell Cancer VaccinesDendritic Cell Cancer Vaccines by Type (CreaVax, Sipuleucel-T (Provenge), Others), by Application (Pediatrics, Adults), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
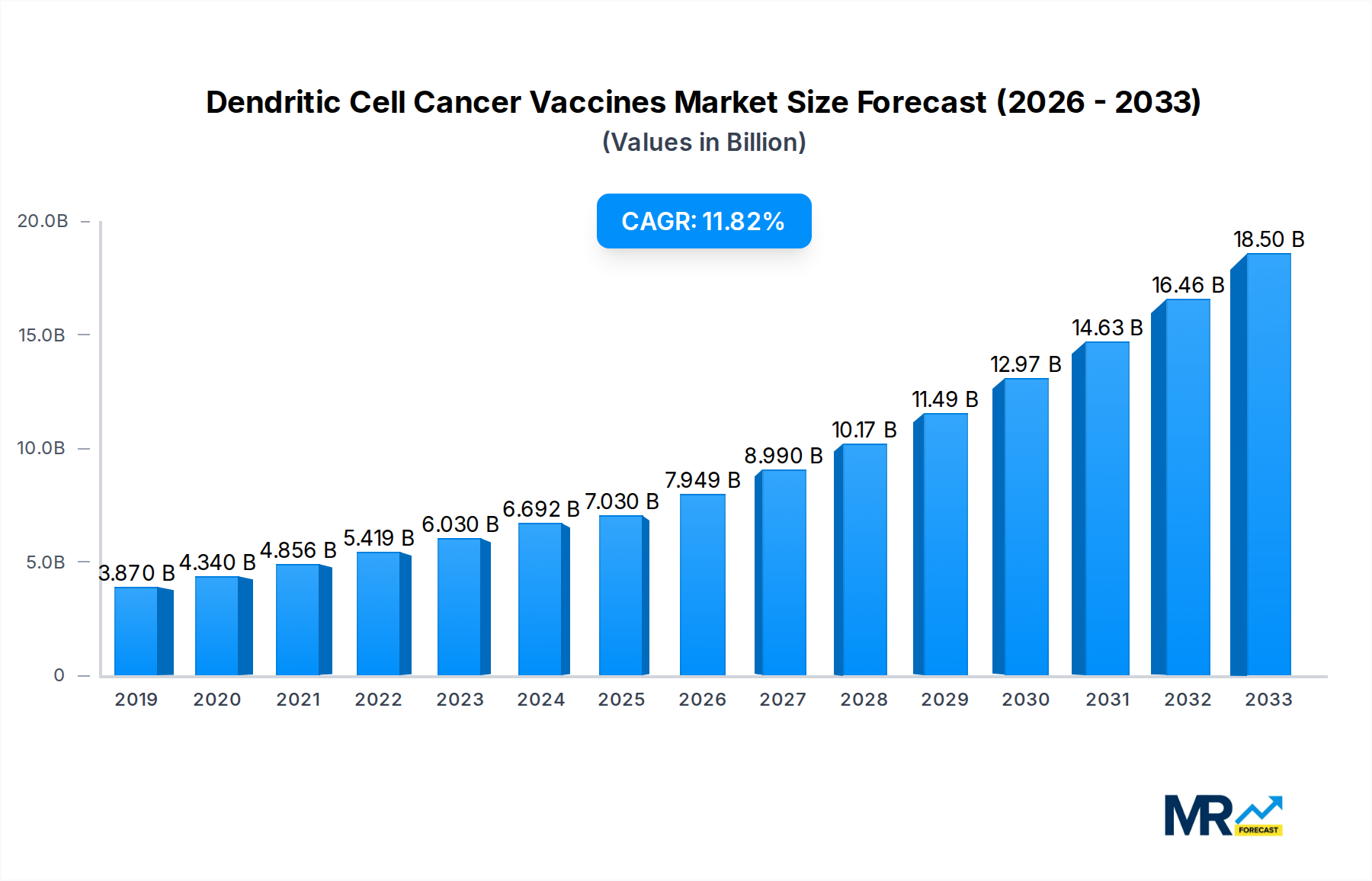
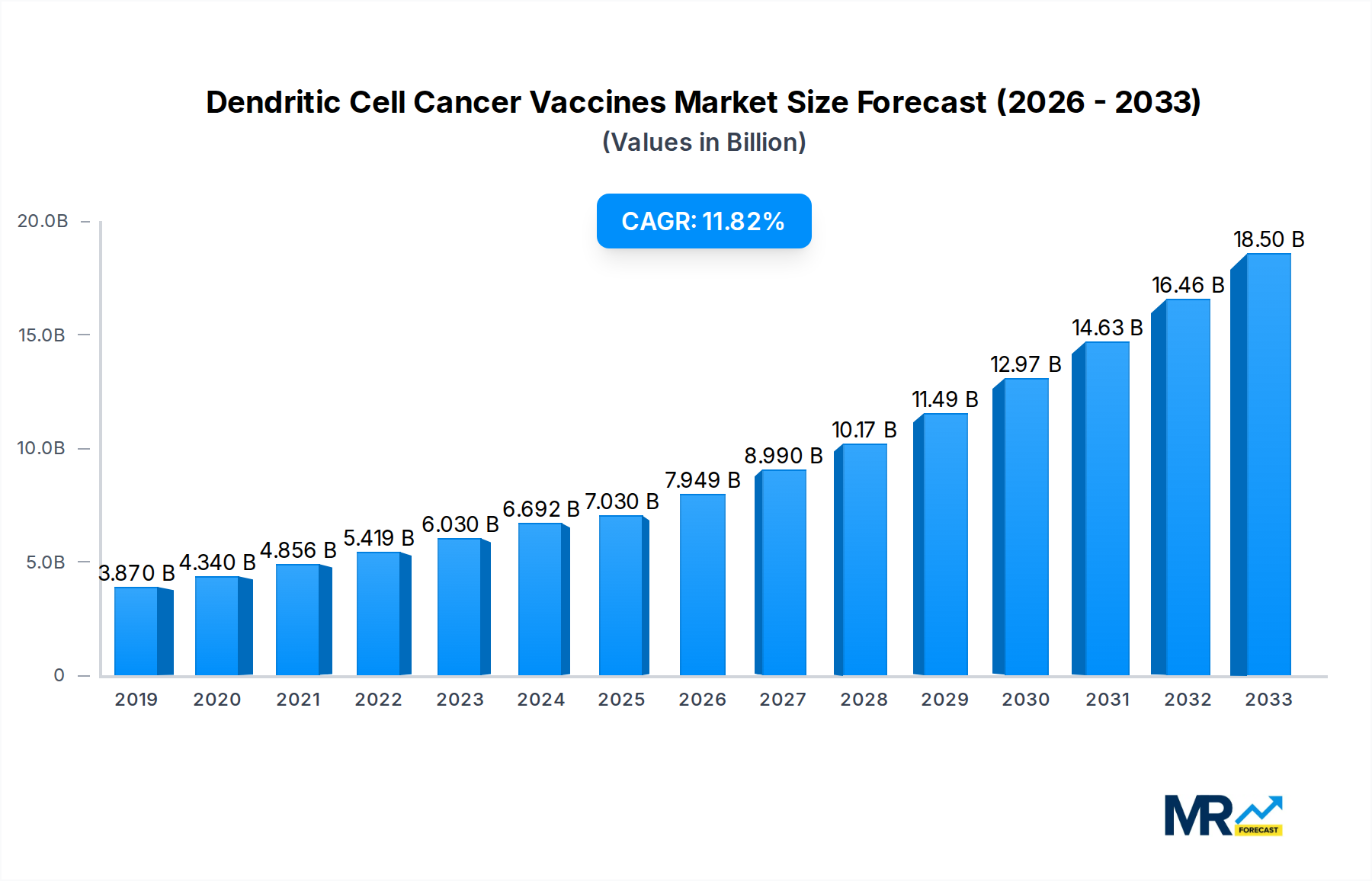
The global Dendritic Cell Cancer Vaccines market is poised for substantial growth, projected to reach approximately $618.7 million by 2025, with an impressive Compound Annual Growth Rate (CAGR) of 12.6%. This robust expansion is fueled by a confluence of factors, including increasing investments in cancer research and development, a growing understanding of the immune system's role in combating malignancies, and advancements in cell-based therapy technologies. The market's trajectory is further bolstered by the rising incidence of various cancers worldwide, creating a persistent demand for innovative and effective treatment modalities. Key drivers include the development of personalized cancer vaccines, aiming to tailor treatments to individual patient profiles for enhanced efficacy and reduced side effects. Early detection initiatives and improved diagnostic tools are also contributing to a larger patient pool amenable to advanced therapies like dendritic cell vaccines.

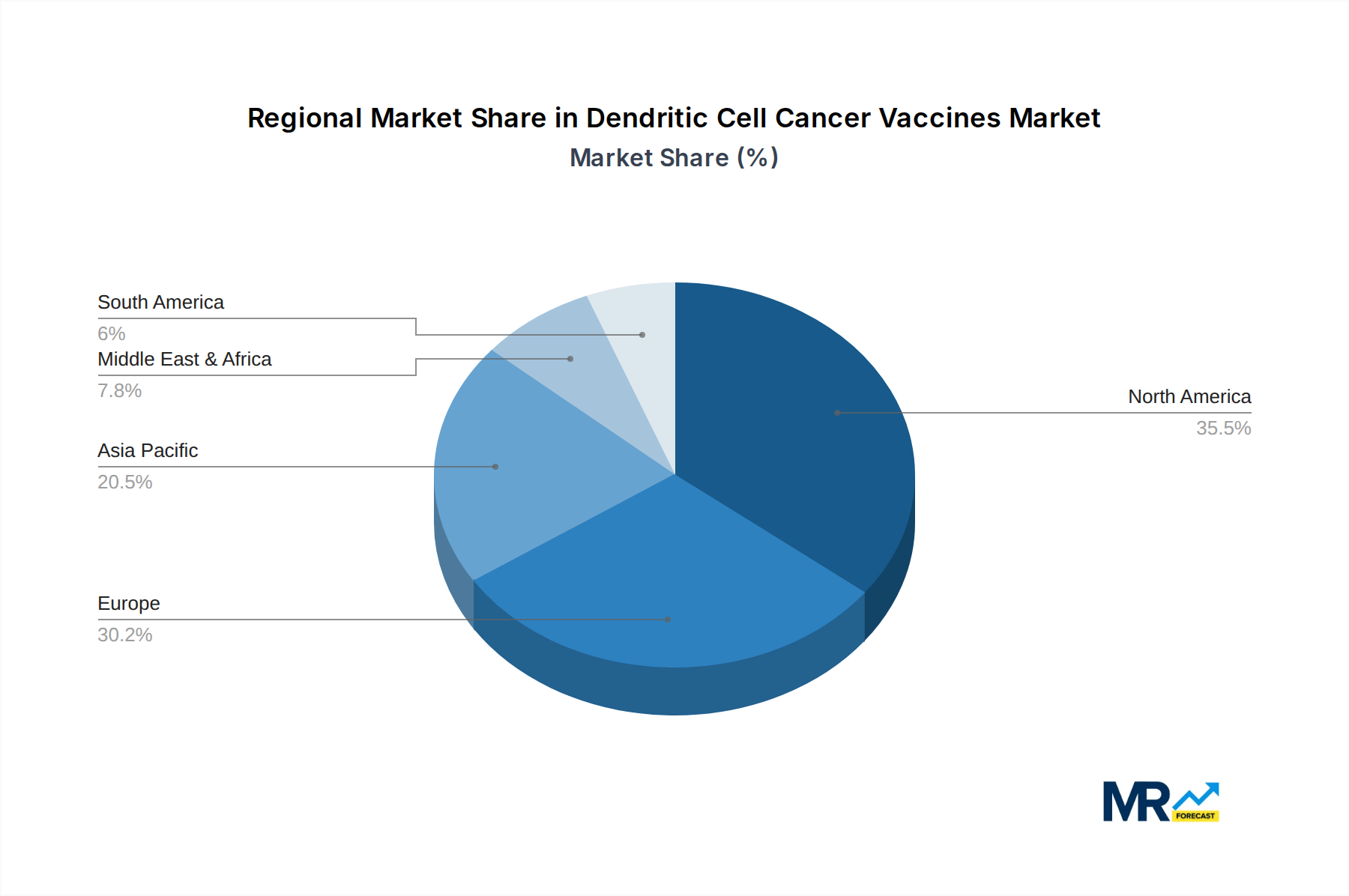
The competitive landscape features a diverse range of companies, from established pharmaceutical giants like Merck and Glaxo Smith Kline to specialized biotech firms such as ImmmunoCellular Therapeutics and Northwest Biotherapeutics. These players are actively engaged in research, clinical trials, and strategic collaborations to bring novel dendritic cell cancer vaccines to market. The market is segmented by type, with CreaVax and Sipuleucel-T (Provenge) being prominent examples, and application areas encompassing both pediatrics and adults. Geographically, North America, particularly the United States, is expected to lead the market, driven by significant healthcare expenditure, advanced research infrastructure, and a strong pipeline of ongoing clinical trials. Europe and Asia Pacific are also anticipated to witness considerable growth, owing to expanding healthcare access and increasing adoption of novel cancer immunotherapies. Challenges such as high treatment costs and the need for specialized infrastructure remain, but the overwhelming potential of dendritic cell therapy in addressing unmet medical needs in oncology suggests a bright future for this market.

Here's a unique report description on Dendritic Cell Cancer Vaccines, incorporating your specified elements:
The global Dendritic Cell Cancer Vaccines market is poised for significant expansion, projected to reach an estimated value of $2,450 million by 2025, a substantial leap from the $875 million recorded in the historical period of 2019-2024. This growth trajectory, meticulously analyzed within the Study Period of 2019-2033, highlights a burgeoning interest and investment in this advanced form of immunotherapy. The Base Year of 2025 serves as a critical juncture, reflecting the current momentum and the anticipated acceleration of market adoption. Several key market insights underscore this optimistic outlook. The increasing understanding of the human immune system's role in fighting cancer has propelled dendritic cell (DC) vaccines to the forefront of oncological research. These vaccines leverage the unique antigen-presenting capabilities of dendritic cells to prime the immune system to recognize and attack tumor cells. The personalized nature of many DC vaccine approaches, where patient-specific tumor antigens are used, offers a distinct advantage over traditional therapies, leading to improved efficacy and reduced side effects. Furthermore, the market is witnessing a steady stream of clinical trial successes across various cancer types, bolstering investor confidence and paving the way for regulatory approvals. The anticipated Compound Annual Growth Rate (CAGR) for the Forecast Period of 2025-2033 is robust, suggesting a market value that could surpass $7,200 million by the end of the forecast horizon. This expansion is not merely driven by incremental improvements but by a fundamental shift towards immunotherapeutic strategies in cancer treatment. The integration of advanced manufacturing techniques and the growing pipeline of DC-based therapies are also contributing factors. The market’s evolution is characterized by increasing collaboration between academic institutions and pharmaceutical giants, fostering innovation and accelerating the translation of research into clinical practice. The demand for more targeted and less toxic cancer treatments is a persistent driver, and DC vaccines are uniquely positioned to meet this demand.
The escalating incidence of cancer globally is a primary impetus behind the surging demand for advanced treatment modalities, including dendritic cell cancer vaccines. As the healthcare landscape shifts towards personalized medicine, DC vaccines, with their inherent ability to elicit a highly specific immune response against cancer cells, are gaining significant traction. The mechanism of action, wherein dendritic cells are engineered to present tumor-associated antigens to T-cells, effectively "educating" the immune system to identify and eliminate cancerous growths, is a paradigm shift in oncology. This personalized approach holds the promise of improved treatment outcomes and a reduction in the debilitating side effects associated with conventional chemotherapy and radiation. Furthermore, a substantial increase in research and development funding, both from public and private sectors, has accelerated the clinical evaluation of DC-based therapies across a spectrum of malignancies. This sustained investment is crucial for navigating the complex regulatory pathways and bringing these innovative treatments to patients. The growing recognition of the immune system's potential in cancer eradication has fostered a climate of optimism and innovation within the field, encouraging further exploration and refinement of DC vaccine technologies. The market is also benefiting from advancements in biotechnology, particularly in cell processing, genetic engineering, and antigen identification, which are making DC vaccine development more efficient and scalable.
Despite the promising outlook, the dendritic cell cancer vaccine market grapples with several significant challenges that temper its growth potential. A primary hurdle remains the high cost of manufacturing and development. The intricate process of isolating, culturing, and activating patient-specific dendritic cells, coupled with rigorous quality control measures, contributes to substantial production expenses. This can translate into prohibitively high treatment costs for patients, limiting accessibility, particularly in resource-constrained regions. Furthermore, complex manufacturing processes and logistical complexities in handling autologous (patient-derived) cell products pose operational challenges, demanding specialized infrastructure and trained personnel. The regulatory landscape for cell-based therapies is still evolving, with lengthy and stringent approval processes that can delay market entry and increase R&D expenditure. Variability in patient immune responses and the potential for limited efficacy in certain cancer types or stages also present clinical challenges. Tumor heterogeneity and the immunosuppressive tumor microenvironment can hinder the effectiveness of even well-designed DC vaccines. Finally, limited patient and physician awareness and understanding of this novel therapeutic modality can act as a restraint, requiring concerted efforts in education and outreach to facilitate broader adoption.
The Adults application segment is projected to hold a commanding share of the Dendritic Cell Cancer Vaccines market, driven by several pivotal factors. Adults represent the vast majority of cancer diagnoses globally, and their more robust immune systems often respond more favorably to immunotherapy. The extensive clinical research and development efforts are predominantly focused on adult indications, leading to a richer pipeline of approved and investigational dendritic cell vaccines for common adult cancers such as prostate, lung, and melanoma. For instance, Sipuleucel-T (Provenge), a well-established dendritic cell vaccine, has demonstrated significant efficacy in treating advanced prostate cancer in adults, setting a precedent and validating the therapeutic potential of this modality. The market penetration of Provenge, alongside ongoing advancements in developing personalized dendritic cell vaccines for other adult malignancies, contributes to the dominance of this segment.
Adults Application Dominance:
North America and Europe Leading Regions:
The 'Others' Segment under Type:
The projected dominance of the Adults application segment within the Dendritic Cell Cancer Vaccines market is underpinned by a confluence of epidemiological, clinical, and economic factors. The sheer volume of adult cancer diagnoses worldwide creates an enormous patient pool for which these therapies are being developed and tested. Clinical trial success and subsequent regulatory approvals have predominantly focused on adult indications, solidifying the position of dendritic cell vaccines as a viable treatment option for a range of common and complex adult cancers. Furthermore, the economic landscape within these regions, characterized by higher disposable incomes and advanced healthcare systems, enables greater accessibility to expensive but potentially life-saving immunotherapies. The presence of leading research institutions and pharmaceutical companies, particularly in North America and Europe, further fuels this dominance. These regions are hubs for cutting-edge research, clinical development, and manufacturing capabilities, attracting significant investment and fostering an environment conducive to innovation and market penetration for dendritic cell vaccines. The established reimbursement policies and the increasing acceptance of personalized medicine approaches in these areas also play a crucial role in driving the demand and adoption of dendritic cell cancer vaccines for adult patients. The 'Others' segment under 'Type' also contributes significantly, as it captures the potential of the myriad of experimental and novel dendritic cell vaccine designs that are continuously being explored, representing the future growth frontiers of the market.
Several factors are acting as significant growth catalysts for the dendritic cell cancer vaccines industry. The burgeoning understanding of cancer immunology and the immense potential of harnessing the patient's own immune system to combat tumors is a fundamental driver. Advancements in genetic engineering and bioinformatics are enabling more precise identification of tumor-specific antigens, leading to the development of highly personalized and effective vaccines. Furthermore, the growing pipeline of clinical trials demonstrating positive efficacy signals across various cancer types is bolstering investor confidence and accelerating R&D investments, projected to exceed $1,200 million in R&D spending by 2025.
This comprehensive report delves into the intricacies of the global Dendritic Cell Cancer Vaccines market, offering a detailed analysis of its trajectory from 2019 to 2033. It provides in-depth insights into market dynamics, including segmentation by Type (CreaVax, Sipuleucel-T (Provenge), Others), Application (Pediatrics, Adults), and key Industry Developments. The report utilizes 2025 as the Base Year and Estimated Year, with a thorough examination of the Historical Period (2019-2024) and projections for the Forecast Period (2025-2033). It highlights the significant growth catalysts, such as the rising incidence of cancer and advancements in immunotherapy research, projected to drive the market value to an estimated $2,450 million by 2025. Furthermore, the report addresses the challenges and restraints, including high manufacturing costs and regulatory complexities, while identifying key regions and segments poised for dominance. Leading players and significant developments are meticulously documented, providing a holistic view of this rapidly evolving therapeutic landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.05% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 13.05%.
Key companies in the market include 3M Company, Activarti, Argos Therapeutics, Batavia Bioservices, Bellicum Pharmaceuticals, Creagene, DanDrit Biotech, DCPrime, Sanpower Corporation, Elios Therapeutics, ImmunoCellular Therapeutics, Immunicum, Kiromic, Medigene, Merck, Northwest Biotherapeutics, Glaxo Smith Kline, Tellaorporation, Vaxil BioTherapeutics, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Dendritic Cell Cancer Vaccines," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Dendritic Cell Cancer Vaccines, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.