1. What is the projected Compound Annual Growth Rate (CAGR) of the Continuous Flow Apheresis Device?
The projected CAGR is approximately 12.2%.
 Continuous Flow Apheresis Device
Continuous Flow Apheresis DeviceContinuous Flow Apheresis Device by Type (Plasma Separators, Plasma Component Separators, Immunoadsorption Columns, Plasma Perfusion Columns, Hemoperfusion Columns), by Application (Therapeutic Plasma Exchange (TPE), Therapeutic Cytapheresis, Photopheresis, Extracorporeal Immunoadsorption (ECI)), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Continuous Flow Apheresis Device market is experiencing robust expansion, projected to reach approximately $4673.2 million by the estimated year of 2025. This growth is underpinned by an impressive Compound Annual Growth Rate (CAGR) of 12.2%, indicating a dynamic and rapidly evolving sector. The increasing prevalence of chronic diseases, such as autoimmune disorders, kidney failure requiring dialysis, and certain hematological conditions, is a primary driver for the demand for apheresis procedures. These therapies play a crucial role in removing harmful substances from the blood, thereby offering vital treatment options for patients. Advancements in device technology, leading to improved efficiency, patient comfort, and safety, are further propelling market adoption. Furthermore, rising healthcare expenditure globally and a growing awareness among healthcare professionals and patients regarding the benefits of apheresis are contributing significantly to this upward trajectory. The market's expansion is also facilitated by an increasing number of clinical applications being explored and approved for apheresis, broadening its therapeutic reach.
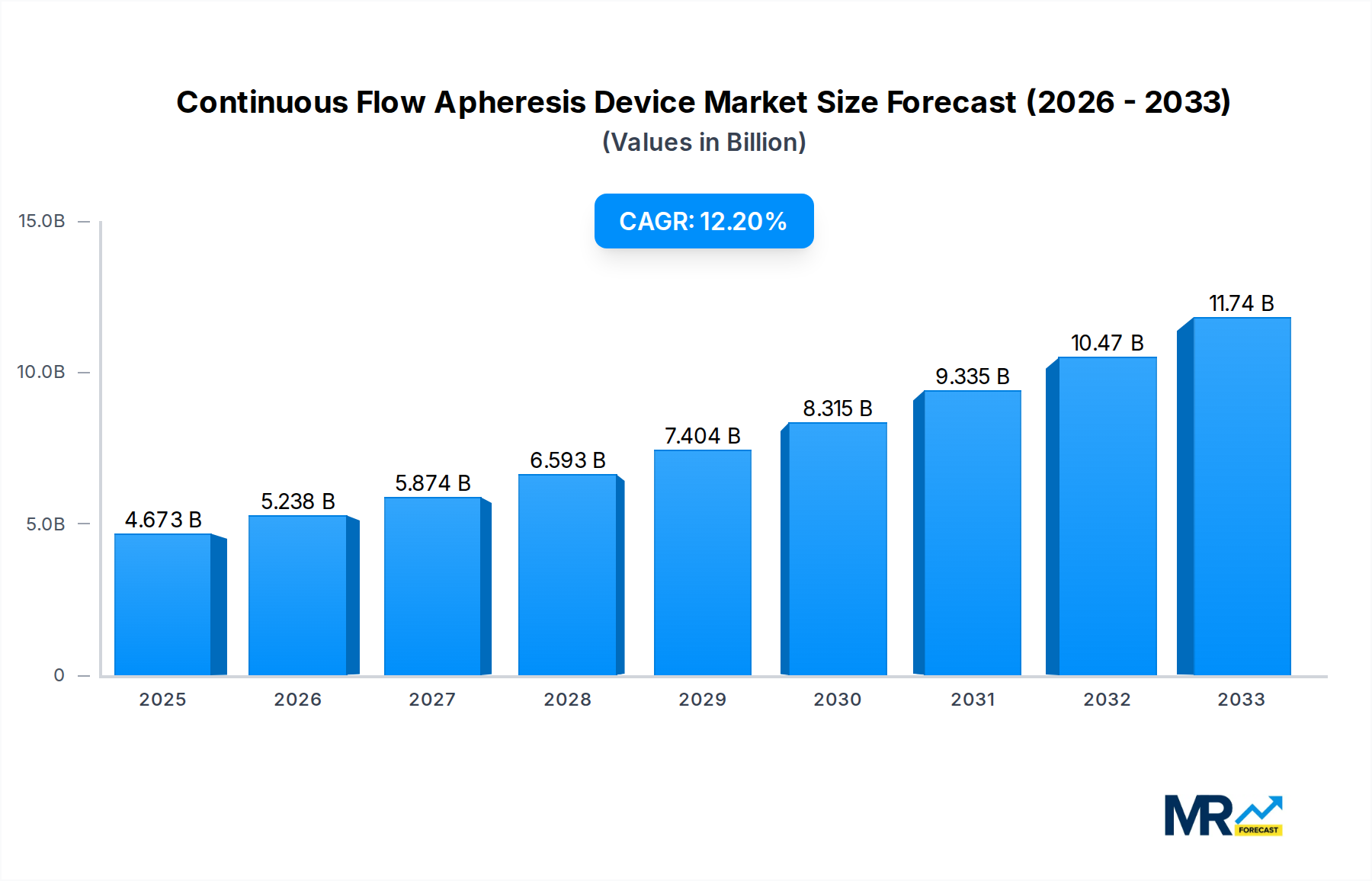

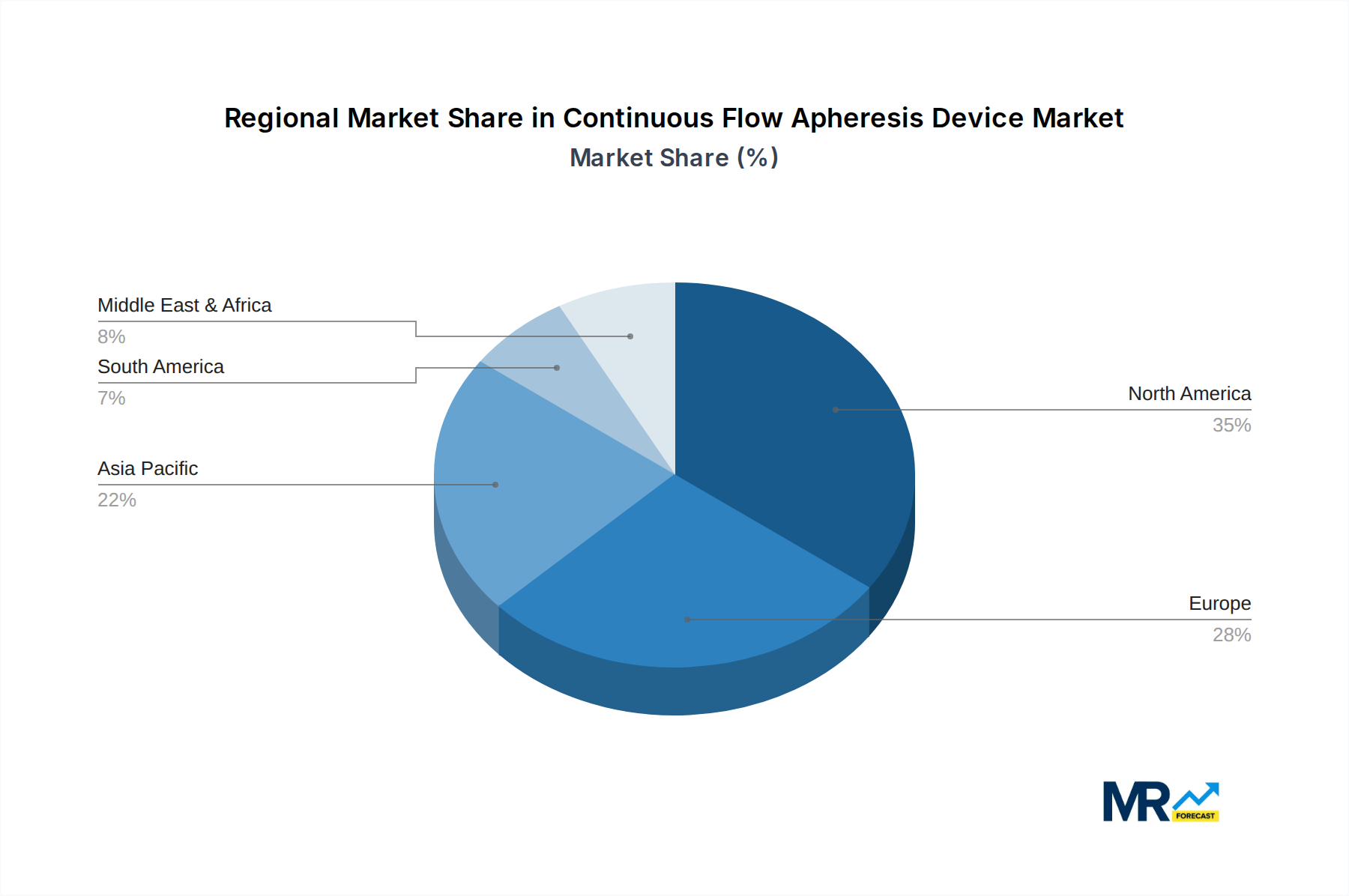
The market segmentation reveals a diverse landscape with significant opportunities across various device types and applications. Plasma Separators and Plasma Component Separators are foundational to many apheresis procedures, while specialized devices like Immunoadsorption Columns and Hemoperfusion Columns are gaining traction for targeted therapies. Therapeutic Plasma Exchange (TPE) remains a cornerstone application, but the growth in Therapeutic Cytapheresis, Photopheresis, and Extracorporeal Immunoadsorption (ECI) highlights the expanding utility of apheresis in treating a wider spectrum of conditions. Leading global players such as Fresenius Medical Care, B. Braun Melsungen AG, and Terumo BCT, Inc. are at the forefront of innovation and market penetration, driven by extensive research and development and a strong distribution network. The geographical distribution indicates a strong presence in North America and Europe, with the Asia Pacific region poised for substantial growth due to improving healthcare infrastructure and increasing patient access to advanced medical treatments. The forecast period from 2025 to 2033 suggests continued strong performance, driven by ongoing technological innovations and the expanding clinical evidence supporting apheresis therapies.

Here is a unique report description for a Continuous Flow Apheresis Device market analysis, incorporating your specified elements:
The global Continuous Flow Apheresis Device market is poised for substantial growth, projected to expand from an estimated $1,500 million in the base year of 2025 to a remarkable $3,200 million by the end of the forecast period in 2033, exhibiting a compound annual growth rate (CAGR) of approximately 8.5%. This robust expansion is underpinned by several converging trends that are reshaping the landscape of blood component separation and therapeutic interventions. The increasing prevalence of chronic diseases, such as autoimmune disorders, hematological malignancies, and kidney diseases, is a primary driver, necessitating advanced apheresis procedures for disease management and treatment. Furthermore, the ongoing advancements in technology, leading to more efficient, precise, and patient-friendly apheresis devices, are significantly contributing to market penetration. The shift towards minimally invasive therapeutic approaches further bolsters the demand for these sophisticated devices. The market is witnessing a growing adoption of point-of-care apheresis systems, enhancing accessibility and reducing treatment times, which is particularly beneficial in critical care settings. Regulatory bodies are also playing a crucial role, with streamlined approval processes for innovative devices encouraging R&D investments and fostering market competition. The historical period from 2019 to 2024 laid the groundwork for this optimistic outlook, characterized by steady innovation and increasing clinical adoption. The study period, encompassing 2019 to 2033, will delve deep into these evolving dynamics, analyzing market segmentation by device type, application, and key regional influences. The estimated year of 2025 serves as a pivotal point, offering a snapshot of the current market valuation and setting the stage for future projections. The report will meticulously analyze the interplay of these factors, providing actionable insights for stakeholders aiming to capitalize on the burgeoning opportunities within the continuous flow apheresis device sector. The market's trajectory is not just about incremental improvements but about transformative shifts in patient care paradigms, driven by the precision and efficacy of these advanced apheresis systems. The increasing focus on personalized medicine further amplifies the need for devices capable of targeted therapeutic interventions, a key area of innovation within this market.
Several potent driving forces are propelling the continuous flow apheresis device market towards unprecedented growth. A paramount factor is the escalating global burden of chronic and rare diseases, including autoimmune disorders like Lupus Erythematosus and Rheumatoid Arthritis, as well as various hematological conditions and certain types of cancer. These conditions often necessitate regular and precise removal or modification of specific blood components, making therapeutic apheresis an indispensable treatment modality. The growing awareness and acceptance of apheresis as a viable therapeutic option among healthcare professionals and patients alike are further fueling demand. Technological advancements are another significant catalyst. Modern continuous flow apheresis devices are engineered for enhanced efficiency, faster processing times, and improved patient comfort through optimized extracorporeal circuits and sophisticated automation. This innovation not only leads to better clinical outcomes but also reduces the overall treatment duration and resource utilization, making apheresis more appealing to healthcare providers. The increasing complexity of treatments, particularly in oncology and immunology, where targeted removal of disease-causing cells or autoantibodies is crucial, is directly translating into a higher demand for sophisticated apheresis systems capable of precise component separation. The integration of artificial intelligence and machine learning in apheresis devices for real-time monitoring and adaptive treatment adjustments is also a nascent but rapidly growing trend that promises to enhance efficacy and safety, further driving market expansion.
Despite the robust growth trajectory, the continuous flow apheresis device market is not without its challenges and restraints. A significant hurdle is the high initial cost of these sophisticated devices. The substantial capital investment required for acquiring and maintaining continuous flow apheresis machines can be a deterrent for smaller healthcare facilities or those in resource-limited regions, thereby limiting market penetration. Furthermore, the specialized training and skilled personnel required to operate these complex devices pose another challenge. A lack of adequately trained apheresis technicians and clinicians can impede the widespread adoption and effective utilization of the technology. Reimbursement policies also play a critical role; inconsistent or inadequate reimbursement for apheresis procedures in various healthcare systems can impact the economic viability of offering these treatments, thus restraining market growth. The development of alternative therapeutic strategies, such as novel drug therapies that target the underlying causes of diseases previously managed by apheresis, could also present a competitive restraint. Regulatory complexities and the stringent approval processes for new devices and associated consumables, although designed to ensure safety and efficacy, can also lead to extended development timelines and increased costs for manufacturers. Lastly, patient comfort and potential side effects, while minimized with modern devices, can still be a concern for some individuals, influencing treatment adherence and overall demand for apheresis procedures.
The North America region is poised to dominate the continuous flow apheresis device market, driven by a confluence of factors including advanced healthcare infrastructure, high prevalence of target diseases, and significant R&D investments. The United States, in particular, stands out due to its well-established reimbursement landscape for advanced medical technologies and a strong emphasis on patient outcomes, facilitating the adoption of cutting-edge apheresis devices. The region's robust healthcare expenditure allows for the procurement of expensive, yet effective, continuous flow apheresis systems.
Within the North American context, the segment of Therapeutic Plasma Exchange (TPE) is expected to be a key driver of market growth and dominance. This is directly attributable to:
Furthermore, the Plasma Separators segment, which forms the technological backbone for TPE and other plasma-based apheresis procedures, will also exhibit substantial growth and dominance, particularly in conjunction with TPE applications. The demand for high-quality, reliable plasma separators is intrinsically linked to the demand for TPE itself. As the understanding of the therapeutic benefits of plasma manipulation expands, the need for sophisticated plasma separation technologies that can isolate specific plasma components or remove harmful ones will continue to rise. The presence of leading global manufacturers of plasma separators in or serving the North American market, such as Fresenius Medical Care and Terumo BCT, further solidifies the region's dominance in this segment. The synergy between advanced therapeutic applications like TPE and the underlying separation technology will ensure that North America and the TPE segment, supported by the robust Plasma Separators technology, remain at the forefront of the continuous flow apheresis device market throughout the forecast period. The market size for TPE procedures in North America alone is estimated to contribute significantly to the overall market value, potentially accounting for over $700 million by 2025 and growing to over $1,500 million by 2033.
The continuous flow apheresis device industry is experiencing robust growth fueled by several key catalysts. The increasing incidence and prevalence of autoimmune diseases, hematological disorders, and certain neurological conditions worldwide are creating a sustained demand for effective therapeutic interventions like apheresis. Furthermore, continuous advancements in technology are leading to more efficient, safer, and patient-friendly apheresis devices, driving their adoption in clinical settings. The expanding indications for apheresis, coupled with growing physician and patient awareness, are also key growth drivers.
This comprehensive report offers an in-depth analysis of the continuous flow apheresis device market, encompassing a detailed market segmentation by device type (e.g., Plasma Separators, Immunoadsorption Columns) and application (e.g., Therapeutic Plasma Exchange, Therapeutic Cytapheresis). It provides historical data from 2019-2024 and forecasts market trends from 2025-2033, with a base year of 2025. The report meticulously examines regional market dynamics, with a particular focus on dominating regions and segments. It delves into the driving forces, challenges, growth catalysts, and significant industry developments, offering strategic insights for stakeholders.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.2% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 12.2%.
Key companies in the market include Fresenius Medical Care (Germany), B. Braun Melsungen AG (Germany), HemaCare Corporation (US), Kaneka Corporation (Japan), Nikkiso Co., Ltd. (Japan), Terumo BCT, Inc (US), Haemonetics Corporation (US), Asahi Kasei Medical Co., Ltd. (Japan), Kawasumi Laboratories Inc. (Japan), Cerus Corporation (US), .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Continuous Flow Apheresis Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Continuous Flow Apheresis Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.