1. What is the projected Compound Annual Growth Rate (CAGR) of the Bioresorbable Ophthalmic Drug Delivery System?
The projected CAGR is approximately 5%.
 Bioresorbable Ophthalmic Drug Delivery System
Bioresorbable Ophthalmic Drug Delivery SystemBioresorbable Ophthalmic Drug Delivery System by Type (Anterior Drug Delivery Methods, Posterior Drug Delivery Methods, World Bioresorbable Ophthalmic Drug Delivery System Production ), by Application (Hospitals, Clinics, Others, World Bioresorbable Ophthalmic Drug Delivery System Production ), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
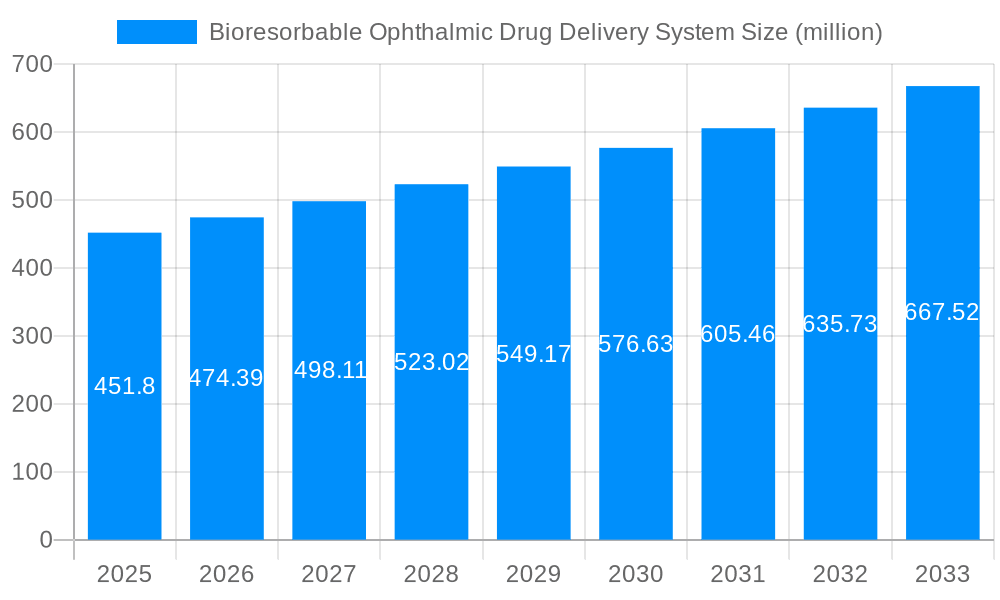
The global Bioresorbable Ophthalmic Drug Delivery System market is projected for robust expansion, reaching an estimated USD 451.8 million by 2025 and exhibiting a Compound Annual Growth Rate (CAGR) of 5% throughout the forecast period (2025-2033). This growth is propelled by a confluence of factors, including the increasing prevalence of chronic eye diseases such as glaucoma, diabetic retinopathy, and age-related macular degeneration (AMD), which necessitate sustained and targeted drug delivery. The inherent advantages of bioresorbable systems, such as improved patient compliance due to reduced dosing frequency, enhanced therapeutic efficacy through precise drug release kinetics, and minimized side effects compared to conventional methods like frequent injections or eye drops, are significant market drivers. Furthermore, advancements in material science leading to novel biodegradable polymers and micro/nanoparticle-based delivery platforms are fueling innovation and broadening the application spectrum. The market's segmentation into anterior and posterior drug delivery methods highlights the diverse therapeutic needs being addressed, with a growing emphasis on innovative solutions for posterior segment diseases.
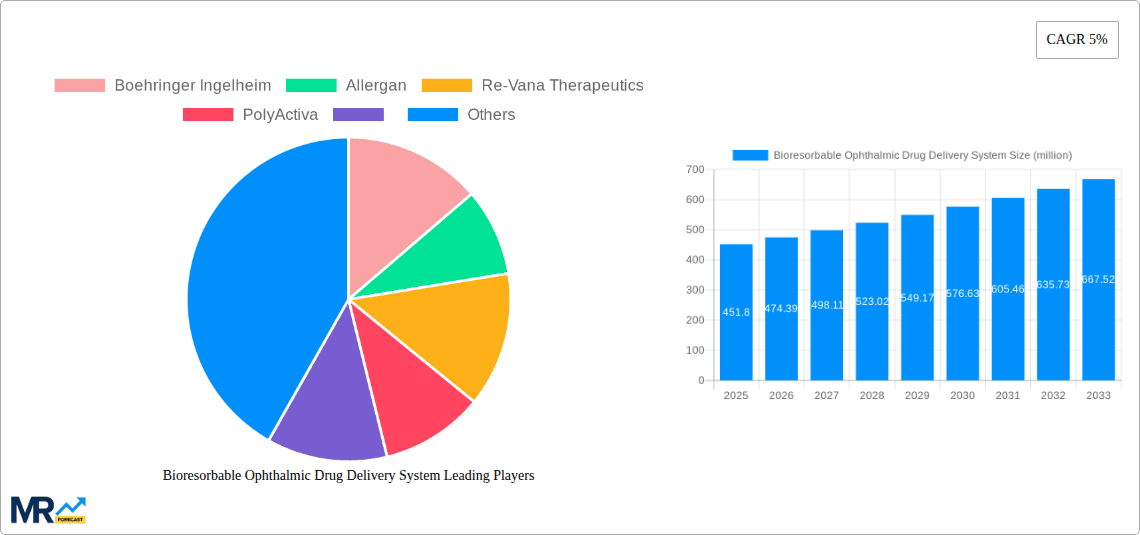

The market landscape is characterized by increasing research and development activities, strategic collaborations, and acquisitions by key players like Boehringer Ingelheim, Allergan, Re-Vana Therapeutics, and PolyActiva, aiming to leverage cutting-edge technologies and expand their product portfolios. Emerging trends such as the development of smart drug delivery systems that respond to physiological cues and the integration of bioresorbable implants for long-term drug release are expected to shape the future trajectory of the market. While the market is experiencing strong growth, potential restraints may include the high cost of development and manufacturing, regulatory hurdles for novel technologies, and the need for extensive clinical validation. However, the persistent demand for safer, more effective, and patient-friendly ophthalmic treatment options, coupled with a growing global elderly population more susceptible to eye conditions, underscores a highly promising outlook for bioresorbable ophthalmic drug delivery systems across hospitals, clinics, and other healthcare settings worldwide.

This report offers an in-depth analysis of the global Bioresorbable Ophthalmic Drug Delivery System market, providing a comprehensive outlook on its trajectory from the historical period of 2019-2024 to an estimated year of 2025 and projecting robust growth through the forecast period of 2025-2033. Our study encompasses a detailed examination of market trends, driving forces, challenges, regional dominance, key growth catalysts, leading players, and significant industry developments.
The global Bioresorbable Ophthalmic Drug Delivery System market is experiencing a significant paradigm shift, driven by an increasing prevalence of ocular diseases, a growing elderly population, and a continuous pursuit of improved patient outcomes and compliance. For decades, traditional ophthalmic drug delivery methods, primarily eye drops, have been the mainstay of treatment. However, these methods suffer from limitations such as poor bioavailability, frequent dosing requirements, and inconsistent therapeutic levels, leading to suboptimal treatment efficacy and patient dissatisfaction. Bioresorbable ophthalmic drug delivery systems represent a revolutionary advancement, offering the potential for sustained drug release directly to the target site within the eye. These systems, typically implants or inserts, are designed to degrade naturally within the body over a predetermined period, eliminating the need for surgical removal. This inherent biodegradability significantly enhances patient comfort and reduces the risk of complications associated with reusable or permanent devices.
The market is witnessing a surge in research and development activities focused on innovative bioresorbable materials and advanced drug encapsulation techniques. Polymers such as polylactic acid (PLA), polyglycolic acid (PGA), and their copolymers are gaining traction due to their biocompatibility and controllable degradation rates. Furthermore, advancements in nanotechnology are enabling the development of micro- and nano-sized bioresorbable carriers that can precisely deliver therapeutic agents to specific ocular tissues. The focus is shifting towards personalized medicine, with systems being designed to deliver tailored dosages for specific conditions like neovascular age-related macular degeneration (nAMD), diabetic retinopathy (DR), glaucoma, and uveitis. The ability to achieve prolonged therapeutic drug levels, potentially reducing the frequency of injections or other invasive procedures from monthly to quarterly or even less frequent intervals, is a major driver of market adoption. For instance, a successful bioresorbable implant for wet AMD could drastically improve patient quality of life by reducing the burden of frequent intravitreal injections, which currently impact millions of patients annually worldwide. The estimated market size in the base year of 2025 is projected to reach several hundred million units globally, with a compound annual growth rate (CAGR) anticipated to exceed XX% in the coming years. This growth is fueled by the clinical advantages, patient preference, and the economic benefits of reduced healthcare resource utilization associated with improved treatment adherence and fewer adverse events. The ongoing clinical trials and product launches by key players are further solidifying the market's robust upward trend. The transition from conventional to bioresorbable solutions is not merely an incremental improvement but a fundamental evolution in ophthalmic therapeutics.
The bioresorbable ophthalmic drug delivery system market is being propelled by a confluence of powerful drivers, fundamentally reshaping how ocular diseases are managed. Foremost among these is the escalating global burden of age-related and chronic eye conditions. The aging demographic, coupled with lifestyle factors, is contributing to a significant increase in the incidence of conditions such as age-related macular degeneration (AMD), diabetic retinopathy (DR), glaucoma, and dry eye syndrome. These conditions often require long-term, consistent therapeutic intervention. Traditional drug delivery methods, predominantly eye drops, face inherent limitations in achieving sustained therapeutic drug concentrations, leading to suboptimal treatment outcomes and the need for frequent administration. Bioresorbable systems offer a compelling solution by providing sustained and targeted drug release directly to the affected ocular tissues, thereby improving efficacy and patient compliance. For instance, a patient with wet AMD, historically requiring monthly intravitreal injections, could potentially benefit from a bioresorbable implant offering sustained drug delivery for several months, drastically reducing the treatment burden.
Furthermore, the inherent advantages of bioresorbable systems, such as enhanced patient comfort and reduced invasiveness, are significant motivators for their adoption. The elimination of frequent injections or the need for topical eye drop application, which can be challenging for elderly patients or those with dexterity issues, significantly improves the patient experience. The self-degrading nature of these systems negates the need for surgical removal, further simplifying the treatment process and minimizing the risk of complications. The economic implications are also substantial; improved treatment adherence and reduced healthcare utilization due to fewer doctor visits and procedures translate into considerable cost savings for healthcare systems and patients alike. Research and development investments by pharmaceutical and biotechnology companies are also a critical driving force, leading to the continuous innovation of novel biomaterials and drug delivery platforms that offer improved performance and therapeutic benefits, further solidifying the market's growth trajectory. The estimated market in 2025 for bioresorbable ophthalmic drug delivery systems is projected to be in the hundreds of millions, with strong growth anticipated.
Despite the promising trajectory, the bioresorbable ophthalmic drug delivery system market faces several significant challenges and restraints that could temper its growth. A primary hurdle is the high cost of development and manufacturing. The research, preclinical testing, clinical trials, and specialized manufacturing processes required for bioresorbable implants are considerably more expensive than for traditional ophthalmic formulations. This elevated cost translates into higher product pricing, which can pose a barrier to widespread adoption, especially in resource-constrained healthcare systems and for patients with limited insurance coverage. The estimated market in 2025, while substantial, could see its expansion hampered by affordability concerns.
Another critical challenge is the regulatory landscape and the time-consuming approval process. Developing a novel medical device, especially one implanted within the eye, involves rigorous safety and efficacy evaluations by regulatory bodies such as the FDA and EMA. The extensive clinical data required to demonstrate the long-term safety, efficacy, and biodegradability of these systems can lead to protracted approval timelines, delaying market entry and return on investment for manufacturers. Furthermore, limited long-term clinical data and potential for unforeseen adverse events remain a concern. While bioresorbable materials are generally considered safe, any novel material introduced into the delicate ocular environment necessitates extensive long-term monitoring to identify any delayed complications, such as inflammation, immune responses, or incomplete degradation. Building physician and patient confidence in the long-term safety profile of these systems is crucial for their widespread acceptance. The variability in drug release rates, influenced by factors like ocular fluid composition and individual patient physiology, can also present a challenge in achieving consistent therapeutic outcomes, requiring further optimization of material science and drug formulation.
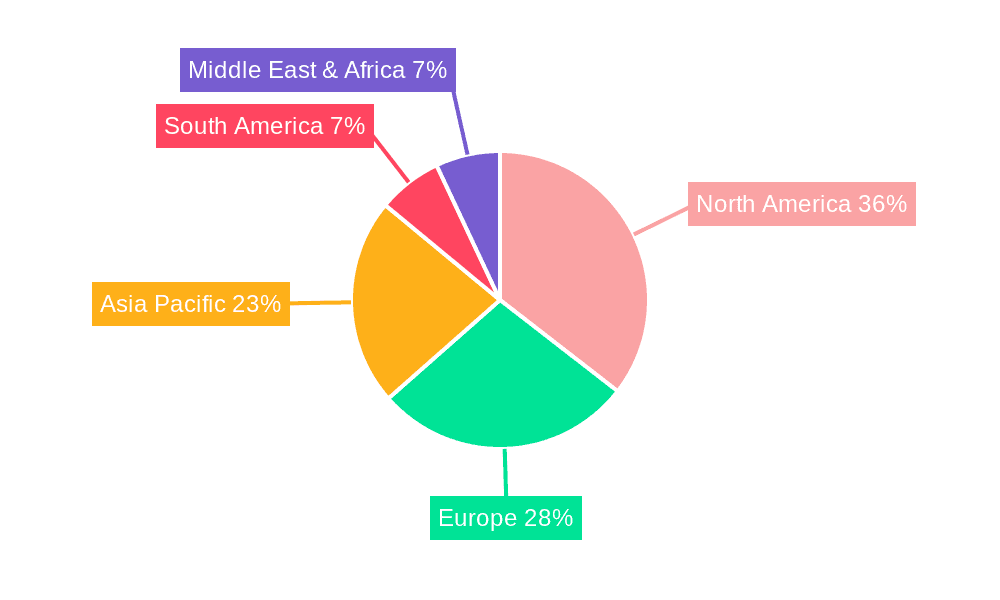
The global Bioresorbable Ophthalmic Drug Delivery System market is poised for significant expansion, with certain regions and segments expected to lead this growth. Among the regions, North America, particularly the United States, is anticipated to dominate the market in the forecast period (2025-2033). This dominance is underpinned by several key factors, including a high prevalence of ocular diseases, a robust healthcare infrastructure with advanced medical facilities, a high disposable income, and a strong emphasis on adopting innovative medical technologies. The significant patient population suffering from conditions like wet AMD and DR, coupled with the willingness of both healthcare providers and patients to embrace advanced treatment modalities for improved outcomes, fuels the demand for bioresorbable solutions. The presence of leading pharmaceutical and biotechnology companies with substantial R&D capabilities and a well-established regulatory framework that supports the timely approval of novel medical devices further solidifies North America's leading position.
Within the segments, Posterior Drug Delivery Methods are projected to command a substantial market share and drive significant growth. This is primarily due to the high unmet need and the therapeutic challenges associated with treating diseases affecting the posterior segment of the eye, such as age-related macular degeneration (AMD), diabetic retinopathy (DR), and retinal vein occlusions. Traditional drug delivery methods, including frequent intravitreal injections, are often associated with significant patient discomfort, procedural risks, and compliance issues. Bioresorbable implants and inserts designed for posterior segment delivery offer a paradigm shift by enabling sustained drug release directly to the retina or vitreous cavity. This localized delivery minimizes systemic exposure and offers the potential for less frequent administration, thereby improving patient quality of life and treatment adherence. The estimated market for posterior drug delivery methods in 2025 is expected to be in the hundreds of millions of units, with a projected CAGR that outpaces that of anterior segment delivery methods. The continuous innovation in developing longer-lasting and more precise posterior delivery systems, alongside ongoing clinical trials demonstrating their efficacy and safety, will be critical in maintaining this dominance. The successful introduction and adoption of bioresorbable devices for posterior segment diseases will likely represent a major step forward in ophthalmic therapeutics, significantly impacting the overall market landscape. Furthermore, the Hospitals application segment is expected to be a major contributor to market growth. Hospitals, being the primary centers for advanced ophthalmological procedures and chronic disease management, are well-equipped to adopt and integrate these sophisticated drug delivery systems. Their ability to handle complex procedures, manage post-operative care, and access advanced technologies makes them ideal settings for the implementation of bioresorbable ophthalmic drug delivery systems.
The bioresorbable ophthalmic drug delivery system industry is experiencing robust growth due to several key catalysts. The escalating global prevalence of chronic ocular diseases, such as age-related macular degeneration (AMD) and diabetic retinopathy (DR), is a primary driver, creating a sustained demand for more effective and patient-friendly treatment options. Advancements in biomaterial science are continuously yielding innovative, biocompatible, and biodegradable polymers, enabling the development of systems with tailored drug release profiles and improved therapeutic outcomes. Furthermore, the increasing patient preference for minimally invasive and convenient treatment modalities, coupled with a growing awareness of the limitations of traditional eye drop therapies, is accelerating the adoption of bioresorbable solutions. The estimated market size in 2025 for these systems is in the hundreds of millions, and these catalysts are expected to propel it further.
This comprehensive report provides a deep dive into the Bioresorbable Ophthalmic Drug Delivery System market, offering valuable insights for stakeholders. It meticulously analyzes market dynamics, including historical trends from 2019-2024, the estimated landscape of 2025, and a robust forecast from 2025-2033. The report delves into the crucial driving forces propelling this market forward, such as the rising burden of ocular diseases and technological advancements, while also critically examining the challenges and restraints, including high development costs and regulatory hurdles. Furthermore, it identifies key regions and segments poised for dominance, with a specific focus on Posterior Drug Delivery Methods and the Hospitals application segment, highlighting their significant growth potential and market share. The report also spotlights the leading players and significant developments within the sector, providing a forward-looking perspective on the industry's evolution. This extensive coverage ensures stakeholders are equipped with the knowledge necessary to navigate and capitalize on the burgeoning opportunities within the bioresorbable ophthalmic drug delivery system market. The estimated market in 2025 for bioresorbable ophthalmic drug delivery systems is projected to reach several hundred million units, underscoring the significant current and future market potential.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5%.
Key companies in the market include Boehringer Ingelheim, Allergan, Re-Vana Therapeutics, PolyActiva, .
The market segments include Type, Application.
The market size is estimated to be USD 451.8 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Bioresorbable Ophthalmic Drug Delivery System," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Bioresorbable Ophthalmic Drug Delivery System, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.