1. What is the projected Compound Annual Growth Rate (CAGR) of the Critical Care Therapeutics?
The projected CAGR is approximately 10.96%.
 Critical Care Therapeutics
Critical Care TherapeuticsCritical Care Therapeutics by Application (Pulmonary Embolism (PE), Acute Coronary Syndrome, Atrial Fibrillation, Other), by Type (Albumin, Prothrombin Complex Concentrates, Antithrombin Concentrates, Factor XIII Concentrates, Fibrinogen Concentrates), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Critical Care Therapeutics market is projected to reach approximately $5.86 billion by 2025, exhibiting a Compound Annual Growth Rate (CAGR) of 10.96% during the forecast period. This robust growth is fueled by the rising prevalence of chronic diseases, an aging global population, and advancements in therapeutic interventions. Significant application segments like Pulmonary Embolism (PE) and Acute Coronary Syndrome (ACS) are expected to drive demand, alongside increasing incidences of cardiovascular and thromboembolic disorders. The robust demand for Albumin and Prothrombin Complex Concentrates is attributed to their proven efficacy in managing bleeding disorders and critical liver-related illnesses. Continuous research and development in novel drug delivery systems and targeted therapies will further accelerate market expansion.
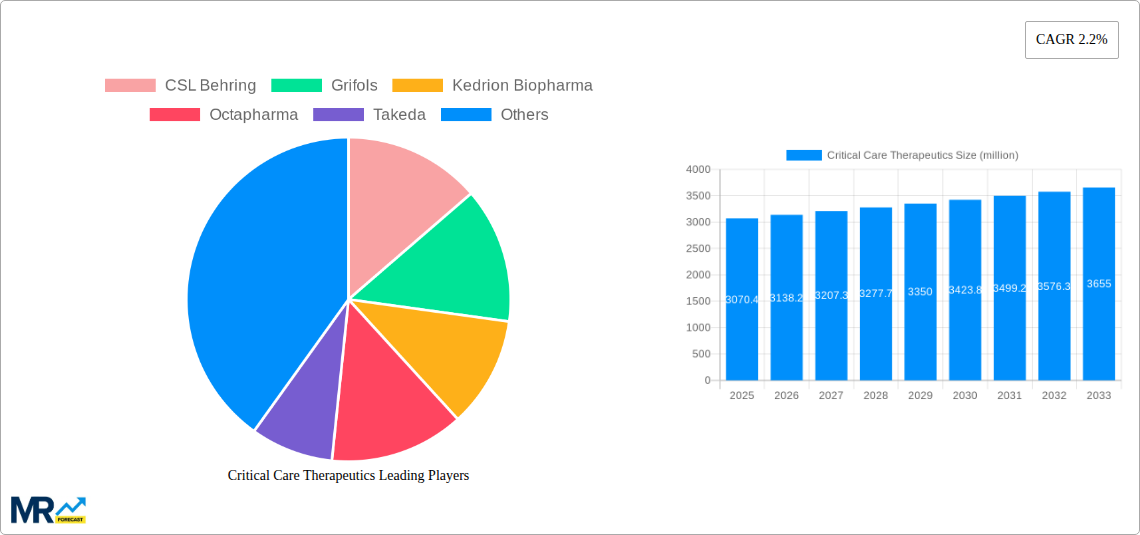

Key market drivers include escalating healthcare expenditure in emerging economies, increased focus on early diagnosis and treatment of critical conditions, and strategic collaborations and acquisitions among leading pharmaceutical and biotechnology firms. Restraints include the high cost of certain therapeutics, stringent regulatory pathways, and the potential for adverse drug reactions. North America and Europe are anticipated to lead the market, supported by advanced healthcare infrastructures and high adoption rates of cutting-edge treatments. The Asia Pacific region offers substantial growth potential due to its expanding patient demographic and improving healthcare accessibility. Leading players such as CSL Behring, Grifols, and Takeda are pivotal in shaping the market through innovation and comprehensive product portfolios.

This report provides an in-depth analysis of the Critical Care Therapeutics market, covering market size, growth projections, and key trends.
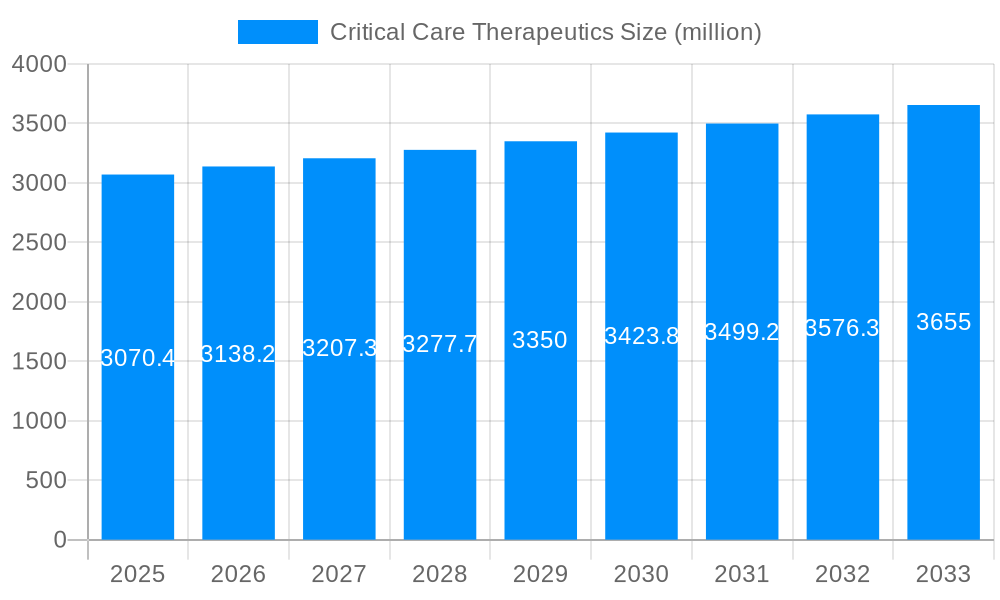
The global Critical Care Therapeutics market is poised for significant expansion, projected to reach an estimated $25,500 million in the base year of 2025. This robust growth trajectory, expected to continue through the forecast period of 2025-2033, is underpinned by a confluence of factors including an aging global population, a rising incidence of chronic diseases, and advancements in diagnostic and therapeutic technologies. The historical period from 2019 to 2024 saw steady market development, with the critical care segment demonstrating resilience and adaptability. Key market insights reveal a notable shift towards more targeted and personalized therapies, particularly in managing life-threatening conditions like Pulmonary Embolism (PE) and Acute Coronary Syndrome (ACS). The demand for plasma-derived therapeutics, such as Albumin and Prothrombin Complex Concentrates, remains exceptionally high due to their crucial role in fluid management and hemostasis in critically ill patients. Furthermore, increasing healthcare expenditure globally and the expansion of healthcare infrastructure in emerging economies are contributing to broader market access and adoption of these vital treatments. The study period of 2019-2033 highlights a sustained upward trend, driven by both unmet clinical needs and ongoing innovation. The estimated market size for 2025 underscores the current demand, while the forecast period indicates a strong future outlook for the critical care therapeutics landscape. The report will delve into the nuanced dynamics shaping this market, from the prevalence of specific critical conditions to the evolving regulatory frameworks and reimbursement policies that influence market access and patient care.
The escalating prevalence of chronic diseases worldwide serves as a primary impetus for the growth of the critical care therapeutics market. Conditions such as cardiovascular diseases, respiratory illnesses, and bleeding disorders, which often necessitate intensive medical intervention, are becoming more common, particularly in aging populations. This demographic shift, characterized by a larger proportion of individuals susceptible to critical illnesses, directly translates into a higher demand for therapeutic solutions. Moreover, advancements in medical technology and a deeper understanding of disease pathophysiology are enabling the development of more sophisticated and effective critical care treatments. The availability of novel therapeutic agents and improved diagnostic tools allows for earlier and more accurate identification of critical conditions, leading to timely intervention and better patient outcomes. This, in turn, fuels market growth as healthcare providers adopt these advanced therapies. Increased healthcare spending and a growing emphasis on patient-centric care also contribute significantly. Governments and private entities are investing more in healthcare infrastructure and research, fostering an environment conducive to innovation and market expansion. The continuous drive to improve patient survival rates and quality of life in critical care settings ensures sustained demand for innovative therapeutic solutions.
Despite the promising growth trajectory, the critical care therapeutics market faces several significant challenges. One of the most prominent is the high cost associated with the development and manufacturing of these specialized therapies. The complex research and development processes, stringent regulatory approvals, and specialized production requirements contribute to substantial upfront investments and ongoing operational expenses. This can lead to high price points for critical care therapeutics, posing a barrier to access, particularly in resource-limited settings and for healthcare systems with constrained budgets. Furthermore, the stringent regulatory landscape governing the approval of new drugs and biologics in critical care can be a lengthy and arduous process. Meeting the exacting standards set by regulatory bodies requires extensive clinical trials and data submission, which can delay market entry and increase development costs. Another significant restraint is the limited pool of specialized healthcare professionals equipped to administer and manage complex critical care treatments. A shortage of trained intensivists, pharmacists, and nurses can hinder the effective utilization of available therapeutics. Finally, the evolving nature of critical care itself, with new pathogens and resistant strains emerging, necessitates continuous adaptation and innovation, presenting an ongoing challenge for therapeutic development.
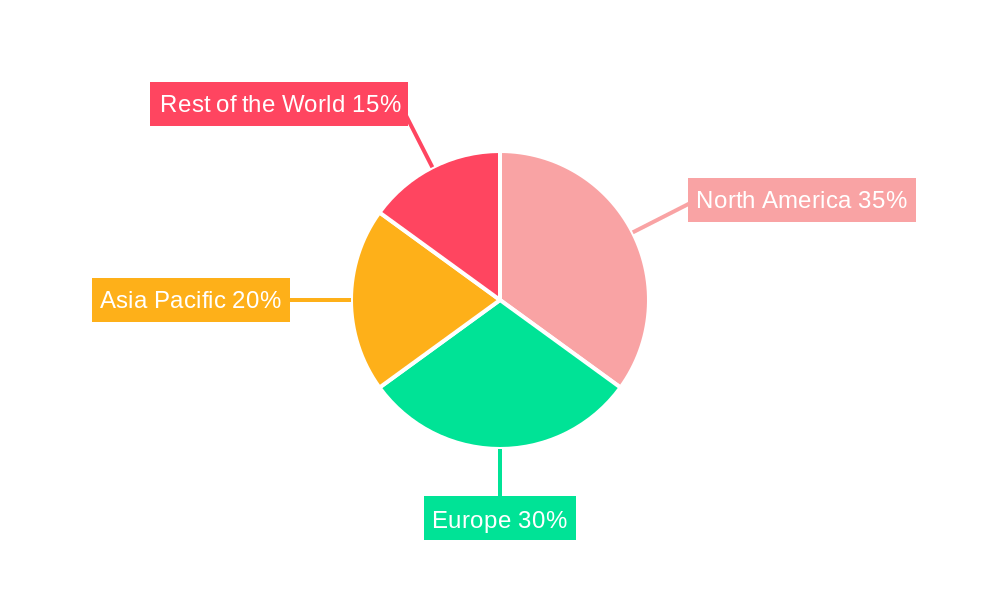
The North America region is projected to dominate the critical care therapeutics market, driven by a robust healthcare infrastructure, high healthcare expenditure, and a strong emphasis on advanced medical research and development. The United States, in particular, stands out due to its well-established reimbursement policies for critical care interventions, a high prevalence of chronic diseases, and a leading role in pharmaceutical innovation. The presence of major biopharmaceutical companies and a sophisticated network of research institutions further solidify North America's leadership position.
Within the Application segment, Pulmonary Embolism (PE) and Acute Coronary Syndrome (ACS) are anticipated to be major contributors to market growth. The increasing incidence of these life-threatening cardiovascular and pulmonary conditions, often linked to sedentary lifestyles, aging populations, and other comorbidities, necessitates a significant demand for effective therapeutic interventions. For PE, anticoagulants and thrombolytics are crucial, while ACS management relies heavily on antiplatelets, anticoagulants, and supportive therapies aimed at stabilizing cardiac function and preventing further damage. The widespread adoption of advanced diagnostic techniques for early detection further fuels the demand for treatments in these areas.
In terms of Type, Albumin is expected to maintain a dominant position. Albumin plays a critical role in maintaining oncotic pressure, fluid resuscitation, and transporting various substances in critically ill patients, making it an indispensable therapeutic agent in intensive care units. Its application spans a wide range of conditions, including sepsis, shock, and severe burns. Following closely are Prothrombin Complex Concentrates (PCCs) and Antithrombin Concentrates. PCCs are vital for reversing anticoagulation and managing bleeding in patients on anticoagulant therapy or those with specific coagulation factor deficiencies. Antithrombin concentrates are essential for managing patients with hereditary or acquired antithrombin deficiency, particularly those at risk of thromboembolism. The increasing use of anticoagulant therapies has, in turn, created a greater need for reversal agents like PCCs.
The Other application segment, which encompasses a broad spectrum of critical conditions such as sepsis, severe infections, and multi-organ dysfunction syndrome, is also expected to exhibit substantial growth. This is due to the complexity and heterogeneity of critical care medicine, where a diverse range of therapeutic needs arise.
The market segmentation analysis reveals a dynamic interplay between the types of therapeutics and the specific critical conditions they address. The continued innovation in plasma-derived products and the development of recombinant alternatives will further shape the competitive landscape. The increasing focus on personalized medicine and the development of targeted therapies for specific patient populations will also play a crucial role in segment growth.
The critical care therapeutics industry is experiencing robust growth fueled by several key catalysts. The burgeoning global elderly population, inherently more susceptible to critical illnesses, presents a sustained demand for these life-saving treatments. Concurrently, the rising incidence of chronic diseases, such as cardiovascular and respiratory ailments, necessitates intensive medical intervention, further propelling market expansion. Significant investments in research and development by leading pharmaceutical companies are yielding a pipeline of innovative therapies, including novel biologics and gene therapies, designed to address unmet clinical needs in critical care settings. Furthermore, advancements in diagnostic technologies are enabling earlier and more accurate identification of critical conditions, leading to timely and effective treatment.
This comprehensive report provides an in-depth analysis of the Critical Care Therapeutics market, encompassing a detailed examination of market trends, growth drivers, and prevailing challenges. The study meticulously analyzes key market segments across applications like Pulmonary Embolism (PE), Acute Coronary Syndrome, Atrial Fibrillation, and Other critical conditions. It also delves into the market dynamics of various therapeutic types, including Albumin, Prothrombin Complex Concentrates, Antithrombin Concentrates, Factor XIII Concentrates, and Fibrinogen Concentrates. The report offers precise market size estimations for the base year (2025) and projects market value for the forecast period (2025-2033), based on a comprehensive study period from 2019 to 2033. Furthermore, it highlights significant industry developments, identifies leading market players, and provides insights into key regional market dominance, offering a holistic view of the global critical care therapeutics landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 10.96% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 10.96%.
Key companies in the market include CSL Behring, Grifols, Kedrion Biopharma, Octapharma, Takeda, Bayer HealthCare, Merck, Mitsubishi Tanabe Pharma, REVO Biologics, Rockwell Medical, Thermo Fisher Scientific, Shanghai RAAS, China Biologic Products, Cosmo Pharmaceuticals, King Pharmaceuticals, .
The market segments include Application, Type.
The market size is estimated to be USD 5.86 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion.
Yes, the market keyword associated with the report is "Critical Care Therapeutics," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Critical Care Therapeutics, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.