1. What is the projected Compound Annual Growth Rate (CAGR) of the Biocompatibility Testing for Medical Device?
The projected CAGR is approximately 15.19%.
 Biocompatibility Testing for Medical Device
Biocompatibility Testing for Medical DeviceBiocompatibility Testing for Medical Device by Type (Cytotoxicity Testing, Sensitization Testing, Irritation Testing, Implantation Testing, Hemocompatibility Testing, Others), by Application (Medical Device Company, Hospital, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
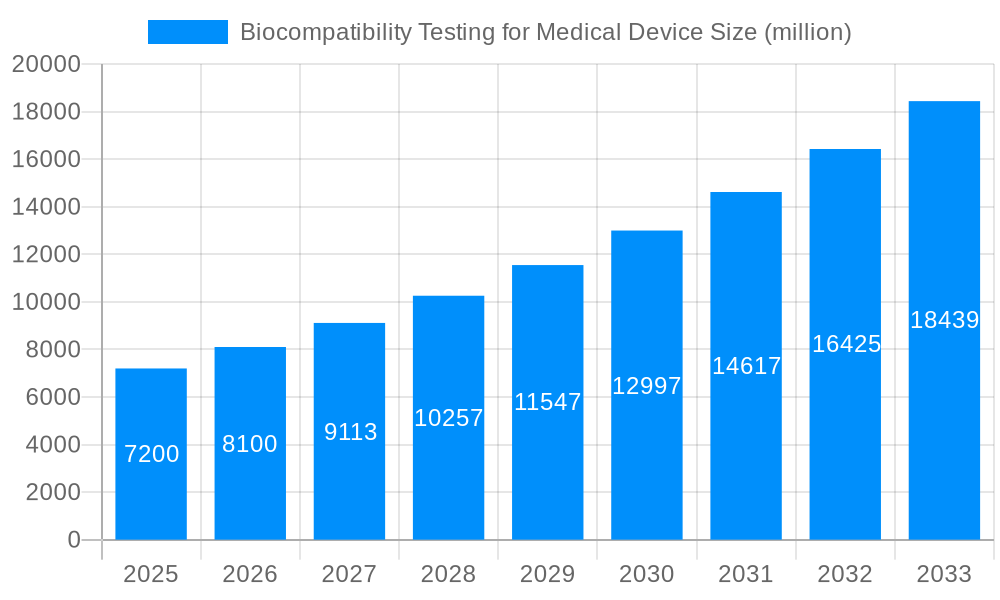
The global Biocompatibility Testing for Medical Devices market is projected for substantial expansion, expected to reach USD 9.21 billion by 2025. This growth is fueled by increasing demand for advanced, safe medical devices and evolving global regulatory standards. The market is anticipated to grow at a CAGR of 15.19% from the base year 2025 through 2033. Key drivers include the rising incidence of chronic diseases, an aging global demographic, and the advancement of technologies such as implantable devices and sophisticated drug delivery systems. Stringent regulations from bodies like the FDA and EMA are compelling manufacturers to invest in comprehensive biocompatibility testing, ensuring material safety and enhancing patient well-being.
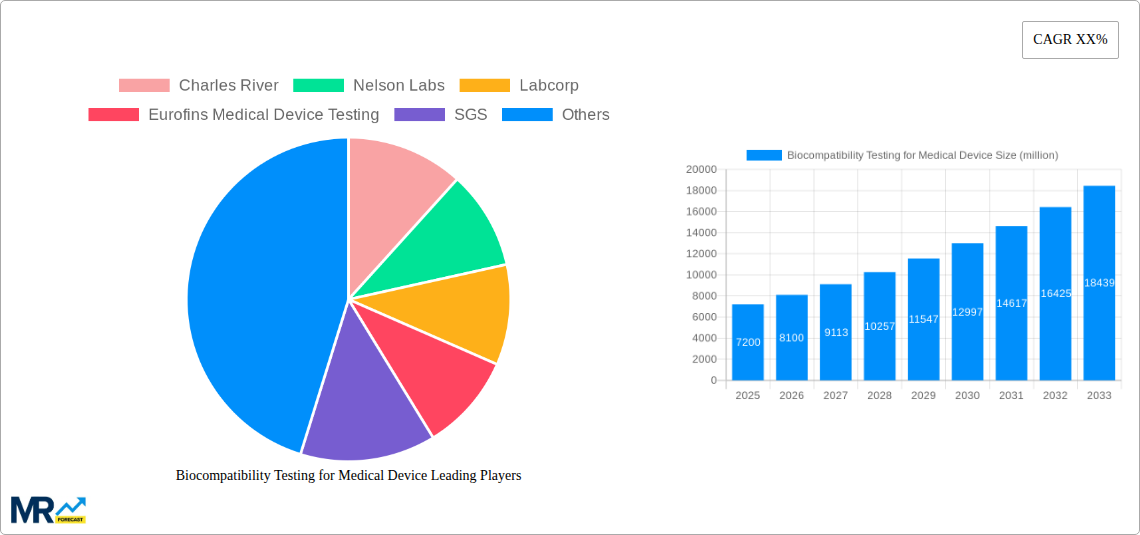

Market segmentation includes testing types such as Cytotoxicity, Sensitization, and Irritation Testing, which are crucial for evaluating basic biological interactions. Medical Device Companies are the primary end-users, followed by healthcare providers and research institutions. North America and Europe currently lead the market due to robust healthcare infrastructure, significant R&D investment, and a high concentration of leading medical device manufacturers. The Asia Pacific region is emerging as a significant growth hub, driven by increasing healthcare investments, a growing medical device manufacturing sector, and rising awareness of safety standards in key economies like China and India. Key market players, including Charles River, Nelson Labs, Labcorp, and Eurofins Medical Device Testing, are actively expanding their services and global reach to meet this dynamic market demand.

This report provides an in-depth analysis of the Biocompatibility Testing for Medical Devices market, outlining its size, growth trajectory, and future projections.
The global Biocompatibility Testing for Medical Device market is experiencing robust expansion, driven by an increasing demand for advanced medical devices and a heightened focus on patient safety. The study period, spanning from 2019 to 2033, with a base year of 2025 and a forecast period from 2025 to 2033, illustrates a significant upward trajectory. The historical period (2019-2024) laid the groundwork for this growth, characterized by evolving regulatory landscapes and early adoption of rigorous testing protocols. In the estimated year of 2025, the market is projected to reach a valuation in the hundreds of millions of dollars, a figure expected to ascend considerably as the forecast period unfolds. Key market insights reveal a growing emphasis on sophisticated in-vitro testing methodologies, which offer faster turnaround times and reduced reliance on animal models, aligning with ethical considerations and cost-efficiency. The rising prevalence of chronic diseases worldwide, such as cardiovascular conditions, diabetes, and orthopedic ailments, directly fuels the need for an ever-expanding array of medical devices, from implantable sensors to advanced surgical tools, each necessitating stringent biocompatibility validation. Furthermore, the burgeoning field of personalized medicine and the development of novel biomaterials are introducing new testing paradigms, demanding adaptability and innovation from testing service providers. The increasing integration of AI and machine learning in data analysis of biocompatibility studies is also a notable trend, promising enhanced predictive capabilities and a deeper understanding of material-tissue interactions. Regulatory bodies globally are continuously updating guidelines, such as ISO 10993, to encompass emerging technologies and materials, thereby mandating comprehensive and standardized biocompatibility testing for all medical devices entering the market. This evolving regulatory environment acts as a powerful impetus for market growth, compelling manufacturers to invest more heavily in specialized testing services. The market's expansion is not merely quantitative; it reflects a qualitative shift towards more comprehensive risk assessment and a proactive approach to ensuring device safety throughout its lifecycle.
Several potent forces are actively propelling the Biocompatibility Testing for Medical Device market forward. Foremost among these is the escalating global demand for medical devices, driven by an aging population, the increasing incidence of chronic diseases, and advancements in healthcare infrastructure, particularly in emerging economies. As these devices come into direct or indirect contact with the human body, ensuring their biocompatibility becomes paramount for patient safety and regulatory compliance. This fundamental requirement translates directly into increased demand for specialized testing services. Moreover, stringent regulatory frameworks established by authorities like the FDA in the U.S., the EMA in Europe, and other national bodies worldwide, mandate comprehensive biocompatibility evaluations as a prerequisite for market approval. These regulations, continually being updated to address new technologies and potential risks, compel manufacturers to engage third-party testing laboratories, thereby stimulating market growth. The accelerating pace of innovation in the medical device sector, with the introduction of novel materials, complex designs, and sophisticated functionalities, also contributes significantly. Each new material or design innovation requires thorough biocompatibility assessment to ensure it does not elicit adverse biological responses. The growing awareness among both healthcare professionals and patients regarding the potential risks associated with poorly characterized medical devices further amplifies the demand for robust testing, pushing manufacturers to invest in thorough biocompatibility assessments to build trust and ensure patient well-being.
Despite the robust growth, the Biocompatibility Testing for Medical Device market encounters several significant challenges and restraints that can temper its expansion. A primary hurdle is the considerable cost associated with comprehensive biocompatibility testing. These studies, especially those involving in-vivo evaluations, can be time-consuming and expensive, posing a financial burden, particularly for small and medium-sized enterprises (SMEs) and start-ups. This cost factor can sometimes lead to compromises in the extent of testing, especially in resource-constrained environments. Another significant challenge lies in the complexity and evolving nature of regulatory requirements. Keeping abreast of ever-changing international standards and guidelines, such as the ISO 10993 series, and ensuring compliance across different global markets can be a daunting task for manufacturers. This complexity necessitates specialized expertise, which may not be readily available within all organizations. Furthermore, the limited availability of skilled personnel proficient in conducting and interpreting biocompatibility tests is a growing concern. The specialized nature of this field requires a deep understanding of toxicology, biology, and material science, and a shortage of such experts can bottleneck testing capacity. The ethical considerations and societal pressures surrounding the use of animal testing, although diminishing with the rise of alternative methods, still present challenges in certain jurisdictions and for specific test types, potentially leading to longer validation processes for new in-vitro methods. Finally, long lead times for certain in-vivo studies can impact the overall product development timeline, a critical factor in the fast-paced medical device industry, potentially delaying market entry.
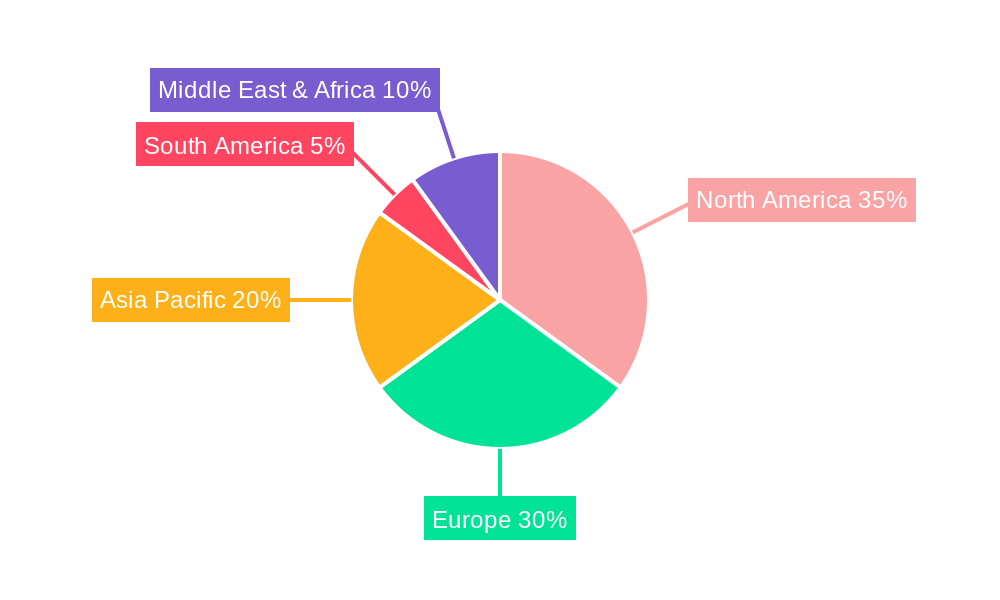
The Biocompatibility Testing for Medical Device market is characterized by dominance across both specific regions and critical market segments.
Key Dominating Regions and Countries:
Key Dominating Segments:
The synergy between these dominant regions and segments creates a robust market ecosystem. The innovation originating from medical device companies in North America and Europe, coupled with the increasing regulatory stringency and manufacturing growth in Asia Pacific, ensures a consistent and growing demand for essential tests like Cytotoxicity Testing.
Several key growth catalysts are fueling the expansion of the Biocompatibility Testing for Medical Device industry. The relentless pace of innovation in medical technology, leading to the development of novel and complex devices, necessitates advanced biocompatibility assessments. Furthermore, increasingly stringent global regulatory requirements, such as the EU's Medical Device Regulation (MDR), are mandating more comprehensive testing protocols, driving up demand for specialized services. The growing awareness and demand for patient safety from both healthcare providers and end-users also play a crucial role, encouraging manufacturers to prioritize thorough biocompatibility evaluations.
This comprehensive report offers an in-depth analysis of the Biocompatibility Testing for Medical Device market, providing crucial insights for stakeholders. It details market size, growth projections, and key trends across the study period (2019-2033), with a detailed forecast from 2025-2033. The report elaborates on the driving forces, challenges, and restraints influencing market dynamics, supported by specific market data and valuations in the millions of dollars. It meticulously examines regional and segment-specific dominance, highlighting key players and significant industry developments. The report also delves into growth catalysts, offering strategic perspectives for market participants to leverage. This all-encompassing view ensures that readers gain a thorough understanding of the current landscape and future trajectory of this vital sector.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 15.19% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 15.19%.
Key companies in the market include Charles River, Nelson Labs, Labcorp, Eurofins Medical Device Testing, SGS, BDC Laboratories, Eurofins, Measurlabs, TÜV SÜD, UL Solutions, Pace Analytical, Pacific BioLabs, WuXi AppTec, .
The market segments include Type, Application.
The market size is estimated to be USD 9.21 billion as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in billion.
Yes, the market keyword associated with the report is "Biocompatibility Testing for Medical Device," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Biocompatibility Testing for Medical Device, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.