1. What is the projected Compound Annual Growth Rate (CAGR) of the Blow Fill Seal Solutions?
The projected CAGR is approximately 11.11%.
 Blow Fill Seal Solutions
Blow Fill Seal SolutionsBlow Fill Seal Solutions by Type (Polypropylene, High-density Ethylene, Low-density Ethylene), by Application (Medical Institutions, Research Institutes, Pharmaceutical Company, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The global Blow-Fill-Seal (BFS) solutions market is poised for significant expansion, projected to reach an estimated USD 3,150 million by 2025, with a robust Compound Annual Growth Rate (CAGR) of 5% anticipated through 2033. This growth is fundamentally driven by the increasing demand for sterile and unit-dose packaging solutions across the pharmaceutical, medical, and biotechnology sectors. The inherent benefits of BFS technology, including its ability to minimize contamination risk, ensure product integrity, and offer cost-effectiveness for high-volume production, are key accelerators. Furthermore, advancements in BFS machinery, offering greater automation, speed, and versatility for a wider range of product viscosities and container formats, are continuously enhancing its adoption. The market's trajectory is further supported by the growing emphasis on single-use medical devices and the escalating need for safe and reliable drug delivery systems, especially for parenteral and ophthalmic preparations.

The BFS market is characterized by distinct segmentation, with Polypropylene (PP) leading as the preferred material due to its excellent chemical resistance, sterilizability, and cost-efficiency, followed by High-Density Ethylene (HDE) and Low-Density Ethylene (LDE). In terms of application, Medical Institutions and Pharmaceutical Companies represent the largest share, driven by their continuous need for sterile vials, ampoules, and syringes. Research Institutes also contribute to demand for specialized packaging. Key restraints include the initial capital investment for BFS machinery and stringent regulatory compliance, which can pose challenges for smaller players. However, emerging economies in the Asia Pacific region, fueled by burgeoning healthcare infrastructure and increasing disposable incomes, present significant untapped growth opportunities. Innovations in BFS technology, such as the integration of inline inspection systems and the development of sustainable packaging materials, are expected to shape future market dynamics.
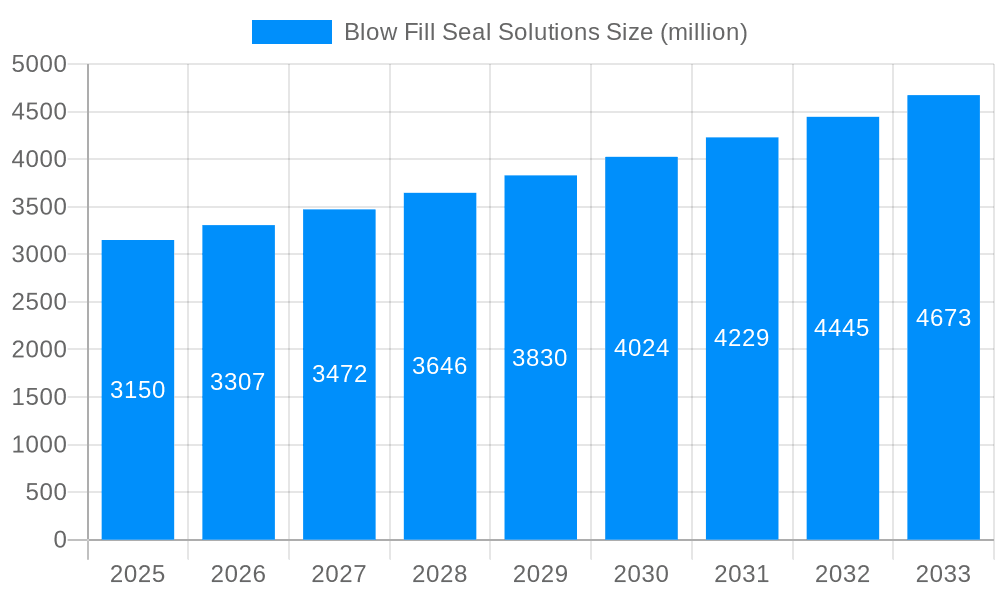

The Blow Fill Seal (BFS) solutions market is poised for significant expansion, driven by an increasing demand for sterile, single-dose packaging across diverse sectors. From a robust historical performance during the 2019-2024 period, the market is projected to witness sustained growth, reaching an estimated value in the millions of units by the Base Year of 2025 and continuing its upward trajectory through the Forecast Period of 2025-2033. This growth is intrinsically linked to the pharmaceutical industry's relentless pursuit of enhanced drug delivery systems and improved patient safety. BFS technology offers a compelling solution by manufacturing, filling, and sealing containers in a single, continuous, automated process, thereby minimizing contamination risks and ensuring product integrity. The study period, spanning from 2019 to 2033, encompasses a dynamic evolution of this technology, characterized by advancements in materials, automation, and regulatory compliance. Key market insights highlight a pronounced shift towards smaller volume BFS containers, catering to specialized pharmaceutical applications and the growing biologics sector. Furthermore, the adoption of sustainable packaging materials, such as advanced grades of Polypropylene and Ethylene-based polymers, is gaining momentum, reflecting a broader industry commitment to environmental responsibility. The inherent sterility assurance of BFS systems, coupled with their cost-effectiveness in high-volume production, positions them as a preferred choice for a wide array of liquid, semi-liquid, and even some powder-based pharmaceuticals. Innovations in container designs, including tamper-evident features and user-friendly dispensing mechanisms, are also contributing to market vibrancy. The growing prevalence of chronic diseases and the subsequent rise in the demand for injectable and ophthalmic drugs are significant tailwinds for the BFS market. As healthcare infrastructure expands globally, particularly in emerging economies, the adoption of advanced packaging solutions like BFS is expected to accelerate. The market's intricate interplay between technological innovation, regulatory landscapes, and evolving consumer preferences will continue to shape its trajectory, with a strong emphasis on sterile, reliable, and patient-centric packaging solutions. The overall market sentiment indicates a positive outlook, with ongoing investments in research and development expected to unlock new applications and further solidify BFS's position as a cornerstone of modern pharmaceutical packaging. The projected market size, measured in millions of units, underscores the substantial scale and importance of this segment within the broader healthcare ecosystem.
The burgeoning demand for sterile and aseptic packaging solutions across the pharmaceutical and healthcare industries stands as a primary engine for the growth of the Blow Fill Seal (BFS) market. The inherent ability of BFS technology to produce, fill, and seal containers in a single, integrated process, significantly mitigates the risk of microbial contamination. This is paramount in the manufacturing of injectables, ophthalmic solutions, and other sensitive pharmaceutical products where sterility is non-negotiable. Furthermore, the increasing global burden of chronic diseases necessitates a continuous supply of medications, many of which are administered via liquid or semi-liquid dosage forms that benefit from BFS packaging. The expanding biologics sector, characterized by complex and sensitive therapeutic agents, also presents a significant growth avenue for BFS solutions due to their ability to maintain product integrity. Regulatory bodies worldwide are increasingly emphasizing product safety and traceability, which aligns perfectly with the controlled and automated nature of BFS processes. The drive towards enhanced patient convenience and adherence is also playing a crucial role. BFS containers can be designed with user-friendly features, such as pre-scored tips for easy opening and accurate dosing, thereby improving the patient experience. Moreover, the cost-effectiveness of BFS, particularly in high-volume production runs, makes it an attractive proposition for pharmaceutical manufacturers seeking to optimize their operational expenditures without compromising on quality or safety. The continuous innovation in BFS machinery and materials, enabling the handling of a wider range of formulations and container types, further fuels its adoption.
Despite its robust growth prospects, the Blow Fill Seal (BFS) market faces certain challenges and restraints that could temper its expansion. The initial capital investment required for sophisticated BFS machinery can be substantial, posing a barrier to entry for smaller manufacturers or those with limited financial resources. While the long-term cost-effectiveness is undeniable, the upfront expenditure can be a significant consideration. Furthermore, the highly specialized nature of BFS technology necessitates a skilled workforce for operation, maintenance, and validation. Sourcing and retaining adequately trained personnel can be a hurdle in certain regions, potentially impacting production efficiency. The stringent regulatory requirements governing pharmaceutical packaging, including BFS systems, add another layer of complexity. Manufacturers must adhere to rigorous quality control standards and undergo extensive validation processes, which can be time-consuming and costly. While BFS offers inherent sterility, the prevention of particle generation during the manufacturing process remains a critical concern, requiring meticulous control over the manufacturing environment. Additionally, the flexibility of BFS in terms of container material and design, while improving, can still be perceived as somewhat limited compared to other packaging technologies for highly specific applications or novel materials. Market competition from alternative sterile packaging methods, such as pre-filled syringes and vials, also presents a competitive pressure. The development of new drug formulations or delivery systems that might not be optimally suited for existing BFS capabilities could also pose a restraint. Finally, fluctuations in raw material prices, particularly for polymers like Polypropylene and Ethylene, can impact production costs and, consequently, the overall pricing of BFS solutions.
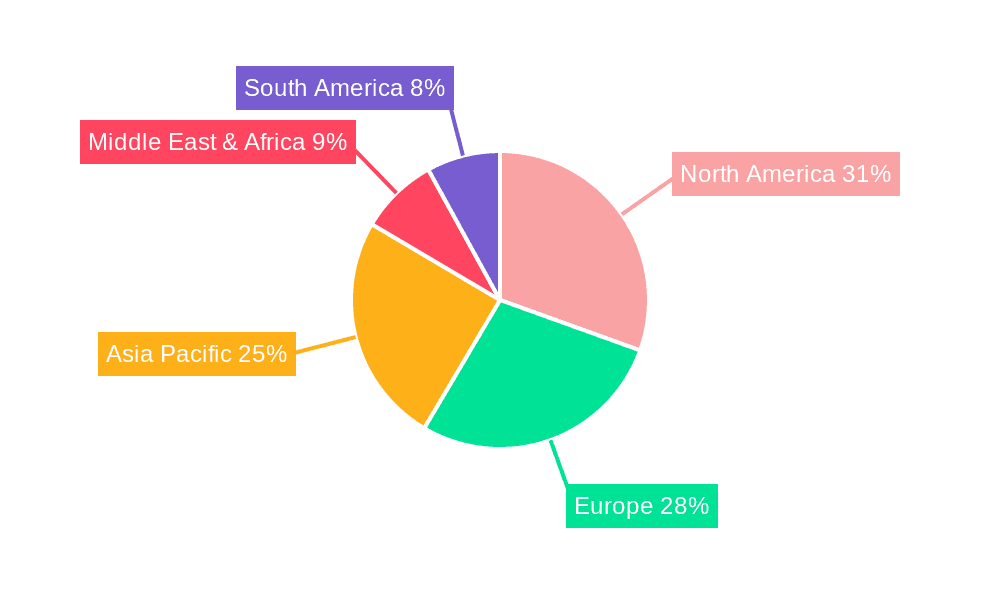
The Blow Fill Seal (BFS) solutions market is characterized by a significant dominance stemming from Pharmaceutical Companies as a key application segment, coupled with a strong regional presence in North America and Europe.
Dominant Segment: Pharmaceutical Company (Application)
Dominant Regions: North America and Europe
While other regions like Asia-Pacific are showing significant growth potential due to expanding healthcare access and manufacturing capabilities, North America and Europe are expected to maintain their leadership positions in the Blow Fill Seal solutions market due to the sustained demand from pharmaceutical companies and the advanced healthcare ecosystems present.
The Blow Fill Seal (BFS) industry is experiencing significant growth catalysts, primarily driven by the escalating demand for sterile and single-dose pharmaceutical packaging. The expanding biologics market, with its sensitive therapeutic agents requiring stringent containment, is a major propellant. Furthermore, the increasing global prevalence of chronic diseases necessitates a continuous supply of injectable and ophthalmic medications, which are optimally packaged using BFS technology. Advancements in BFS machinery, including higher speeds, increased automation, and the ability to handle a wider range of container sizes and shapes, are enhancing its appeal. The growing emphasis on patient convenience and adherence, with BFS containers offering user-friendly designs, also fuels demand. Finally, the inherent cost-effectiveness and sterility assurance of BFS solutions make them an attractive option for pharmaceutical companies seeking to optimize production and ensure product safety.
This comprehensive report delves into the intricate landscape of the Blow Fill Seal (BFS) solutions market, offering invaluable insights for stakeholders. It meticulously analyzes market trends, identifying key growth drivers such as the burgeoning pharmaceutical sector and the increasing demand for sterile packaging. The report also addresses significant challenges and restraints, including high initial investment costs and the need for specialized expertise. Detailed regional and segment analyses highlight the dominance of key markets and applications, such as Pharmaceutical Companies in North America and Europe. Furthermore, it outlines crucial growth catalysts and provides a detailed overview of leading industry players. The report's extensive coverage ensures stakeholders are equipped with the knowledge to navigate this dynamic market, make informed strategic decisions, and capitalize on future opportunities, projecting market values in the millions of units.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 11.11% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 11.11%.
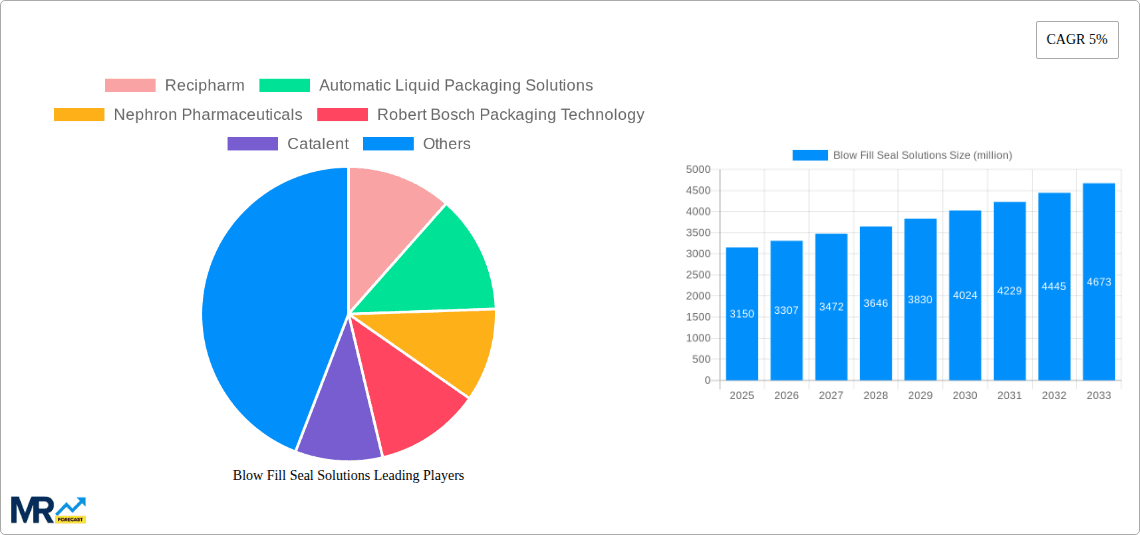
Key companies in the market include Recipharm, Automatic Liquid Packaging Solutions, Nephron Pharmaceuticals, Robert Bosch Packaging Technology, Catalent, Horizon Pharmaceuticals, Unither, ApiJect Systems Corporation, LESVI, Asept Pak, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Blow Fill Seal Solutions," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Blow Fill Seal Solutions, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.