1. What is the projected Compound Annual Growth Rate (CAGR) of the Biological Small-Caliber Artificial Blood Vessel?
The projected CAGR is approximately 5.02%.
 Biological Small-Caliber Artificial Blood Vessel
Biological Small-Caliber Artificial Blood VesselBiological Small-Caliber Artificial Blood Vessel by Type (Autologous Cell-Derived Blood Vessels, Blood Vessels Derived From Non-Autologous Cells, Others), by Application (General Hospital, Specialized Hospital, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
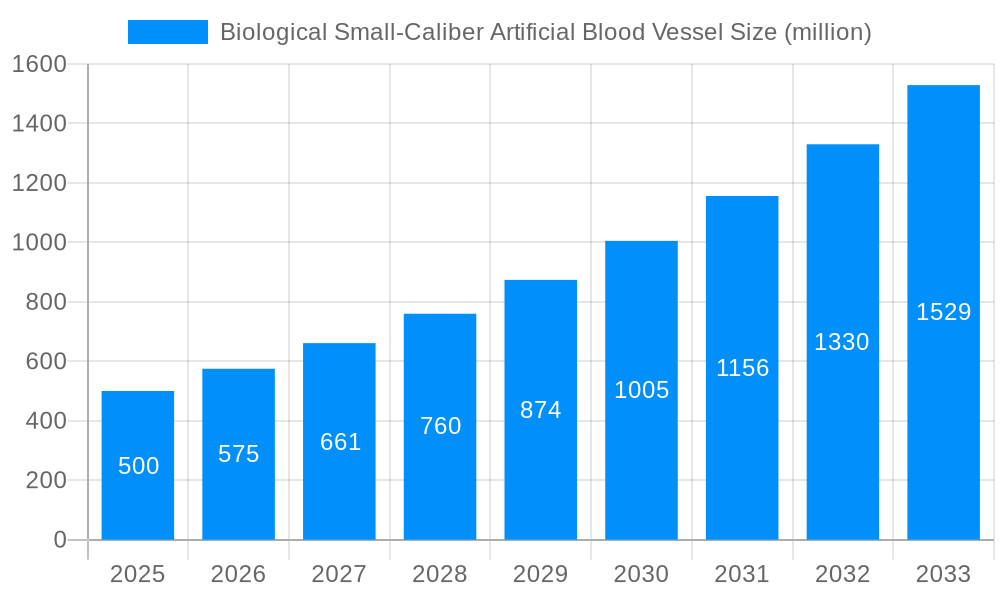
The Biological Small-Caliber Artificial Blood Vessel market is experiencing robust expansion, projected to reach approximately $500 million by 2025, with a Compound Annual Growth Rate (CAGR) of around 15% from 2019 to 2033. This significant growth is propelled by a confluence of escalating cardiovascular disease prevalence globally, increasing demand for minimally invasive surgical procedures, and continuous advancements in biomaterial science and tissue engineering. The aging global population, coupled with a rise in lifestyle-related health issues, directly contributes to a higher incidence of conditions requiring vascular reconstruction, thereby fueling the demand for effective small-caliber artificial blood vessel solutions. Furthermore, the growing preference for autologous cell-derived blood vessels, offering superior biocompatibility and reduced rejection rates, is a key driver, though non-autologous cell-derived options are also gaining traction due to their scalability and potential for off-the-shelf availability. The market's trajectory is significantly influenced by the pursuit of improved patient outcomes and the reduction of long-term complications associated with traditional vascular grafts.
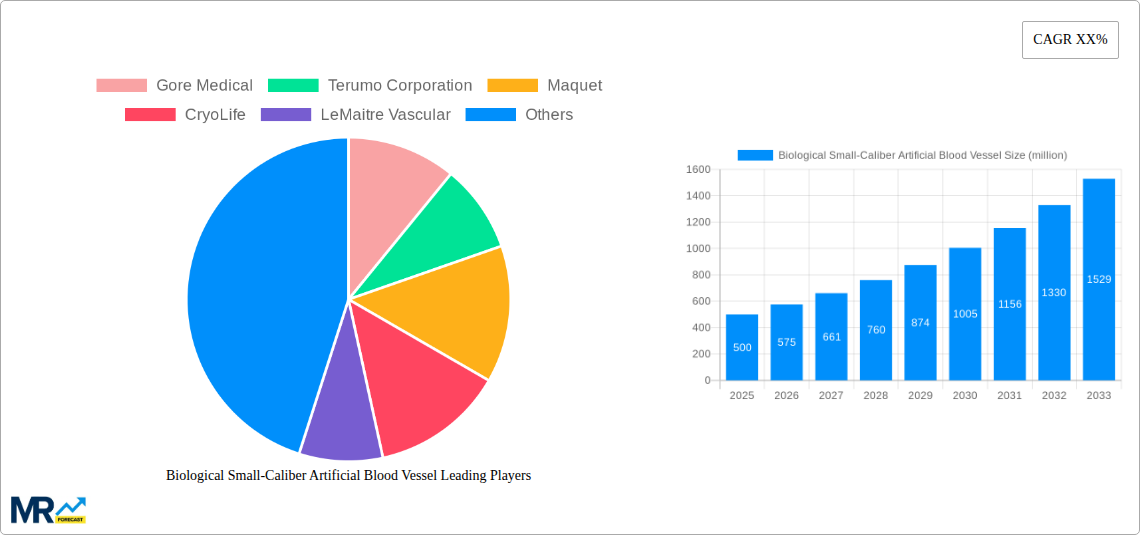

The market faces certain restraints, primarily revolving around the high cost of research and development, stringent regulatory approval processes for novel medical devices, and the need for extensive clinical validation. However, these challenges are being systematically addressed through collaborative efforts between research institutions and leading medical device manufacturers. Emerging trends like the integration of smart technologies for enhanced monitoring and the development of patient-specific vascular grafts are poised to further shape the market landscape. Specialized hospitals are anticipated to be the dominant application segment, leveraging these advanced solutions for complex cardiovascular surgeries. Geographically, North America and Europe are expected to lead the market due to their advanced healthcare infrastructure and high adoption rates of cutting-edge medical technologies. The Asia Pacific region, however, presents a significant growth opportunity driven by a burgeoning healthcare sector and increasing patient awareness. Companies like Gore Medical, Terumo Corporation, and Maquet are at the forefront, investing heavily in innovation to capture a larger market share.

Here is a unique report description for "Biological Small-Caliber Artificial Blood Vessel," incorporating your specified elements:
The global biological small-caliber artificial blood vessel market is poised for substantial expansion, driven by a confluence of escalating cardiovascular disease prevalence, technological advancements in regenerative medicine, and a growing demand for minimally invasive treatment options. Our comprehensive analysis, spanning the Study Period: 2019-2033, with a Base Year: 2025 and an Estimated Year: 2025, reveals a dynamic landscape characterized by increasing R&D investments and a shifting preference towards biomimetic and bioresorbable solutions. During the Historical Period: 2019-2024, the market witnessed steady growth, primarily fueled by the limitations of traditional synthetic grafts in small-diameter applications, such as coronary artery bypass grafting and peripheral arterial disease treatment. The market is projected to reach an estimated value of $1,200 million units by 2025, with a robust compound annual growth rate (CAGR) anticipated throughout the Forecast Period: 2025-2033. Key trends include the increasing adoption of autologous cell-derived blood vessels, promising enhanced biocompatibility and reduced immunogenicity, and the continuous refinement of fabrication techniques to mimic the intricate structure and function of native vessels. Furthermore, the integration of advanced imaging and surgical navigation technologies is expected to further optimize the implantation and long-term performance of these crucial medical devices. The market's trajectory is also influenced by favorable reimbursement policies in developed nations and the expanding healthcare infrastructure in emerging economies, creating a fertile ground for innovation and commercialization. The development of off-the-shelf, readily available allogeneic or xenogeneic grafts, engineered to possess superior hemocompatibility and reduced thrombogenicity, represents another significant area of focus, aiming to overcome the limitations of autologous tissue harvesting and reduce procedural complexities.
The burgeoning demand for effective treatments for cardiovascular diseases, which remain a leading cause of mortality worldwide, is a primary catalyst for the biological small-caliber artificial blood vessel market. The inherent limitations of synthetic grafts in small-diameter applications, such as high rates of thrombosis, stenosis, and infection, have created a critical unmet need for biocompatible and functional alternatives. Biological grafts, particularly those derived from autologous cells, offer superior hemocompatibility and a reduced risk of immune rejection, paving the way for improved patient outcomes. The rapid advancements in tissue engineering and regenerative medicine, coupled with significant investments in research and development by leading players, are instrumental in the creation of innovative and effective biological vascular substitutes. These advancements are enabling the development of grafts that not only replace damaged vessels but also actively integrate with the host tissue, promoting neovascularization and long-term patency. Furthermore, the increasing global focus on preventive healthcare and early intervention for cardiovascular conditions is leading to a larger patient pool requiring advanced surgical solutions, thereby stimulating market growth. The growing acceptance of these advanced biomaterials by the medical community, supported by positive clinical trial data demonstrating enhanced efficacy and safety profiles, further fuels their adoption.
Despite the promising outlook, the biological small-caliber artificial blood vessel market faces several significant challenges and restraints that can impede its growth trajectory. The high cost associated with research, development, and manufacturing of these complex biological constructs remains a primary barrier to widespread adoption, particularly in resource-limited settings. The intricate regulatory approval processes for novel regenerative medicine products also contribute to extended development timelines and increased financial burdens for manufacturers. Furthermore, ensuring the long-term biocompatibility and durability of these grafts, especially in challenging anatomical locations subjected to high hemodynamic stress, continues to be an area of ongoing research and development. The potential for immunogenic responses, even with autologous cell-derived grafts, and the risk of graft failure due to thrombosis or infection, though reduced compared to synthetics, are still concerns that require diligent management. The lack of widespread awareness and physician education regarding the benefits and optimal use of biological small-caliber artificial blood vessels can also hinder market penetration. Addressing these multifaceted challenges through sustained innovation, cost-reduction strategies, streamlined regulatory pathways, and comprehensive educational initiatives will be crucial for unlocking the full potential of this vital medical technology.
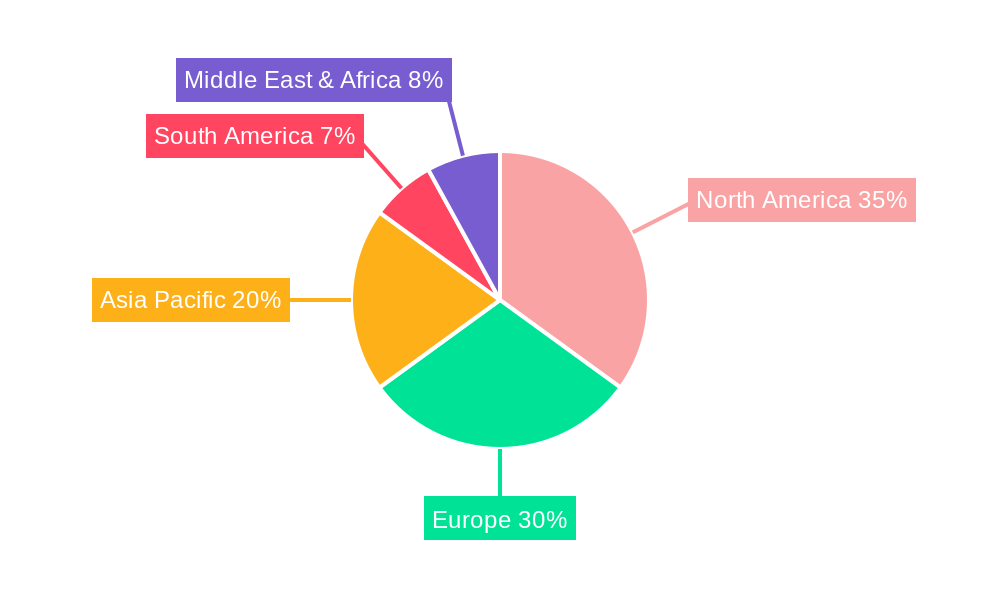
Dominant Region/Country: North America is anticipated to remain a dominant region in the biological small-caliber artificial blood vessel market, driven by a confluence of factors including a high prevalence of cardiovascular diseases, advanced healthcare infrastructure, significant investments in R&D, and favorable reimbursement policies. The United States, in particular, is a major hub for medical device innovation and adoption, with a strong presence of leading biotechnology and medical device companies actively engaged in developing and commercializing next-generation vascular grafts. The robust clinical trial ecosystem and a well-established network of specialized cardiovascular centers further bolster market growth in this region.
Dominant Segment: Within the market, Autologous Cell-Derived Blood Vessels are projected to exhibit the most significant growth and market share. This segment's dominance is attributable to its inherent advantage of minimal immunogenicity and superior biocompatibility, leading to enhanced integration with host tissues and reduced risks of rejection and infection. The ability of autologous grafts to potentially mimic the structural and functional properties of native blood vessels makes them highly desirable for critical applications like coronary artery bypass grafting and repairs of damaged smaller arteries. While the procurement and culturing of autologous cells can involve more complex and time-consuming procedures compared to off-the-shelf options, the long-term clinical benefits and reduced post-operative complications observed in numerous studies are driving their increasing preference among both surgeons and patients. The continuous advancements in cell culture techniques, bioreactor technology, and decellularization/recellularization methods are further refining the efficacy and scalability of autologous cell-derived blood vessels. The market value for this segment is expected to reach approximately $850 million units by 2025, highlighting its pivotal role in shaping the future of vascular reconstruction. The focus on personalized medicine further bolsters the appeal of autologous solutions, as they offer a tailored approach to individual patient needs, minimizing the risk of adverse reactions.
Secondary Dominant Segment: Following closely, the Blood Vessels Derived From Non-Autologous Cells segment, encompassing allogeneic and xenogeneic sources, also holds substantial market potential. While facing greater challenges in terms of immunogenicity and potential rejection, ongoing research in decellularization and recellularization techniques, along with strategies to mitigate immune responses, is improving the viability and application of these grafts. The advantage of being readily available, eliminating the time-consuming process of autologous cell harvesting, makes this segment attractive for emergency procedures and for patients where autologous tissue is not viable. The market value for this segment is estimated to reach around $250 million units by 2025.
Application Dominance: Within the application segment, Specialized Hospitals are expected to be the primary drivers of market demand. These institutions typically house advanced surgical departments, cutting-edge technology, and a higher concentration of experienced cardiovascular surgeons specializing in complex procedures requiring small-caliber vascular grafts. The ability of specialized hospitals to handle intricate surgeries and manage the advanced post-operative care associated with biological grafts positions them at the forefront of adoption. The estimated market share for specialized hospitals is projected to be around $950 million units by 2025.
The biological small-caliber artificial blood vessel industry is propelled by several key growth catalysts. Foremost is the persistent and growing global burden of cardiovascular diseases, necessitating innovative and effective surgical interventions. Advancements in regenerative medicine and tissue engineering are continuously producing more sophisticated and biocompatible vascular grafts. Furthermore, the increasing preference for minimally invasive procedures and improved patient outcomes, driven by a greater understanding of the limitations of traditional synthetic grafts in small-diameter applications, is a significant driver. Supportive government initiatives and increasing R&D investments by leading companies further fuel innovation and market expansion.
This report provides a comprehensive analysis of the biological small-caliber artificial blood vessel market, offering invaluable insights for stakeholders. It delves into the intricate trends, driving forces, and challenges that shape the industry's trajectory. With detailed market segmentation based on type, application, and region, the report illuminates key growth areas and identifies dominant players. The analysis extends to significant developments and future projections, providing a holistic view of the market landscape up to 2033. This report is essential for understanding the evolving market dynamics, identifying strategic opportunities, and making informed business decisions within this critical sector of medical technology.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 5.02% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 5.02%.
Key companies in the market include Gore Medical, Terumo Corporation, Maquet, CryoLife, LeMaitre Vascular, Cook Medical, Boston Scientific, Medtronic.
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A and volume, measured in K.
Yes, the market keyword associated with the report is "Biological Small-Caliber Artificial Blood Vessel," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Biological Small-Caliber Artificial Blood Vessel, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.