1. What is the projected Compound Annual Growth Rate (CAGR) of the Coagulation Efficiency Testers?
The projected CAGR is approximately 5.6%.
 Coagulation Efficiency Testers
Coagulation Efficiency TestersCoagulation Efficiency Testers by Type (Standalone Analysers, Table Top Analysers, Portable Analysers), by Application (Hospitals, Clinical Laboratories, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Coagulation Efficiency Testers market is poised for substantial growth, projected to reach a valuation of $2,940.1 million in 2025, with a Compound Annual Growth Rate (CAGR) of 5.6% expected to propel it through 2033. This robust expansion is primarily driven by the increasing prevalence of thrombotic and hemorrhagic disorders, a growing demand for accurate and rapid diagnostic tools in healthcare settings, and significant advancements in analyzer technology. The rising number of diagnostic tests performed globally, coupled with an aging population susceptible to coagulation-related conditions, further fuels market momentum. The trend towards decentralized testing and the integration of automation and AI in laboratory diagnostics are also key factors contributing to market penetration and the development of more sophisticated and user-friendly coagulation testing solutions.
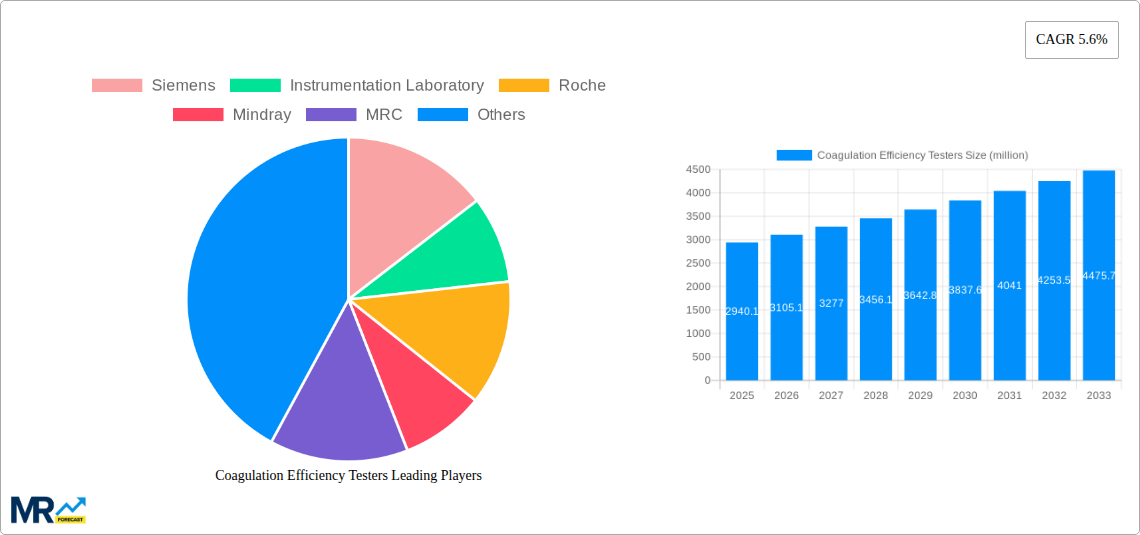

The market segmentation reveals a dynamic landscape. Standalone analyzers currently lead in revenue, catering to specialized needs, while table-top analyzers are gaining traction due to their versatility and cost-effectiveness for a broader range of clinical applications. Portable analyzers are emerging as a critical segment, facilitating point-of-care testing and remote patient monitoring, thereby enhancing accessibility and improving patient outcomes. Hospitals and clinical laboratories represent the dominant application segments, driven by high test volumes and the critical role of coagulation testing in patient management. The market is characterized by intense competition among established players like Siemens, Roche, and Abbott Laboratories, alongside innovative companies such as Mindray and Sysmex, all striving to capture market share through product innovation, strategic partnerships, and geographical expansion. Emerging economies, particularly in the Asia Pacific region, are presenting significant growth opportunities due to improving healthcare infrastructure and increasing awareness of advanced diagnostic techniques.
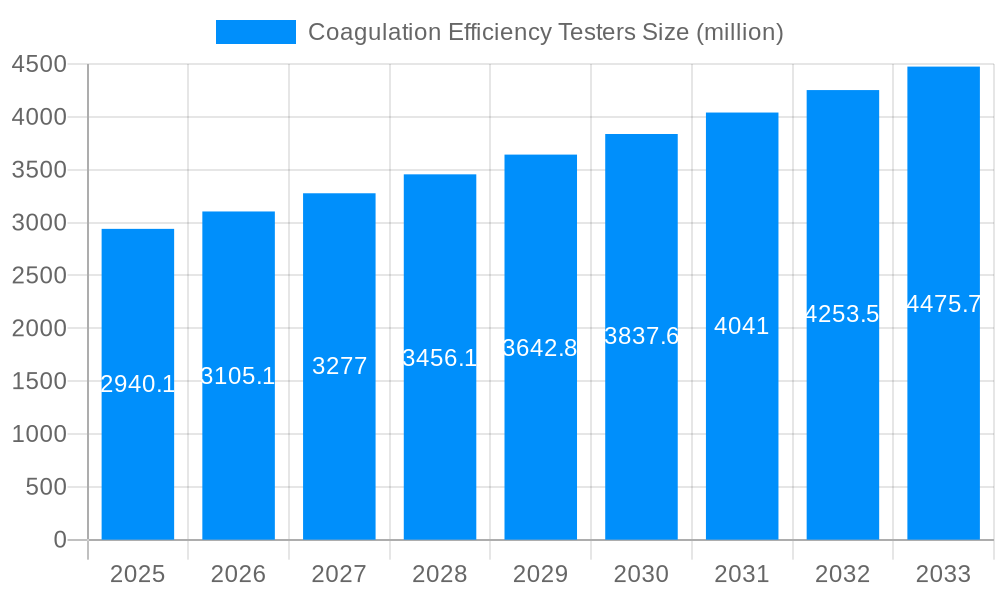

This report offers a comprehensive analysis of the global Coagulation Efficiency Testers market, spanning the historical period of 2019-2024 and projecting significant growth through 2033. The study period for this analysis is from 2019 to 2033, with the base year and estimated year both set to 2025, and the forecast period extending from 2025 to 2033. The market is poised for substantial expansion, with an estimated valuation in the tens of millions of dollars in the base year, projected to reach several hundred million dollars by the end of the forecast period. This growth is driven by a confluence of factors, including increasing prevalence of coagulation disorders, advancements in diagnostic technologies, and rising healthcare expenditure worldwide.
The report delves into the intricate landscape of coagulation testing, examining the various types of analyzers, their applications across diverse healthcare settings, and the evolving industry developments shaping the market's trajectory. It provides granular insights into market segmentation, enabling stakeholders to identify lucrative opportunities and understand the competitive dynamics at play.
XXX report highlights a significant upward trend in the global Coagulation Efficiency Testers market, driven by a confluence of technological advancements and increasing demand for accurate and rapid diagnostic solutions. The market, valued in the tens of millions of dollars in the base year of 2025, is projected to witness a compound annual growth rate (CAGR) that will propel its valuation into the hundreds of millions by 2033. A key trend is the increasing adoption of automated and semi-automated analyzers, which offer enhanced throughput, reduced manual error, and improved precision compared to older, manual methods. This shift is particularly pronounced in hospitals and large clinical laboratories, where the volume of tests performed necessitates efficient and reliable instrumentation. Furthermore, the development of point-of-care (POC) coagulation testing devices is another significant trend. These portable and user-friendly analyzers are enabling faster diagnosis and treatment decisions at the patient's bedside, particularly crucial in emergency settings, intensive care units, and for patients on anticoagulant therapy. The integration of advanced software and data management capabilities within these testers is also a growing trend, facilitating seamless integration with laboratory information systems (LIS) and electronic health records (EHRs), thereby improving workflow efficiency and data traceability. The research and development pipeline for novel assay technologies and reagents also continues to fuel market expansion, promising even greater diagnostic accuracy and the ability to detect a wider spectrum of coagulation abnormalities. The growing awareness and diagnosis of coagulation disorders, coupled with the aging global population, are further augmenting the demand for these essential diagnostic tools.
The Coagulation Efficiency Testers market is experiencing robust growth, propelled by several powerful driving forces. The escalating prevalence of thrombotic and bleeding disorders globally stands as a primary catalyst. Conditions such as deep vein thrombosis (DVT), pulmonary embolism (PE), and hemophilia necessitate accurate and timely coagulation testing for diagnosis, monitoring of treatment, and management of patient care. This increasing disease burden directly translates into a higher demand for sophisticated coagulation analyzers. Furthermore, the rapid advancements in diagnostic technology are playing a pivotal role. Manufacturers are continuously innovating, introducing analyzers with higher throughput, improved accuracy, and expanded testing menus, capable of performing a wider range of coagulation tests. This technological evolution makes these testers more attractive to healthcare providers seeking to enhance their diagnostic capabilities and clinical outcomes. The growing emphasis on personalized medicine and precision therapeutics also fuels the demand. As treatment protocols become more tailored to individual patient needs, particularly for anticoagulant therapies, precise monitoring of coagulation parameters becomes essential. This drives the adoption of advanced testing solutions that can provide reliable and reproducible results. Moreover, the increasing global healthcare expenditure, coupled with rising investments in healthcare infrastructure, particularly in emerging economies, is creating a fertile ground for market expansion. This allows for greater accessibility to advanced diagnostic equipment, including coagulation efficiency testers, across a wider network of healthcare facilities.
Despite the promising growth trajectory, the Coagulation Efficiency Testers market is not without its challenges and restraints. One significant hurdle is the high cost associated with acquiring advanced coagulation analyzers. For smaller laboratories or facilities in resource-limited regions, the initial capital investment can be a substantial deterrent, limiting market penetration. This cost factor can lead to a disparity in access to cutting-edge diagnostic tools. Another challenge lies in the stringent regulatory requirements governing medical devices. The development, manufacturing, and marketing of coagulation efficiency testers are subject to rigorous approval processes by regulatory bodies such as the FDA and EMA. Obtaining these approvals can be time-consuming and expensive, potentially slowing down the introduction of new products into the market. Furthermore, the availability of skilled personnel to operate and maintain these sophisticated analyzers can be a limiting factor, especially in developing countries. Proper training and expertise are crucial for ensuring accurate test results and maximizing the efficiency of the equipment. The market also faces competition from established players and the constant pressure to innovate, which requires continuous investment in research and development. Price sensitivity among some market segments can also pose a restraint, as healthcare providers seek cost-effective solutions without compromising on quality. Finally, the potential for reimbursement challenges in certain healthcare systems can impact the adoption rates of new and more expensive testing methodologies.
The Standalone Analyzers segment, within the Hospitals application, is projected to dominate the global Coagulation Efficiency Testers market. This dominance is driven by several converging factors that underscore the critical role of these analyzers in modern healthcare.
Hospitals as Primary Consumers: Hospitals, across all tiers, represent the largest and most consistent consumers of coagulation efficiency testers. The sheer volume of patients requiring coagulation diagnostics, from routine pre-operative screenings to the management of critical care patients with complex clotting disorders, necessitates robust and high-throughput testing capabilities. Standalone analyzers, designed for dedicated laboratory use, offer the precision, reliability, and comprehensive test menus required to meet these demanding clinical needs. Their integration into the hospital's central laboratory infrastructure ensures standardized testing protocols and efficient workflow management.
Standalone Analyzers: The Backbone of Clinical Diagnostics: Standalone analyzers are characterized by their comprehensive functionalities, advanced automation, and capacity for a broad spectrum of coagulation tests, including PT, aPTT, fibrinogen, D-dimer, and antithrombin assays, among others. These units are typically benchtop or floor-standing models, equipped with sophisticated optical or mechanical detection systems, automated sample handling, and integrated software for data analysis and reporting. Their ability to perform a wide array of tests with high accuracy and minimal manual intervention makes them indispensable for accurate diagnosis and effective patient management in hospital settings. The drive for improved turnaround times and reduced diagnostic errors further solidifies the position of standalone analyzers.
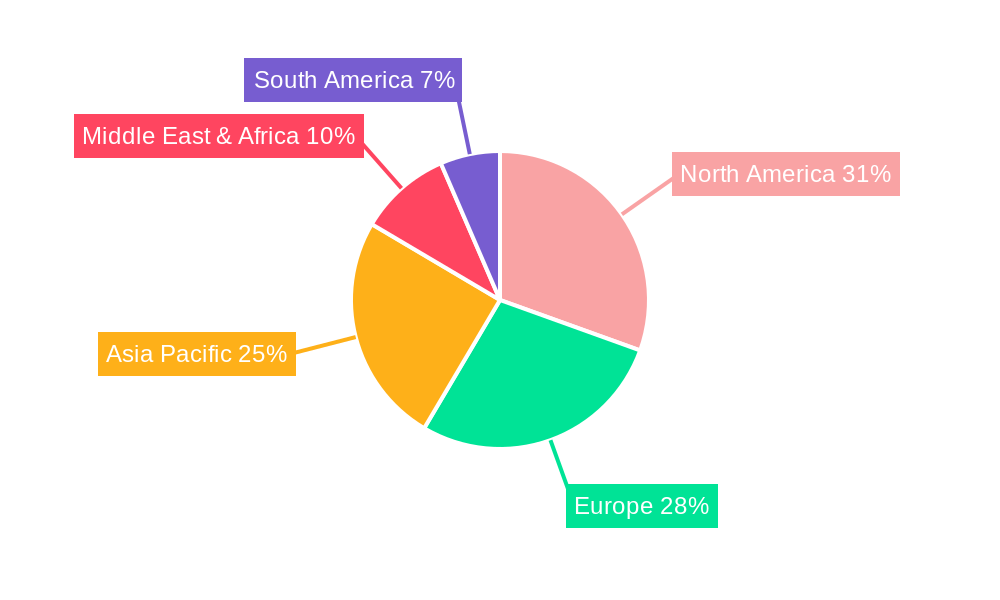
Geographic Dominance – North America and Europe: Within the hospital segment, North America and Europe are expected to continue their dominance in the utilization of standalone coagulation efficiency testers. These regions boast highly developed healthcare infrastructures, significant investments in medical technology, and a high prevalence of coagulation disorders, including an aging population susceptible to thrombotic events. Furthermore, the presence of leading research institutions and a strong emphasis on evidence-based medicine contribute to the early adoption of advanced diagnostic technologies. Stringent regulatory frameworks in these regions also necessitate the use of high-quality, validated diagnostic equipment, favoring sophisticated standalone analyzers. The high per capita healthcare expenditure and robust reimbursement policies further support the acquisition of these advanced systems by hospitals.
Emerging Markets and Future Growth: While North America and Europe lead, significant growth potential also exists in the Asia-Pacific region, driven by increasing healthcare expenditure, expanding hospital networks, and a growing awareness of coagulation disorders. As these economies develop and healthcare infrastructure improves, the demand for standalone analyzers in hospitals will undoubtedly escalate. The increasing adoption of advanced medical devices by both public and private healthcare providers in countries like China, India, and Southeast Asian nations will contribute significantly to the global market expansion in the coming years.
The Coagulation Efficiency Testers industry is experiencing several potent growth catalysts. The increasing incidence of cardiovascular diseases and the growing use of anticoagulants for their management are directly fueling the demand for accurate coagulation monitoring. Furthermore, advancements in assay technologies, leading to improved sensitivity and specificity of tests, are enhancing diagnostic capabilities. The global expansion of healthcare infrastructure, particularly in emerging economies, coupled with rising healthcare expenditure, is making these essential diagnostic tools more accessible. The growing emphasis on personalized medicine and point-of-care diagnostics is also driving innovation and adoption of more user-friendly and rapid testing solutions.
This comprehensive report offers an in-depth exploration of the Coagulation Efficiency Testers market, providing actionable insights for stakeholders. It delves into market dynamics, identifying key trends such as the shift towards automation, the growing demand for point-of-care solutions, and the impact of technological advancements. The report meticulously analyzes the driving forces behind market growth, including the rising prevalence of coagulation disorders and increasing healthcare investments. It also sheds light on the challenges and restraints, such as high acquisition costs and stringent regulatory hurdles. Furthermore, the report provides a detailed regional and segment analysis, highlighting dominant regions and key application areas. It also profiles leading industry players and significant recent developments, offering a holistic view of the competitive landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.6%.
Key companies in the market include Siemens, Instrumentation Laboratory, Roche, Mindray, MRC, Helena Laboratories, Thermo Fisher Scientific, ERBA Diagnostics Mannheim, Rayto Life and Analytical Sciences, Nihon Kohden Corporation, Beckman Coulter Inc. (Danaher Corporation), Sysmex Corporation, Abbott Laboratories, HORIBA Medical, DIRUI Industrial, Seleo, Zircon Biotech, Beijing Succeeder Technology, .
The market segments include Type, Application.
The market size is estimated to be USD 2940.1 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Coagulation Efficiency Testers," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Coagulation Efficiency Testers, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.