1. What is the projected Compound Annual Growth Rate (CAGR) of the Anti-TIGIT Antibody?
The projected CAGR is approximately 13.0%.
 Anti-TIGIT Antibody
Anti-TIGIT AntibodyAnti-TIGIT Antibody by Type (Monotherapy, Combination Therapy), by Application (Cell Carcinoma, Solid Tumor), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
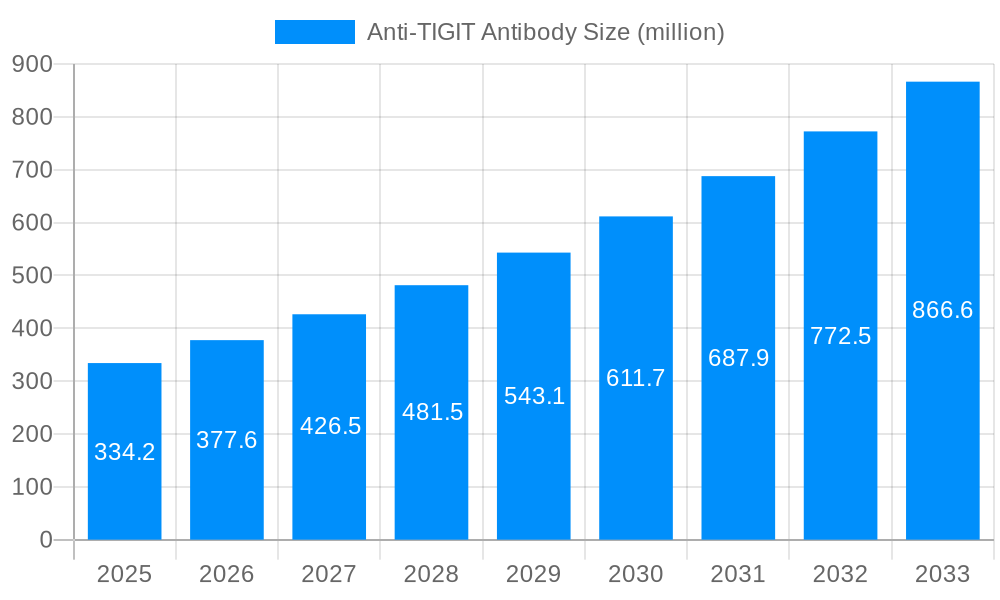
The global Anti-TIGIT Antibody market is poised for significant expansion, projected to reach approximately $334.2 million by 2025, driven by an impressive Compound Annual Growth Rate (CAGR) of 13.0% throughout the forecast period of 2025-2033. This robust growth is primarily fueled by the escalating prevalence of various cancers, particularly cell carcinoma and solid tumors, which represent the key application segments. The increasing understanding of TIGIT's role in immune evasion by cancer cells is spurring intense research and development efforts, leading to the advancement of novel therapeutic strategies. Monotherapy treatments are expected to remain a significant segment, while combination therapies, leveraging the synergistic effects of Anti-TIGIT antibodies with other immunotherapies like checkpoint inhibitors, are rapidly gaining traction and are anticipated to become a dominant force in the market. The growing pipeline of Anti-TIGIT antibody candidates and ongoing clinical trials further underscore the market's positive trajectory.
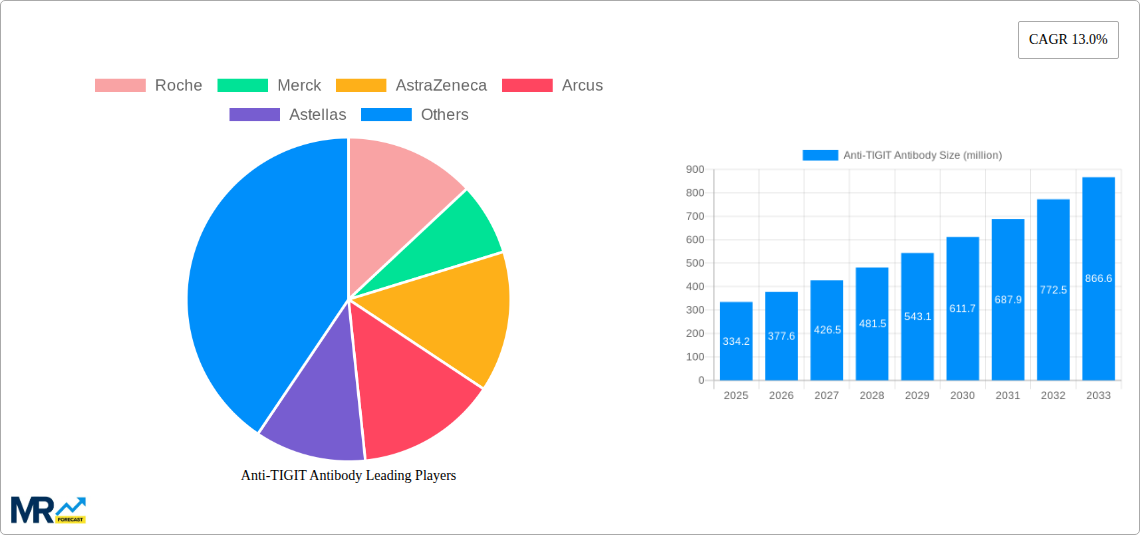

The market's dynamism is further shaped by the active participation of leading pharmaceutical and biotechnology companies, including Roche, Merck, AstraZeneca, and BeiGene, among others, who are investing heavily in the development and commercialization of these promising cancer immunotherapies. Geographically, North America and Europe are anticipated to hold substantial market shares due to advanced healthcare infrastructure, high cancer incidence rates, and significant R&D investments. However, the Asia Pacific region, particularly China and India, is expected to exhibit the fastest growth, driven by increasing healthcare expenditure, a rising cancer burden, and expanding access to innovative treatments. While the market presents substantial opportunities, challenges such as the high cost of development and treatment, stringent regulatory pathways, and the need for extensive clinical validation could temper the pace of market penetration in certain segments. Nevertheless, the compelling clinical data emerging from ongoing trials and the potential for significant patient benefit are expected to drive sustained market growth and innovation in the Anti-TIGIT antibody landscape.

This comprehensive report offers an in-depth analysis of the Anti-TIGIT Antibody market, providing critical insights into its current trajectory and future potential. The study covers a significant period, spanning the Study Period: 2019-2033, with a Base Year: 2025 and Estimated Year: 2025. The Forecast Period: 2025-2033 will meticulously detail anticipated market shifts, building upon a robust Historical Period: 2019-2024. Our analysis delves into the intricate dynamics of this rapidly evolving sector, utilizing a projected market value in the millions of units.
The Anti-TIGIT antibody landscape is experiencing a dynamic surge, driven by increasing recognition of TIGIT as a crucial immune checkpoint target in oncology. This evolving understanding is translating into a robust pipeline of clinical candidates and an intensifying competitive environment. We observe a significant trend towards Combination Therapy as a primary strategy, moving beyond monotherapy applications. The rationale behind this approach is the synergistic potential of targeting multiple immune checkpoints to overcome tumor resistance and enhance anti-tumor immunity. This is particularly evident in the development of novel drug combinations incorporating Anti-TIGIT antibodies with PD-1/PD-L1 inhibitors and other immunotherapies. Furthermore, there's a clear upward trajectory in the exploration of Anti-TIGIT antibodies for a broad spectrum of Solid Tumors, demonstrating their potential as a versatile therapeutic agent. The market is also witnessing a growing emphasis on biomarker discovery to identify patient populations most likely to respond to TIGIT inhibition, a critical step towards personalized medicine. The regulatory landscape, while still maturing for TIGIT-targeted therapies, is showing signs of acceleration with ongoing discussions and potential pathways for accelerated approvals based on promising clinical data. This proactive approach by regulatory bodies underscores the perceived clinical benefit and unmet medical need that Anti-TIGIT antibodies aim to address. The increasing investment in research and development, coupled with strategic partnerships between established pharmaceutical giants and emerging biotech firms, further fuels this positive trend. The potential for significant revenue generation, projected to reach millions of units, highlights the immense commercial promise of this therapeutic class. The iterative nature of clinical development and the ongoing scientific discourse surrounding TIGIT's mechanism of action are continuously shaping the market, leading to innovative approaches and refined therapeutic strategies.
The burgeoning growth of the Anti-TIGIT antibody market is fundamentally propelled by the unmet medical need in oncology, particularly for patients who are refractory to or have relapsed from existing immunotherapies. TIGIT (T cell immunoglobulin and ITM and immunoglobulin domain-containing protein) has emerged as a compelling target due to its role in modulating T cell exhaustion and promoting tumor immune evasion. Research consistently demonstrates that TIGIT acts as an inhibitory receptor on various immune cells, including T cells and NK cells, and its blockade can unleash these cells to mount a more potent anti-tumor response. The synergistic potential of combining Anti-TIGIT antibodies with established immunotherapies, such as PD-1 and PD-L1 inhibitors, represents a significant driving force. This combination approach aims to overcome resistance mechanisms that often limit the efficacy of monotherapy treatments, thereby expanding the patient population that can benefit from immunotherapy. Furthermore, the increasing understanding of the complex tumor microenvironment and the diverse roles of immune checkpoints is fueling further investigation and development of TIGIT-targeted therapies. The substantial investments being channeled into clinical trials and the progress observed in late-stage studies are also acting as powerful catalysts, instilling confidence in the therapeutic potential of these antibodies. This confluence of scientific discovery, clinical validation, and strategic investment is creating a fertile ground for the rapid expansion of the Anti-TIGIT antibody market, offering a beacon of hope for improved cancer treatment outcomes.
Despite the promising outlook, the Anti-TIGIT antibody market is not without its hurdles. A primary challenge lies in elucidating the precise role and optimal combination strategies for TIGIT inhibition. While promising, the scientific community is still actively defining the nuanced mechanisms by which TIGIT exerts its immune-regulatory effects across different tumor types and in combination with other agents. This complexity can lead to clinical trial designs that may not fully capture the therapeutic potential. The development of effective and reliable predictive biomarkers remains a significant restraint. Identifying patient populations that are most likely to respond to Anti-TIGIT antibody therapy is crucial for optimizing treatment efficacy and avoiding unnecessary toxicity. The current lack of universally validated biomarkers can lead to suboptimal patient selection and potentially disappointing clinical trial outcomes. Furthermore, the high cost associated with developing novel biologics, including extensive preclinical testing and multi-phase clinical trials, poses a significant financial barrier. This can also translate to higher treatment costs for patients, potentially impacting market accessibility and adoption. The competitive landscape, with numerous companies vying for market share, while ultimately beneficial for innovation, can also lead to a crowded pipeline, making it challenging for individual therapies to gain significant traction without robust clinical differentiation. Finally, the potential for off-target effects or immune-related adverse events associated with targeting immune checkpoints necessitates rigorous safety monitoring and management, which can add complexity to clinical practice.
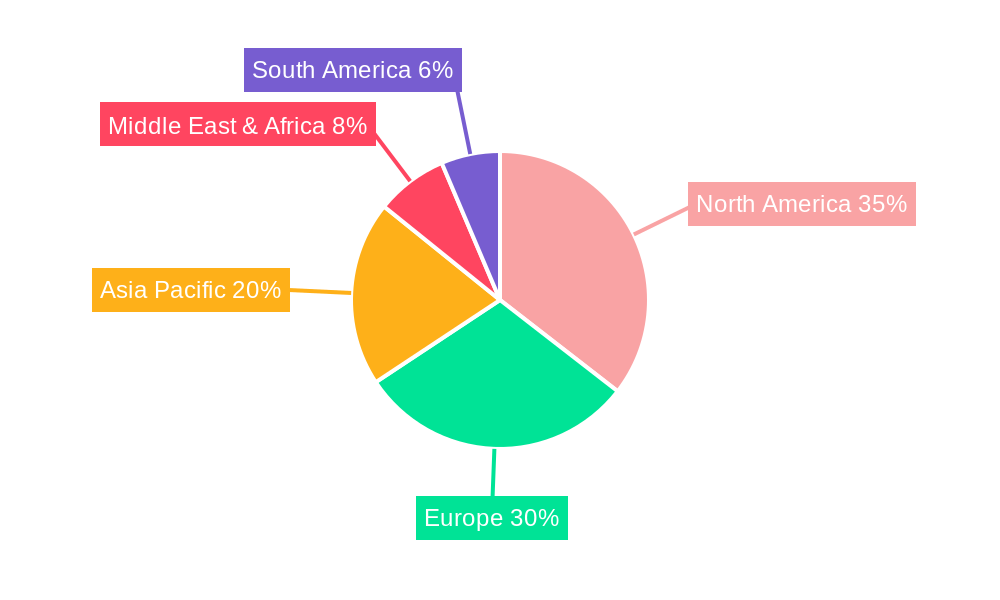
The global Anti-TIGIT antibody market is poised for substantial growth, with a discernible concentration of activity and anticipated dominance in North America and the Asia Pacific region. This dominance is intricately linked to specific market segments, particularly the Combination Therapy application and its widespread use in treating Solid Tumors.
In North America, particularly the United States, the market's leading position is underpinned by several factors. Firstly, the region boasts a highly developed and robust healthcare infrastructure, with a high concentration of leading research institutions and academic medical centers. This ecosystem fosters cutting-edge research and clinical development, attracting substantial investment from both public and private sectors. The well-established pharmaceutical industry in North America, with giants like Roche, Merck, and AstraZeneca, actively involved in TIGIT research, provides a strong foundation for pipeline development and commercialization. Furthermore, the region's advanced regulatory framework, while stringent, is also conducive to the approval of novel therapies, especially those addressing significant unmet medical needs like cancer. The payer landscape in North America is generally more amenable to adopting innovative and potentially expensive treatments, provided there is clear evidence of clinical benefit.
The Asia Pacific region, driven significantly by China, is emerging as a formidable player and is projected to witness the fastest growth. This surge is fueled by a rapidly expanding biopharmaceutical industry, characterized by significant investments in research and development and a growing number of domestic players such as BeiGene, Junshi Biosciences, and Innovent Biologics. The sheer volume of the patient population suffering from various cancers, particularly Solid Tumors like lung cancer and gastric cancer, creates a substantial market demand. Government initiatives aimed at promoting innovation in the healthcare sector and supportive regulatory reforms are further accelerating the development and adoption of new therapies. The increasing disposable income and growing access to advanced healthcare services in countries like China are also contributing to the market's expansion. Moreover, the focus on Combination Therapy is particularly pronounced in this region, driven by the desire to overcome treatment resistance and achieve better patient outcomes in common cancers.
When considering specific segments:
The Anti-TIGIT antibody industry is experiencing robust growth fueled by several key catalysts. The increasing scientific understanding of TIGIT's role in immune suppression and tumor evasion is paramount. This knowledge is directly translating into a wave of promising clinical trials, particularly in Combination Therapy settings, which offer the potential for enhanced efficacy. Furthermore, the growing global burden of cancer, especially Solid Tumors, creates a persistent demand for novel and more effective treatment options. Strategic collaborations between established pharmaceutical giants and innovative biotech companies are accelerating R&D and clinical development timelines. Finally, a supportive regulatory environment for novel oncology therapeutics and increasing investment from venture capital are injecting significant capital into the sector.
This report provides a panoramic view of the Anti-TIGIT antibody market, meticulously analyzing market size, segmentation, and growth projections across the Study Period: 2019-2033. We offer deep dives into key market dynamics, including the pivotal shift towards Combination Therapy and the expansive applications in Solid Tumors. Our analysis further dissects the driving forces, challenges, and restraints, providing a balanced perspective on the industry's trajectory. Crucially, the report identifies dominant regions and countries, alongside specific segments poised for significant market share, offering strategic insights for stakeholders. The report is enriched with an exhaustive list of leading players and a chronological breakdown of significant industry developments, ensuring a comprehensive understanding of the competitive landscape and innovation pipeline. This report is an indispensable resource for pharmaceutical companies, biotech firms, investors, and research institutions seeking to navigate and capitalize on the evolving Anti-TIGIT antibody market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 13.0% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 13.0%.
Key companies in the market include Roche, Merck, AstraZeneca, Arcus, Astellas, BeiGene, BMS, Junshi Biosciences, Innovent Biologics, Mereo BioPharma, Seattle Genetics, Hengrui Medicine, Compugen, Gilead Sciences, .
The market segments include Type, Application.
The market size is estimated to be USD 334.2 million as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in million.
Yes, the market keyword associated with the report is "Anti-TIGIT Antibody," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Anti-TIGIT Antibody, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.