1. What is the projected Compound Annual Growth Rate (CAGR) of the Bioartificial Liver Therapeutic Technology?
The projected CAGR is approximately XX%.
 Bioartificial Liver Therapeutic Technology
Bioartificial Liver Therapeutic TechnologyBioartificial Liver Therapeutic Technology by Type (/> Molecular Adsorbent Recirculating System (MARS), Fractionated Plasma Separation And Adsorption System (Prometheus), Single-Pass Albumin Dialysis (SPAD), Selective Plasma Filtration Therapy), by Application (/> Treatment, R&D), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The Bioartificial Liver Therapeutic Technology market is poised for significant expansion, projected to reach approximately $1,250 million by 2025. This growth is fueled by a substantial Compound Annual Growth Rate (CAGR) of around 15%, indicating a robust and dynamic market trajectory. The primary drivers for this expansion stem from the increasing prevalence of liver diseases, such as cirrhosis and acute liver failure, coupled with a growing demand for advanced treatment alternatives to traditional liver transplantation. The limitations of organ availability for transplantation, coupled with the inherent risks and long recovery periods associated with it, are directly contributing to the rising adoption of bioartificial liver devices. Furthermore, ongoing advancements in cell therapy, biomaterials, and device engineering are enhancing the efficacy and safety of these technologies, making them more attractive therapeutic options for both patients and healthcare providers. The market’s development is also supported by increasing investment in research and development by key players and a growing awareness of the potential of bioartificial liver systems in critical care settings.
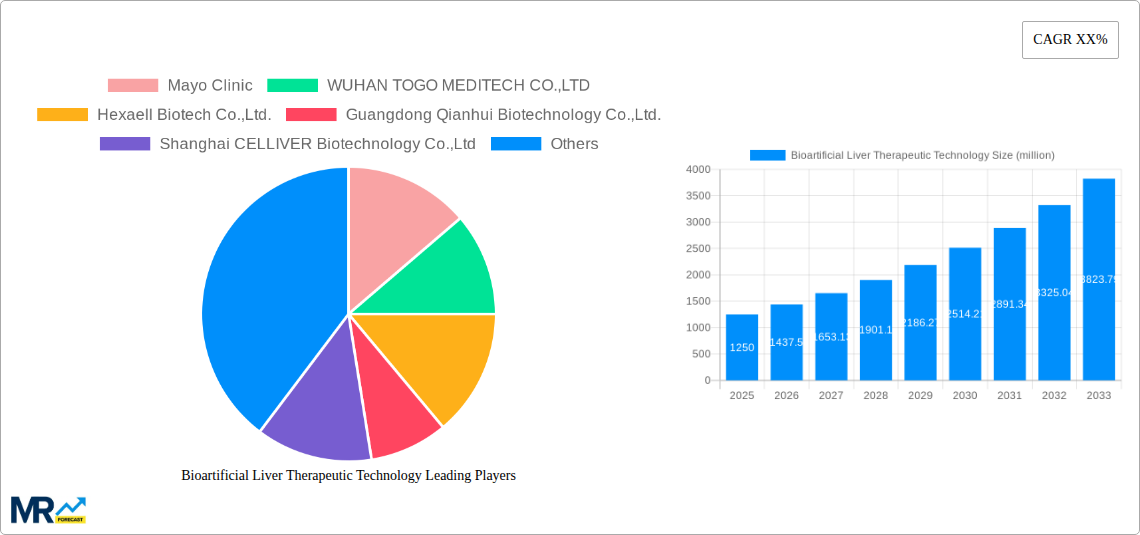

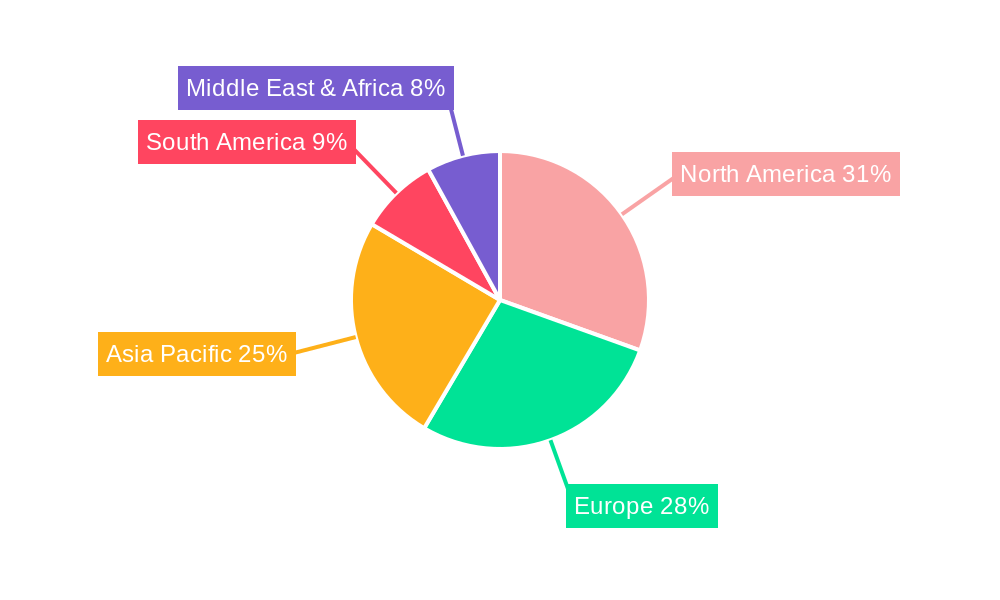
The market is segmented into distinct technology types, with Molecular Adsorbent Recirculating System (MARS) and Fractionated Plasma Separation and Adsorption System (Prometheus) holding significant shares due to their established presence and proven therapeutic benefits in managing liver failure. The Application segment is dominated by Treatment, reflecting the direct use of these technologies in clinical settings to support patients with liver dysfunction. However, Research and Development (R&D) applications are also showing promising growth as new iterations and improved functionalities are continuously being explored. Geographically, North America and Europe currently lead the market, driven by sophisticated healthcare infrastructures, high healthcare spending, and established regulatory frameworks supporting medical device innovation. The Asia Pacific region, particularly China and India, is expected to witness the fastest growth, owing to a large patient pool, increasing healthcare expenditure, and a burgeoning biotechnology sector actively investing in novel therapeutic solutions.
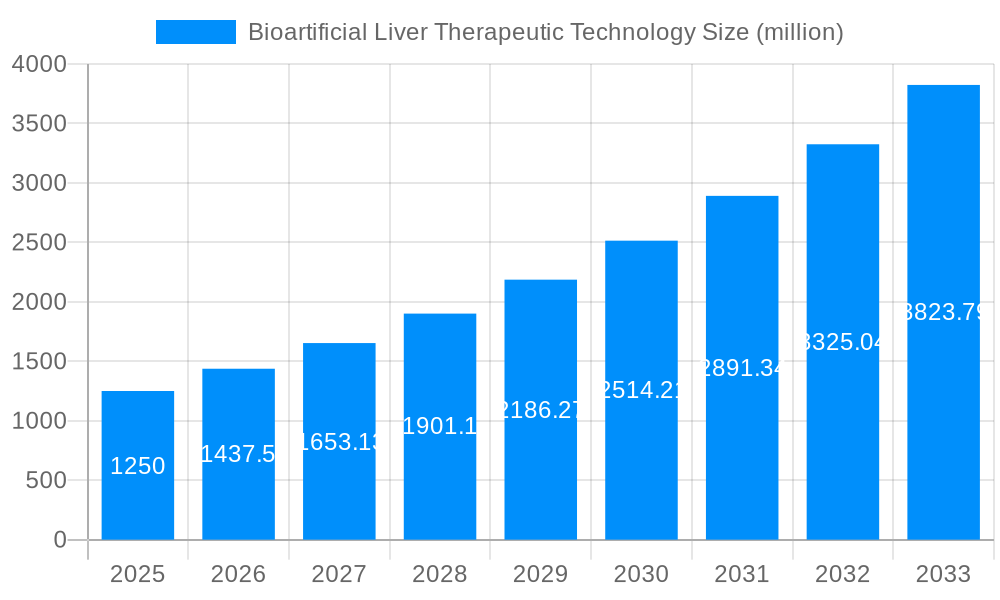

The global bioartificial liver (BAL) therapeutic technology market is poised for significant expansion, driven by an increasing incidence of liver diseases and the limitations of traditional treatments like liver transplantation. XXX projects a robust CAGR, indicating substantial growth from the historical period of 2019-2024 to the projected forecast period of 2025-2033. The base year of 2025 is expected to witness market values in the hundreds of millions of units, a figure anticipated to escalate considerably by the end of the study period. Key market insights reveal a burgeoning demand for advanced therapeutic solutions that can bridge the gap for patients awaiting transplants or those with acute liver failure. Innovations in bioreactor design, cell encapsulation techniques, and the development of more efficient detoxification and metabolic support systems are central to this trend. The market is also experiencing a paradigm shift towards personalized medicine, with a growing focus on developing BAL devices tailored to individual patient needs. Furthermore, the increasing investment in research and development by both academic institutions and private companies is fueling the pipeline of novel BAL technologies. The integration of sophisticated monitoring systems and artificial intelligence to optimize device performance is another emerging trend. The global adoption of these advanced therapies, coupled with favorable regulatory pathways for innovative medical devices, will be instrumental in shaping the future landscape of bioartificial liver technology. The market's trajectory is not merely about numerical growth but also about achieving greater clinical efficacy and accessibility, thereby improving patient outcomes and quality of life.
Several potent forces are propelling the bioartificial liver therapeutic technology market forward. The escalating global burden of chronic liver diseases, including hepatitis, cirrhosis, and non-alcoholic fatty liver disease (NAFLD), coupled with a rise in acute liver failure cases, is creating an urgent need for effective treatment alternatives. Liver transplantation, while life-saving, faces significant challenges such as organ donor shortages and the risk of rejection, making bioartificial liver devices a crucial bridging therapy or a potential long-term solution. Technological advancements in cell culture, biomaterials, and engineering have significantly improved the efficacy and longevity of bioartificial liver systems. The development of novel bioreactors capable of providing optimal cellular environment and efficient nutrient/waste exchange, alongside progress in encapsulating hepatocytes to prevent immune rejection, are key drivers. Furthermore, increased governmental and private funding for regenerative medicine and advanced therapeutic technologies is fostering innovation and accelerating the development pipeline. As research institutions and biotechnology companies invest heavily in R&D, the capabilities of bioartificial livers are continuously being enhanced, offering hope for more effective and accessible treatments for liver failure patients worldwide.
Despite the promising outlook, the bioartificial liver therapeutic technology market faces several significant challenges and restraints that could temper its growth trajectory. One of the primary hurdles is the limited clinical validation and regulatory approval for many emerging technologies. While promising in preclinical settings, translating these innovations into widespread clinical adoption requires extensive and costly human trials, which can be time-consuming and fraught with uncertainty. Another major challenge lies in the complexity and cost of manufacturing and implementation. Producing high-quality, functional bioartificial livers often involves intricate cell culture processes and specialized equipment, leading to high production costs. The subsequent treatment costs can also be prohibitive, limiting accessibility for a significant portion of the patient population. Furthermore, long-term efficacy and patient outcomes remain areas requiring further investigation. While promising as a temporary solution, the durability and sustained metabolic function of bioartificial livers over extended periods are still being explored. Ensuring adequate cellular viability, preventing immune responses, and achieving efficient detoxification and synthesis of essential proteins are ongoing technical challenges. Finally, ethical considerations and public perception surrounding the use of bioengineered organs and cell-based therapies can also pose a restraint, requiring careful communication and education to foster acceptance.
The bioartificial liver therapeutic technology market is anticipated to witness dominance from specific regions and segments, driven by a confluence of factors including advanced healthcare infrastructure, robust research and development capabilities, and a higher prevalence of liver diseases.
Dominant Segments:
Dominant Regions/Countries:
North America (particularly the United States): This region is expected to maintain its leadership due to several contributing factors:
Asia-Pacific (particularly China): This region is projected to witness the fastest growth and emerge as a significant player, driven by:
The bioartificial liver therapeutic technology industry is propelled by several key growth catalysts. The escalating global prevalence of liver diseases, from chronic conditions to acute liver failure, creates an undeniable demand for innovative treatments. Furthermore, the persistent shortage of viable donor organs for transplantation necessitates effective bridging therapies, a role perfectly suited for bioartificial livers. Technological advancements in biomaterials, cell encapsulation, and bioreactor design are continually enhancing the efficacy and safety of these devices, making them more attractive to both clinicians and patients. Finally, increasing governmental and private investments in regenerative medicine and life sciences research are fostering a supportive ecosystem for innovation and commercialization.
This report offers an all-encompassing examination of the bioartificial liver therapeutic technology market, spanning from 2019 to 2033. It meticulously analyzes market trends, growth drivers, and prevailing challenges, providing a nuanced understanding of the industry's dynamics. The report delves into key market insights, projecting significant growth from the historical period of 2019-2024 to the forecast period of 2025-2033, with the base year of 2025 anticipated to reach market values in the hundreds of millions of units. It identifies dominant market segments, such as MARS and Prometheus systems for treatment applications, and highlights leading regions like North America and the rapidly growing Asia-Pacific. Furthermore, the report provides an exhaustive list of key industry players and chronicles significant developments with specific year/month attributions, offering a comprehensive overview for stakeholders seeking to navigate this evolving technological landscape.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XX%.
Key companies in the market include Mayo Clinic, WUHAN TOGO MEDITECH CO.,LTD, Hexaell Biotech Co.,Ltd., Guangdong Qianhui Biotechnology Co.,Ltd., Shanghai CELLIVER Biotechnology Co.,Ltd, Shandong yiruite Biotechnology Co.,Ltd, Guanhao Biotech Co.,Ltd..
The market segments include Type, Application.
The market size is estimated to be USD XXX million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 4480.00, USD 6720.00, and USD 8960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "Bioartificial Liver Therapeutic Technology," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Bioartificial Liver Therapeutic Technology, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.