1. What is the projected Compound Annual Growth Rate (CAGR) of the Continuous Bioprocessing?
The projected CAGR is approximately 8.6%.
 Continuous Bioprocessing
Continuous BioprocessingContinuous Bioprocessing by Type (Bioreactor, Sterilizer, World Continuous Bioprocessing Production ), by Application (Pharmaceutical and Biotech Companies, Academic and Research Institutions, Others, World Continuous Bioprocessing Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Continuous Bioprocessing market is poised for significant expansion, projected to reach approximately $429 million by 2025. Fueled by a robust Compound Annual Growth Rate (CAGR) of 8.6% during the forecast period of 2025-2033, this burgeoning sector is a testament to the increasing adoption of advanced biomanufacturing techniques. The market's growth is primarily driven by the inherent advantages of continuous bioprocessing, including enhanced productivity, reduced capital expenditure, improved product quality, and minimized waste generation. Pharmaceutical and biotech companies are at the forefront of this adoption, seeking to optimize their drug development and manufacturing workflows for both traditional biologics and emerging therapies. Academic and research institutions also play a crucial role, contributing to the innovation and refinement of continuous bioprocessing technologies and methodologies. The development of sophisticated bioreactors and advanced sterilizer systems are key technological advancements enabling this shift towards more efficient and cost-effective biopharmaceutical production.
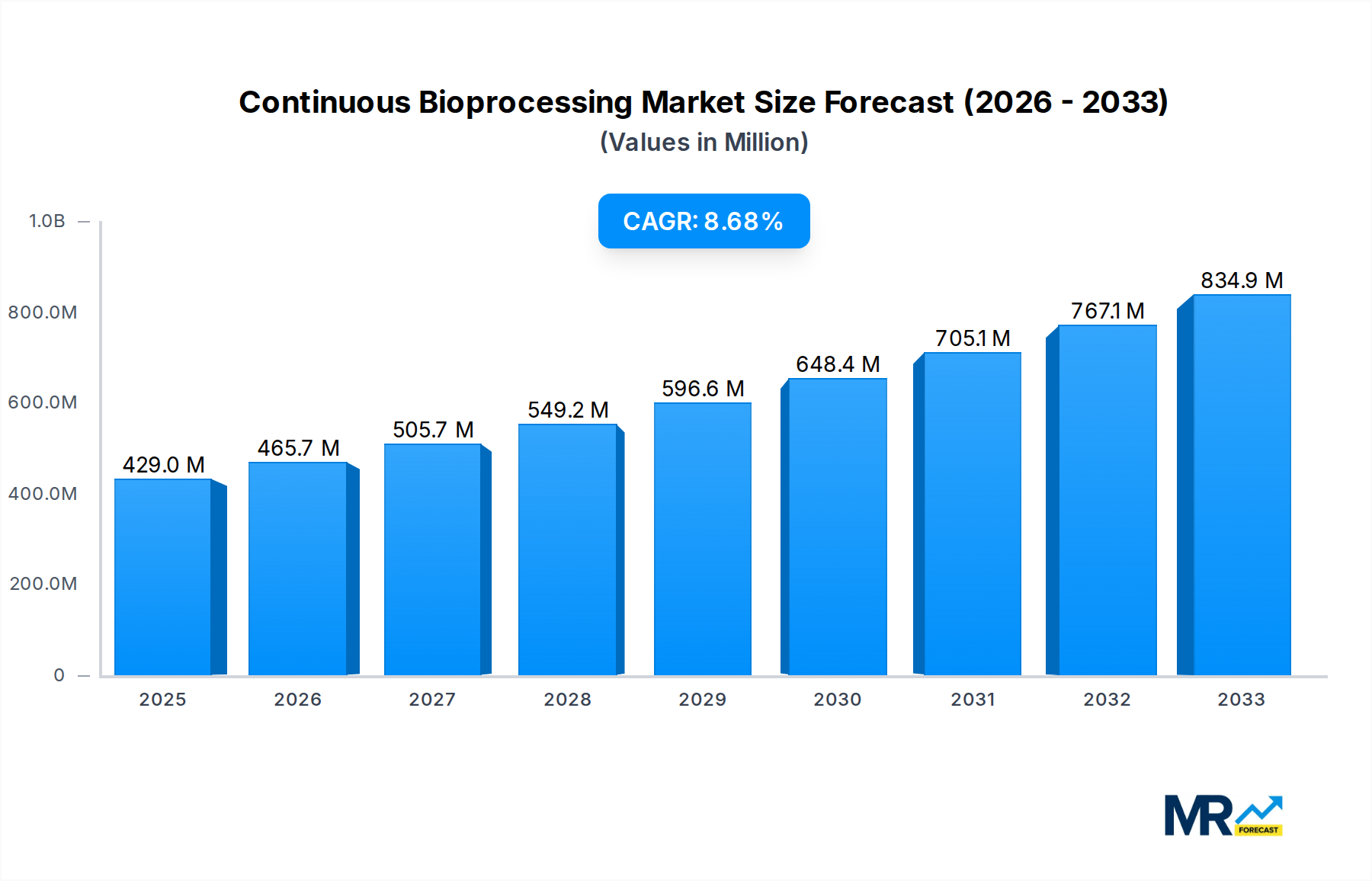

Looking ahead, the continuous bioprocessing landscape is expected to be shaped by several key trends. The increasing demand for personalized medicine and complex biologics necessitates flexible and scalable manufacturing solutions, which continuous processing elegantly provides. Furthermore, the growing emphasis on sustainability and green manufacturing principles aligns perfectly with the waste reduction and energy efficiency benefits offered by these integrated systems. While the market is exceptionally promising, certain restraints may influence its trajectory. High initial investment costs for sophisticated equipment and the need for specialized expertise for implementation and operation could pose challenges. Moreover, regulatory hurdles and the validation of novel continuous processes require diligent attention from industry stakeholders. Nevertheless, the overarching benefits and the relentless pursuit of innovation by leading companies like Thermo Fisher Scientific, Merck KGaA, and Sartorius AG are expected to overcome these obstacles, paving the way for a dynamic and rapidly evolving market.


Here's a report description on Continuous Bioprocessing, incorporating your specified elements:
This comprehensive report delves into the dynamic and rapidly evolving landscape of continuous bioprocessing, offering an in-depth analysis of market trends, driving forces, and future projections from 2019 to 2033. With a base year of 2025 and an estimated year also set for 2025, the report meticulously examines the historical performance (2019-2024) and provides robust forecasts for the period 2025-2033. The global continuous bioprocessing market, estimated to reach figures in the millions of USD, is poised for significant expansion, driven by technological advancements and the growing demand for efficient and cost-effective biopharmaceutical manufacturing.
The report provides granular insights into key market segments, including:
Furthermore, the report profiles leading companies such as 3M, Thermo Fisher Scientific, Merck KGaA, Sartorius AG, Eppendorf SE, Danaher, Bio-Rad Laboratories, Repligen Corporation, Bionet, COLDER PRODUCTS COMPANY, FUJIFILM, and WuXi Biologics, highlighting their strategic initiatives and contributions to the market.
The continuous bioprocessing market is experiencing a paradigm shift, moving away from traditional batch manufacturing towards more integrated and streamlined production methods. Key market insights reveal a substantial upswing in adoption driven by the inherent advantages of continuous systems, including enhanced product quality, reduced footprint, and significant cost efficiencies. Throughout the historical period (2019-2024), we observed a nascent but growing interest, primarily within academic research and early-stage biotech firms exploring the potential. The base year (2025) marks a critical inflection point where larger pharmaceutical entities are increasingly investing in and scaling up continuous bioprocessing technologies, anticipating substantial improvements in their manufacturing pipelines. The forecast period (2025-2033) is projected to witness exponential growth, with market penetration accelerating significantly. This surge is fueled by regulatory bodies showing increased receptiveness to continuous manufacturing, recognizing its potential to deliver safer, more consistent, and higher-quality biopharmaceuticals to patients. The trend towards intensified bioprocessing, where higher productivities are achieved in smaller volumes, is a dominant theme. This is particularly relevant for the development and manufacturing of complex biologics, including monoclonal antibodies, recombinant proteins, and advanced cell and gene therapies, where traditional batch methods can be time-consuming and resource-intensive. The integration of advanced process analytical technology (PAT) and sophisticated control systems is another key trend, enabling real-time monitoring and adjustment of critical process parameters, thereby minimizing variability and maximizing yield. The development of novel bioreactor designs, such as perfusion bioreactors and single-use systems, are also playing a pivotal role in facilitating continuous operations. The market is moving towards a holistic integration of upstream and downstream processing, creating truly end-to-end continuous workflows. This integrated approach not only optimizes efficiency but also significantly reduces the risk of contamination and batch failures. The increasing demand for personalized medicine and orphan drugs, which often require smaller, more flexible manufacturing capabilities, further bolsters the appeal of continuous bioprocessing. The ability to rapidly switch between product campaigns without extensive revalidation further solidifies its strategic importance for biopharmaceutical companies aiming to address diverse therapeutic needs effectively.
Several potent forces are synergistically driving the accelerated adoption of continuous bioprocessing technologies. Foremost among these is the relentless pursuit of cost reduction and increased operational efficiency within the biopharmaceutical industry. Continuous manufacturing offers a distinct advantage over traditional batch processes by minimizing downtime between batches, reducing facility footprint requirements, and lowering raw material consumption. This translates directly into a more competitive cost of goods sold, particularly crucial for the production of biologics which are inherently expensive to manufacture. Furthermore, the escalating demand for complex biologics, including biosimilars and novel therapies for chronic and rare diseases, necessitates more agile and scalable manufacturing solutions. Continuous bioprocessing provides the flexibility to ramp up production quickly and efficiently, adapting to fluctuating market demands. Regulatory agencies worldwide are also increasingly recognizing the benefits of continuous manufacturing, offering more streamlined approval pathways for products manufactured using these advanced techniques. This regulatory support, coupled with the inherent quality advantages of continuous processes – such as improved product consistency and reduced batch-to-batch variability – acts as a significant catalyst. The ongoing advancements in sensor technology, automation, and process analytical technology (PAT) are enabling unprecedented levels of process control and monitoring in real-time, further enhancing the reliability and robustness of continuous bioprocessing. The integration of single-use technologies also plays a crucial role, offering greater flexibility, reduced cleaning validation, and faster changeover times between product campaigns, all of which are vital for efficient continuous operations.
Despite its undeniable promise, the widespread adoption of continuous bioprocessing is not without its hurdles. One of the primary challenges is the significant initial capital investment required for implementing continuous manufacturing platforms. The integration of novel equipment, advanced automation, and sophisticated control systems can represent a substantial financial outlay for companies, particularly small and medium-sized enterprises. Furthermore, the transition from well-established batch manufacturing protocols to continuous processes necessitates considerable re-training of personnel and a fundamental shift in manufacturing paradigms. Developing expertise in operating and maintaining complex continuous systems can be a time-consuming and resource-intensive undertaking. Another significant restraint lies in the regulatory landscape, which, while evolving, still presents complexities in validating and approving continuous manufacturing processes. Demonstrating process robustness and product consistency over extended operational periods requires rigorous validation studies and a deep understanding of process variability. The susceptibility of continuous processes to contamination, though mitigated by advanced sterile techniques, remains a concern. Any contamination event can halt an entire continuous run, potentially leading to significant product loss. The development of suitable downstream processing technologies that can efficiently handle the continuous output from upstream bioreactors is also an ongoing area of research and development. Finally, the perceived complexity and the need for specialized technical expertise can create a barrier to entry for some organizations, slowing down the overall market penetration.
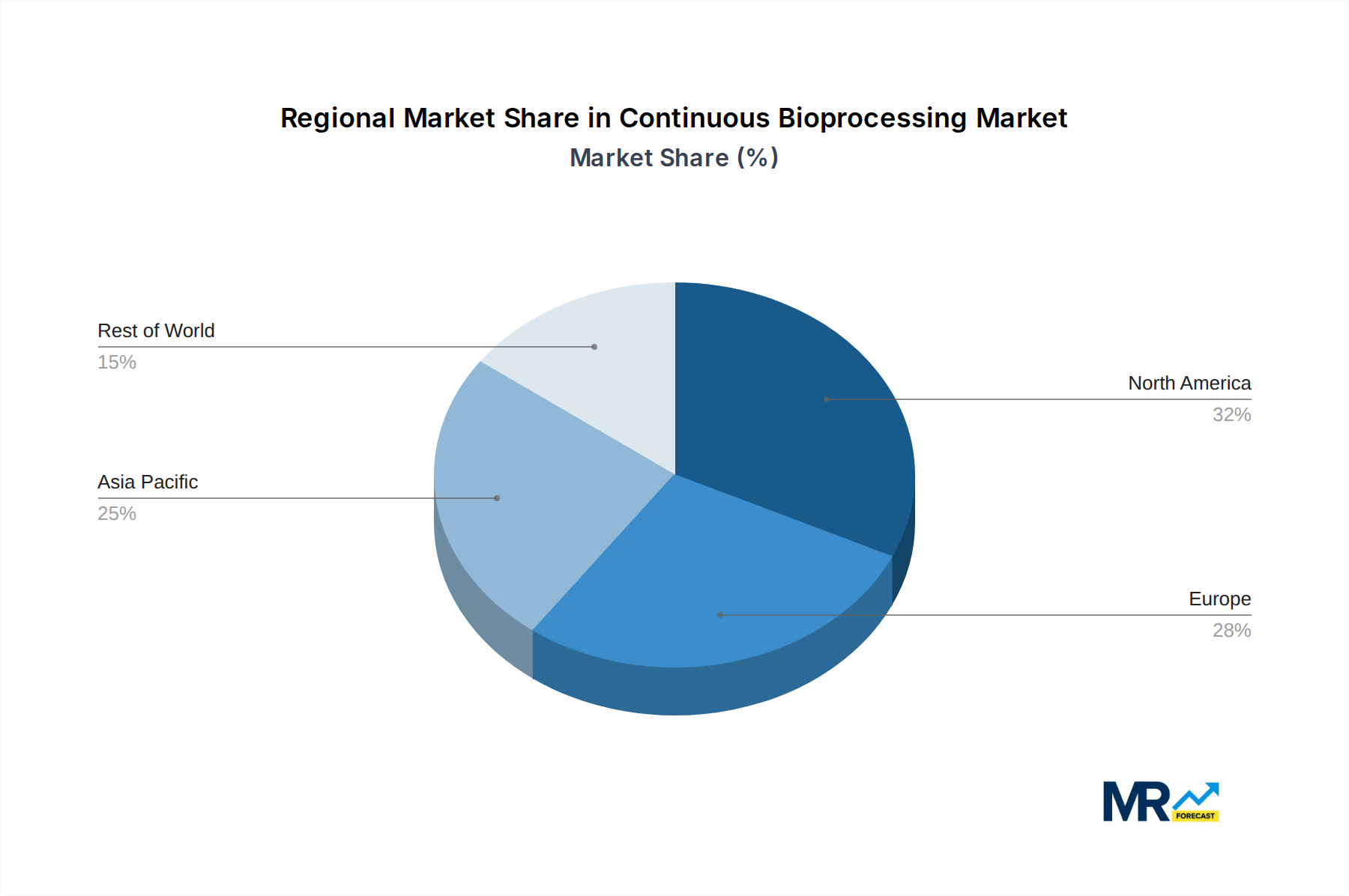
The Pharmaceutical and Biotech Companies segment, particularly within the North America region, is projected to dominate the global continuous bioprocessing market throughout the study period (2019-2033).
Dominance of Pharmaceutical and Biotech Companies:
North America Leading the Charge:
The continuous bioprocessing industry is propelled by several key growth catalysts. The increasing demand for biologics and biosimilars, driven by an aging global population and the rise of chronic diseases, necessitates more efficient and cost-effective manufacturing. Continuous bioprocessing offers a compelling solution by reducing production costs and increasing throughput. Technological advancements in automation, process analytical technology (PAT), and single-use systems are further enabling more robust and reliable continuous workflows. Regulatory support and the increasing acceptance of continuous manufacturing by agencies like the FDA are also significantly de-risking investments and encouraging adoption. Furthermore, the development of more sophisticated bioreactor designs and integrated downstream processing solutions is making continuous bioprocessing more accessible and scalable.
This report provides an exhaustive analysis of the global continuous bioprocessing market, covering historical trends from 2019 to 2024 and projecting future growth until 2033. It offers detailed insights into market dynamics, including key drivers, challenges, and opportunities. The report meticulously examines major segments such as bioreactors, sterilizers, and application areas like pharmaceutical and biotech companies, academic institutions, and others. With a base year of 2025 and an estimated market value in the millions of USD, the report presents a robust forecast for the coming years. Leading players and significant market developments are thoroughly profiled, providing a comprehensive understanding of the competitive landscape and future trajectory of this transformative field in biopharmaceutical manufacturing.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 8.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 8.6%.
Key companies in the market include 3M, Thermo Fisher Scientific, Merck KGaA, Sartorius AG, Eppendorf SE, Danaher, Bio-Rad Laboratories, Repligen Corporation, Bionet, COLDER PRODUCTS COMPANY, FUJIFILM, WuXi Biologics.
The market segments include Type, Application.
The market size is estimated to be USD 429 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Continuous Bioprocessing," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Continuous Bioprocessing, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.