1. What is the projected Compound Annual Growth Rate (CAGR) of the Autoimmune Diagnosis Reagents?
The projected CAGR is approximately 9.1%.
 Autoimmune Diagnosis Reagents
Autoimmune Diagnosis ReagentsAutoimmune Diagnosis Reagents by Application (Hospital, Third-party Testing Agency), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
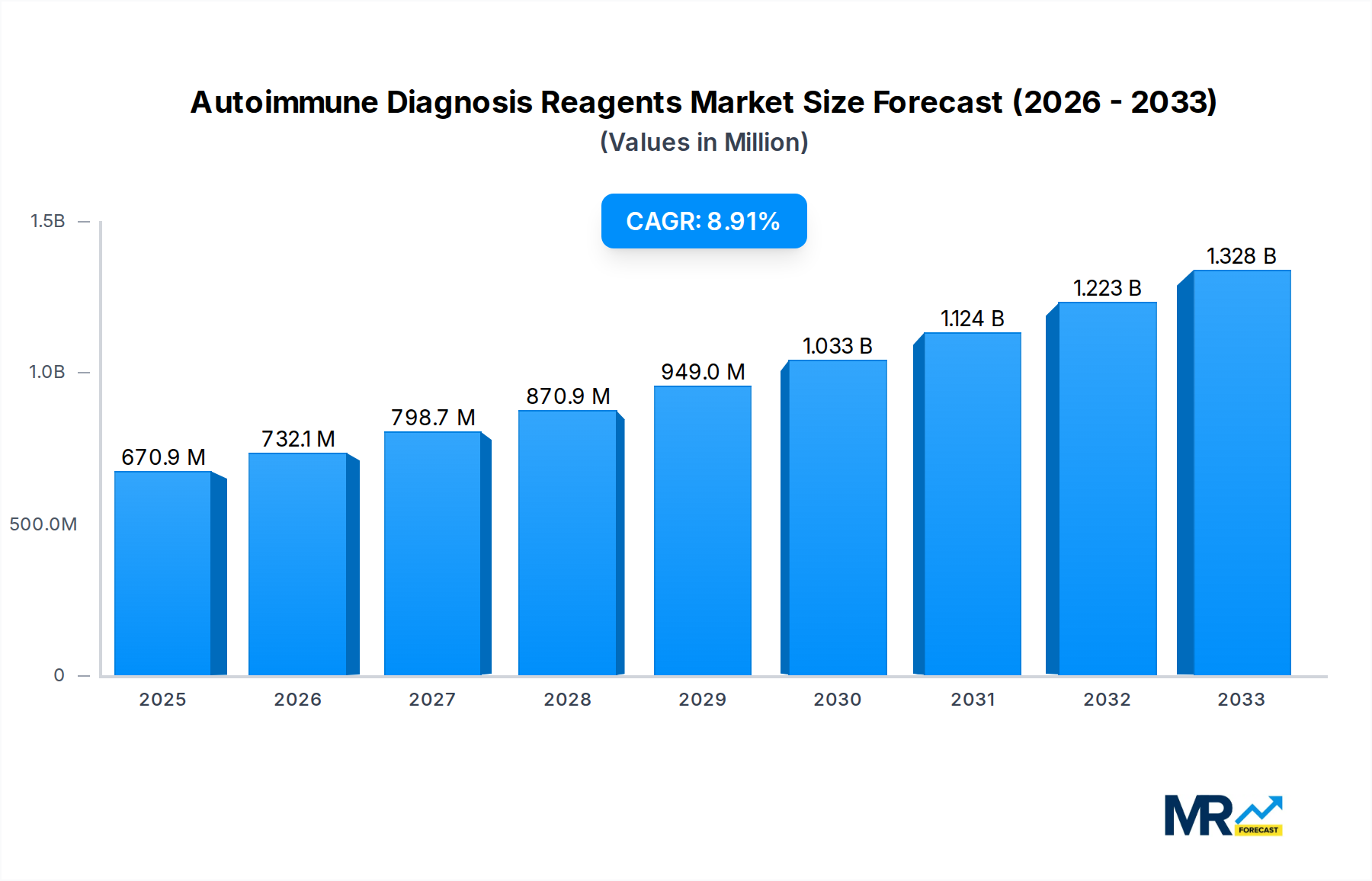
The global Autoimmune Diagnosis Reagents market is projected for robust expansion, currently valued at approximately $670.9 million and anticipated to grow at a Compound Annual Growth Rate (CAGR) of 9.1% from 2025 through 2033. This significant growth is propelled by a confluence of factors, including the increasing prevalence of autoimmune diseases globally, which necessitates more accurate and timely diagnostic solutions. Advancements in diagnostic technologies, such as improved immunoassay platforms and molecular diagnostics, are further fueling market demand by offering enhanced sensitivity and specificity. Furthermore, a growing awareness among healthcare professionals and patients about the early detection and management of autoimmune disorders contributes to the rising adoption of sophisticated diagnostic reagents. The market is also benefiting from increased healthcare expenditure in developing economies and supportive government initiatives aimed at improving diagnostic infrastructure and access to advanced medical testing.
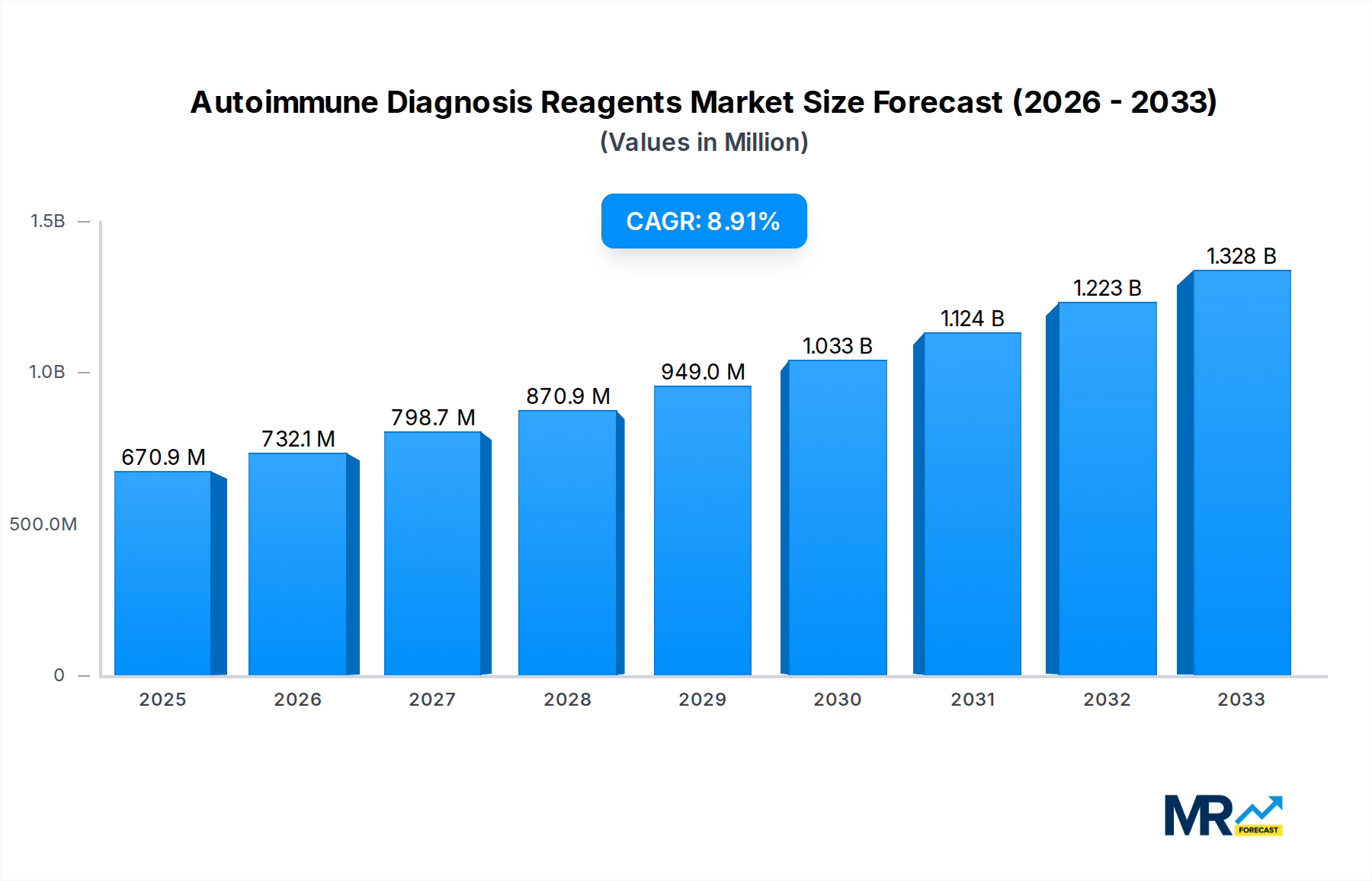

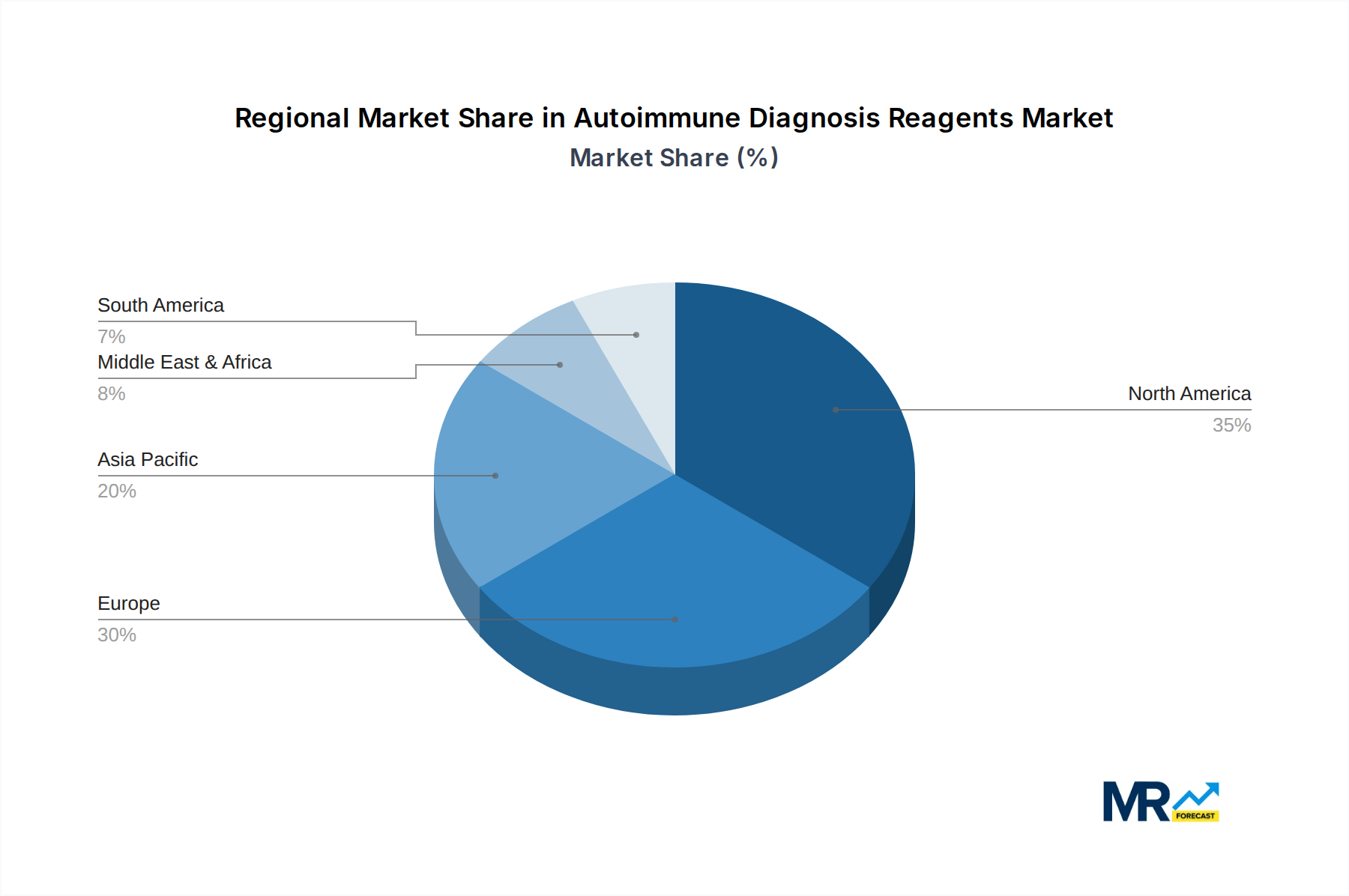
The competitive landscape features prominent players like Thermo Scientific Phadia, Siemens Healthineers, and EUROIMMUN, driving innovation and market penetration through strategic partnerships and product development. The market segments, primarily Hospitals and Third-party Testing Agencies, represent key end-user bases, with hospitals increasingly investing in in-house diagnostic capabilities and third-party agencies expanding their service offerings to meet the growing demand for specialized autoimmune testing. Geographically, North America and Europe are anticipated to remain dominant markets due to well-established healthcare systems and high adoption rates of advanced diagnostics. However, the Asia Pacific region is expected to witness the fastest growth, driven by a burgeoning patient population, increasing disposable incomes, and a growing focus on preventive healthcare and early disease detection. Emerging trends include the development of multiplex assays for simultaneous detection of multiple autoimmune markers and the integration of artificial intelligence in diagnostic interpretation.

Here's a report description on Autoimmune Diagnosis Reagents, incorporating your specified elements:
The autoimmune diagnosis reagents market is on a robust upward trajectory, driven by an increasing prevalence of autoimmune diseases globally and advancements in diagnostic technologies. The market, valued at an estimated USD 3,800 million in the base year 2025, is projected to witness a Compound Annual Growth Rate (CAGR) of approximately 7.5% throughout the forecast period of 2025-2033, reaching an impressive USD 6,800 million by the end of the study period. This significant expansion is underpinned by a growing awareness of autoimmune conditions, leading to earlier and more accurate diagnoses. The increasing demand for high-throughput screening and multiplexed testing platforms is also a key trend, allowing for the simultaneous detection of multiple autoantibodies. Technological innovations, such as the development of more sensitive and specific immunoassay platforms, including ELISA, chemiluminescence immunoassays (CLIA), and multiplex bead arrays, are further fueling market growth. These advancements not only improve diagnostic accuracy but also reduce turnaround times, which is crucial for effective patient management. The rising disposable income in emerging economies is also contributing to increased healthcare expenditure, making advanced diagnostic tools more accessible. Furthermore, the growing emphasis on personalized medicine and the need to identify specific autoantibody profiles for targeted therapies are creating new avenues for market expansion. The integration of artificial intelligence (AI) and machine learning (ML) in interpreting complex autoimmune diagnostics is also an emerging trend that promises to enhance diagnostic efficiency and predictive capabilities.
The autoimmune diagnosis reagents market is propelled by a confluence of powerful forces, with the escalating global burden of autoimmune diseases standing as a primary driver. Conditions such as rheumatoid arthritis, lupus, multiple sclerosis, and type 1 diabetes are becoming increasingly common, necessitating more sophisticated and accessible diagnostic tools. This growing prevalence directly translates into a higher demand for the reagents required for their detection. Concurrently, significant technological advancements in diagnostic methodologies are reshaping the market landscape. The development of highly sensitive and specific immunoassay platforms, including Enzyme-Linked Immunosorbent Assay (ELISA), Chemiluminescence Immunoassay (CLIA), and multiplex bead array technologies, allows for the simultaneous detection of multiple autoantibodies with improved accuracy and speed. This enables earlier diagnosis, which is critical for effective management and improved patient outcomes. Furthermore, the increasing global healthcare expenditure and the rising disposable income in various regions are making these advanced diagnostic solutions more accessible to a wider population. This accessibility, coupled with a growing awareness among healthcare professionals and patients about the importance of early and accurate diagnosis, creates a robust demand for autoimmune diagnosis reagents, fostering continuous market expansion.
Despite the promising growth, the autoimmune diagnosis reagents market faces several challenges and restraints that can temper its expansion. A significant hurdle is the high cost associated with developing and validating novel diagnostic assays, particularly those involving complex autoantibody panels. The initial investment in research and development, coupled with stringent regulatory approval processes, can be a substantial barrier for smaller players and can slow down the introduction of new products. Another challenge is the complexity and heterogeneity of autoimmune diseases themselves. Many autoimmune conditions share overlapping symptoms and autoantibody profiles, making accurate differentiation and diagnosis a complex undertaking. This necessitates the development of highly specific and discriminative assays, which can be technically challenging and expensive to produce. Furthermore, the lack of standardized diagnostic protocols across different regions and healthcare systems can lead to variability in testing and interpretation, hindering widespread adoption of certain reagents. The limited availability of skilled laboratory professionals trained in interpreting complex autoimmune test results in certain geographical areas also poses a restraint. Finally, reimbursement policies for autoimmune diagnostic tests can vary significantly, impacting the economic viability for healthcare providers and potentially limiting patient access to advanced testing.
The Hospital segment, particularly within North America, is poised to dominate the autoimmune diagnosis reagents market. This dominance is a result of a synergistic interplay of factors, including a high prevalence of autoimmune diseases, substantial healthcare expenditure, and a well-established healthcare infrastructure. North America, encompassing the United States and Canada, consistently demonstrates a high incidence of autoimmune disorders, such as rheumatoid arthritis, lupus, and multiple sclerosis. This demographic reality translates directly into a significant and sustained demand for diagnostic reagents. The robust healthcare systems in these countries are characterized by extensive hospital networks, advanced diagnostic laboratories, and a high adoption rate of cutting-edge medical technologies. Hospitals, being the primary point of care for many chronic and complex conditions, are at the forefront of autoimmune disease diagnosis. They possess the necessary infrastructure, skilled personnel, and financial resources to invest in and utilize a wide array of autoimmune diagnosis reagents.
Within the hospital setting, the demand for reagents is further amplified by:
Moreover, the strong reimbursement landscape for diagnostic procedures in North America, coupled with a high per capita income, enables hospitals to procure and utilize a broad spectrum of high-quality autoimmune diagnosis reagents. This financial capacity ensures that hospitals can offer advanced diagnostic services, thereby solidifying their position as the leading segment and region for market dominance.
The autoimmune diagnosis reagents industry is experiencing significant growth catalysts. The ever-increasing global incidence and prevalence of autoimmune diseases are creating a fundamental demand for diagnostic solutions. Advancements in diagnostic technologies, leading to more sensitive, specific, and multiplexed assays, are enhancing diagnostic accuracy and efficiency. The growing awareness among healthcare professionals and patients regarding the importance of early and precise diagnosis is a key driver. Furthermore, rising healthcare expenditures, particularly in emerging economies, coupled with favorable reimbursement policies for diagnostic tests, are making these reagents more accessible.
This comprehensive report delves into the intricate landscape of the autoimmune diagnosis reagents market, meticulously analyzing trends from the historical period of 2019-2024 and projecting future growth through the forecast period of 2025-2033, with a base year of 2025. It offers an in-depth exploration of the driving forces, meticulously examining the escalating prevalence of autoimmune diseases and significant technological advancements in diagnostic methodologies. Conversely, it also scrutinizes the challenges and restraints, including the high costs of R&D, regulatory hurdles, and the inherent complexity of autoimmune conditions. The report provides a granular segmentation analysis, highlighting the dominance of the Hospital application segment, particularly within North America, due to its robust healthcare infrastructure and high disease burden. Furthermore, it identifies key growth catalysts, such as increasing healthcare expenditure and rising awareness, and profiles the leading global players in the market. Finally, the report details significant developments with specific timelines, offering valuable insights for stakeholders seeking to navigate and capitalize on this dynamic and expanding market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 9.1% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 9.1%.
Key companies in the market include Thermo Scientific Phadia, Dr. Fooke Laboratorien, Siemens Healthineers, EUROIMMUN, HOB Biotech, SDR Diagnostics, Shenzhen YHLO, Werfen, Kexin Biotech, HUMAN, AESKU, Bio-Rad, .
The market segments include Application.
The market size is estimated to be USD 670.9 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million and volume, measured in K.
Yes, the market keyword associated with the report is "Autoimmune Diagnosis Reagents," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Autoimmune Diagnosis Reagents, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.