1. What is the projected Compound Annual Growth Rate (CAGR) of the AAV Manufacturing Service?
The projected CAGR is approximately 15.6%.
 AAV Manufacturing Service
AAV Manufacturing ServiceAAV Manufacturing Service by Type (Adherent Culture, Suspension Culture), by Application (Vaccine Development, Gene Therapy, Academic Scientific Research Institution, Others), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The AAV manufacturing service market is experiencing robust growth, projected to reach approximately $3.8 billion in value by 2025. This expansion is fueled by a compelling CAGR of 15.6%, indicating a dynamic and rapidly evolving industry. The increasing prevalence of gene therapies and the critical role of Adeno-Associated Viruses (AAVs) as delivery vectors for these groundbreaking treatments are primary drivers. The demand for AAV manufacturing services is also significantly boosted by vaccine development initiatives, particularly in light of recent global health events, and the persistent need for advanced solutions in academic scientific research. These factors are creating a substantial market opportunity for specialized AAV manufacturing service providers.
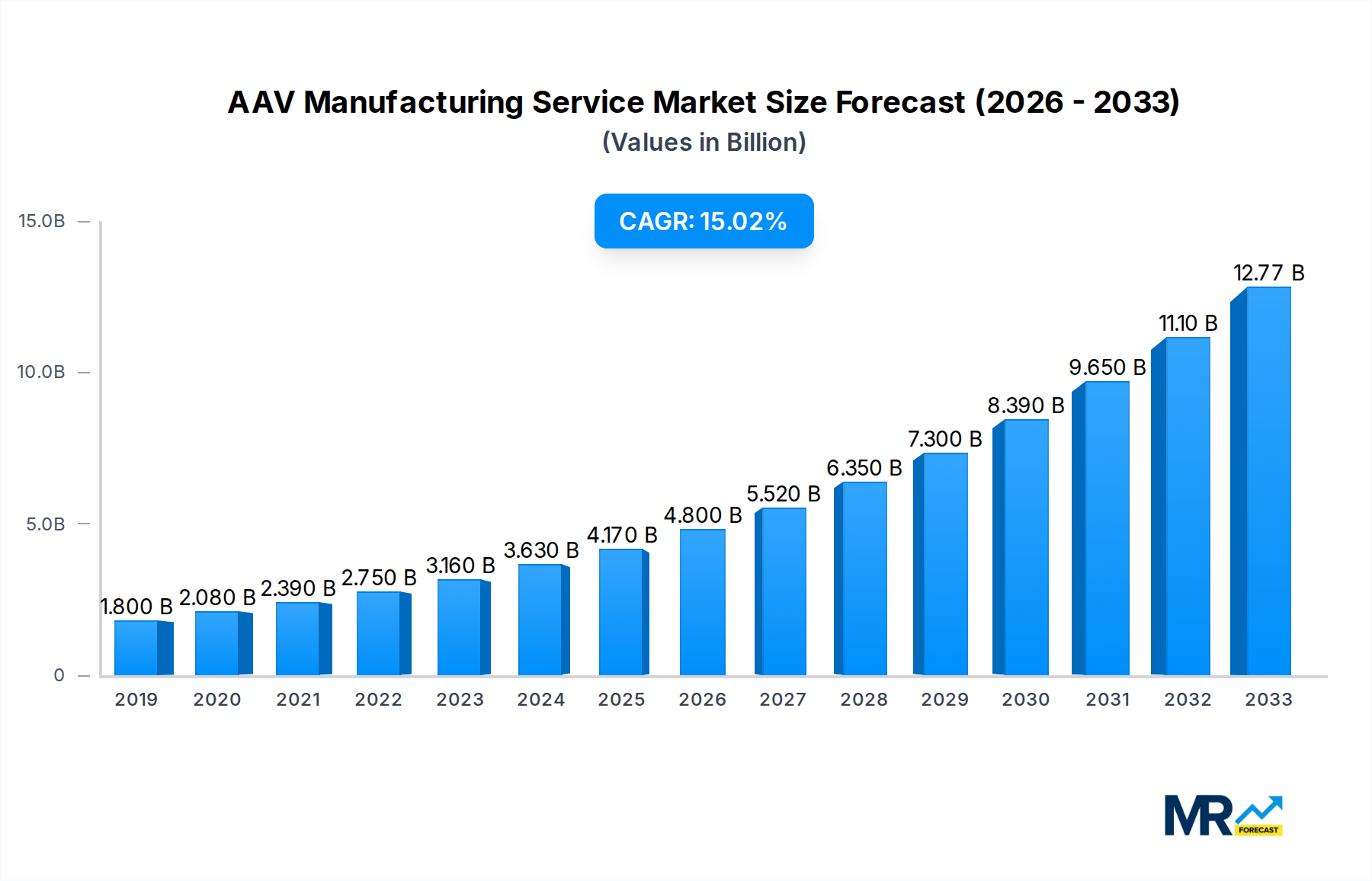

The market's trajectory is further shaped by several key trends and strategic considerations. The shift towards adherent and suspension culture methods reflects advancements in bioprocessing technologies aimed at improving yield and efficiency. Major players, including Thermo Fisher Scientific, Lonza, and Catalent, alongside emerging specialists, are investing heavily in expanding their capacities and enhancing their technological offerings. While the market is poised for significant expansion, certain restraints, such as the complex regulatory landscape and the high cost of specialized manufacturing facilities, need careful navigation. However, the sustained innovation in gene therapy and the growing pipeline of AAV-based therapeutics are expected to outweigh these challenges, ensuring continued market momentum and a bright outlook for AAV manufacturing services.

Here's a unique report description for AAV Manufacturing Services, incorporating your specific requirements:
The Adeno-Associated Virus (AAV) manufacturing service market is experiencing an unprecedented surge, projected to reach figures well into the billions by 2025 and beyond. This explosive growth is fundamentally reshaping the landscape of advanced therapeutics and diagnostics. Our comprehensive report delves deep into the intricate dynamics of this burgeoning sector, offering an unparalleled view of market trends from 2019 through to a projected 2033. We will analyze the pivotal shift from historical CAGR figures to a robust base year of 2025, followed by a detailed forecast for the period 2025-2033. Key insights will highlight the increasing demand for high-quality, scalable AAV production to meet the ever-expanding pipeline of gene therapies and advanced vaccines. The report will meticulously dissect the evolution of manufacturing platforms, from early-stage academic research to large-scale commercial production, reflecting significant advancements in both adherent and suspension culture techniques. Furthermore, we will explore the critical role of AAVs in addressing previously intractable genetic diseases, driving substantial investment and innovation. The market's trajectory is marked by a growing emphasis on regulatory compliance, cost-efficiency, and the development of novel AAV serotypes optimized for specific therapeutic applications. The interplay between academic institutions, biotech startups, and established pharmaceutical giants is fostering a collaborative ecosystem, accelerating the translation of research into life-saving treatments. Understanding these intricate trends is paramount for stakeholders seeking to navigate and capitalize on the immense opportunities within the AAV manufacturing services sector.
The remarkable ascent of the AAV manufacturing service market is underpinned by a confluence of powerful driving forces. Foremost among these is the explosive progress and increasing clinical success of gene therapies. As more gene therapy candidates demonstrate efficacy and gain regulatory approval for a diverse array of genetic disorders, the demand for reliable, large-scale AAV vector production has skyrocketed. This surge is directly translating into billions in market value. Furthermore, the growing understanding of AAV's inherent safety profile and its ability to deliver genetic material to a wide range of tissues and cell types makes it an ideal delivery vehicle for both therapeutic and prophylactic applications, including the development of next-generation vaccines. The expansion of research into rare genetic diseases, where gene therapy offers a potential cure rather than symptom management, is another significant propellant. This intensified research effort necessitates advanced manufacturing capabilities to produce the required quantities of high-purity AAV vectors. The increasing investment from venture capital and established pharmaceutical companies into the gene therapy space, recognizing its transformative potential, further fuels the demand for robust and scalable AAV manufacturing services, pushing the market into multi-billion dollar valuations.
Despite its impressive growth trajectory, the AAV manufacturing service sector faces several significant challenges and restraints that warrant careful consideration. One of the primary hurdles is the inherent complexity and cost associated with producing high-quality, clinical-grade AAV vectors. Achieving sufficient titers, purity, and consistent batch-to-batch reproducibility on a large scale remains a significant technical challenge. The intricate upstream and downstream processing required for AAV production demands specialized expertise and advanced infrastructure, leading to substantial capital investment and operational costs. Regulatory hurdles also present a considerable restraint. Navigating the stringent requirements of global regulatory bodies for gene therapy products, including comprehensive characterization and safety testing of AAV vectors, can be time-consuming and costly. Furthermore, the limited availability of skilled personnel with expertise in viral vector manufacturing and bioprocessing can create bottlenecks. While the market is valued in the billions, these challenges can limit the speed of development and commercialization for some therapies. The scalability of current manufacturing platforms to meet the potentially massive demand for blockbuster gene therapies is another area of concern, necessitating continuous innovation in process development and optimization to ensure cost-effectiveness and accessibility.
The AAV manufacturing service market is poised for significant dominance by specific regions and segments, driven by innovation, investment, and therapeutic application.
Dominant Segments:
Gene Therapy: This segment is unequivocally the primary driver and will continue to dominate the AAV manufacturing service market. The burgeoning pipeline of gene therapies targeting a vast spectrum of genetic disorders, from rare inherited diseases to more prevalent conditions like certain cancers and neurological disorders, directly translates into an insatiable demand for AAV vectors. The successful clinical translation and increasing market approvals of gene therapies, often leveraging AAV as the preferred delivery vehicle, solidify its position. The projected multi-billion dollar valuations are inextricably linked to the success and expansion of this application.
Vaccine Development: While currently a significant, but secondary, application, vaccine development holds immense future potential for AAV manufacturing services. The exploration of AAV as a platform for vaccine delivery, particularly for infectious diseases and cancer vaccines, is gaining significant traction. As research progresses and clinical trials demonstrate efficacy, this segment is expected to experience exponential growth, contributing substantially to the overall market.
Dominant Regions/Countries:
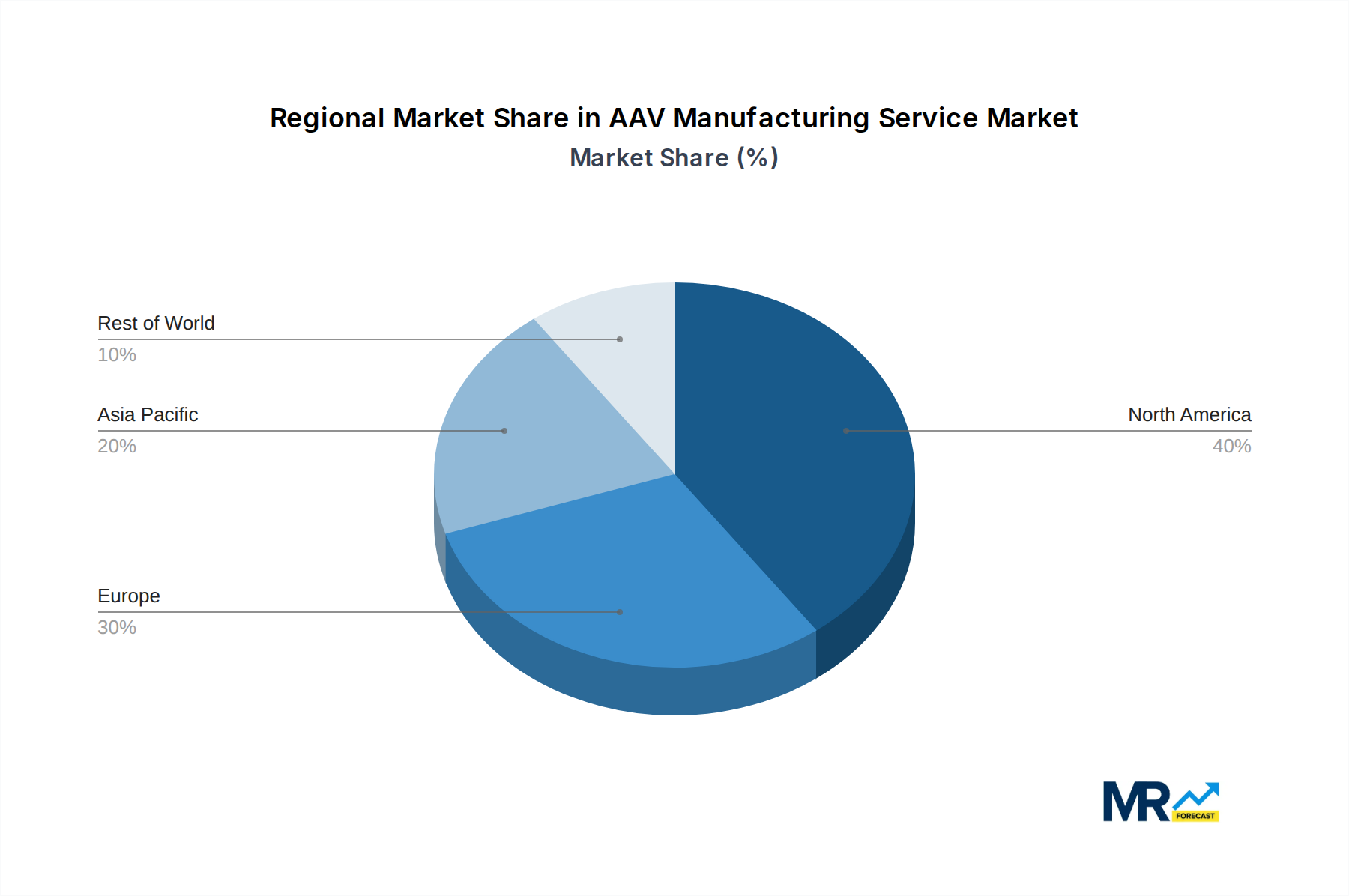
North America (United States): The United States stands as the undisputed leader in the AAV manufacturing service market, driven by a robust ecosystem of innovative biotech and pharmaceutical companies, substantial venture capital funding, and a highly supportive regulatory environment for gene therapies. The presence of leading academic research institutions and a concentration of contract development and manufacturing organizations (CDMOs) specializing in viral vector production further solidifies its dominance. The country is home to a significant portion of the global gene therapy clinical trials and commercialized products, necessitating a vast and sophisticated AAV manufacturing infrastructure, contributing billions to the global market.
Europe (especially UK, Germany, Switzerland): Europe, with a strong emphasis on scientific research and a growing number of gene therapy approvals, presents a significant market for AAV manufacturing services. Countries like the United Kingdom, Germany, and Switzerland are at the forefront, boasting advanced research capabilities and a growing number of CDMOs specializing in viral vector production. The increasing investment in biopharmaceutical innovation and supportive government initiatives are fostering the growth of this sector, making it a critical contributor to the multi-billion dollar market.
Asia-Pacific (especially China and Japan): The Asia-Pacific region, particularly China and Japan, is emerging as a rapidly growing market for AAV manufacturing services. Driven by increasing government support for the biopharmaceutical industry, a burgeoning number of research institutions, and a growing domestic demand for advanced therapeutics, these countries are witnessing significant investments in AAV manufacturing capabilities. While currently smaller in market share compared to North America and Europe, the rapid pace of development and the potential for large patient populations position this region for substantial future growth, contributing billions to the global market over the forecast period. The focus on expanding domestic manufacturing capacity and attracting global players further fuels this expansion.
The dominance of these regions and segments is a testament to the intense scientific innovation, strategic investments, and the critical role AAV manufacturing plays in enabling the next generation of life-saving treatments. The interplay between these factors is creating a dynamic and rapidly expanding multi-billion dollar market.
The AAV manufacturing service industry is experiencing significant growth catalysts that are propelling its expansion into multi-billion dollar valuations. The accelerating pace of gene therapy approvals and clinical advancements for various genetic disorders is a primary driver, creating an unprecedented demand for reliable and scalable AAV production. Furthermore, the expanding applications of AAVs beyond rare diseases, including in areas like neurodegenerative disorders and even oncology, are opening new avenues for market growth. Continuous technological advancements in process development, such as the optimization of suspension culture methods and the development of more efficient downstream purification techniques, are enhancing scalability and reducing manufacturing costs, thereby making AAV therapies more accessible. The increasing number of strategic partnerships and collaborations between CDMOs and pharmaceutical companies, alongside substantial investments from venture capital firms, are also fueling innovation and capacity expansion.
This comprehensive report offers an in-depth analysis of the AAV Manufacturing Service market, encompassing critical trends, driving forces, and challenges. It meticulously details the market’s projected growth into multi-billion dollar valuations from the historical period 2019-2024, through the base year of 2025, and extending to a robust forecast period of 2025-2033. The report highlights the key regions and dominant market segments, such as Gene Therapy, that are shaping the industry's trajectory. Furthermore, it identifies the leading players and significant developments that have occurred within the sector. The growth catalysts, including technological advancements and increasing therapeutic applications, are thoroughly examined, providing stakeholders with actionable insights to navigate this dynamic and rapidly evolving market.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 15.6% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 15.6%.
Key companies in the market include Thermo Fisher Scientific, Lonza, Catalent, Charles River, Merck, SK pharmteco, Oxford Biomedica, AGC Biologics(MolMed), Gene Universal, Polyplus (Sartorius), CCRM, FUJIFILM Diosynth Biotechnologies, Biovian, Miltenyi Bioindustry, Aldevron, Takara Bio, Yposkesi, Genezen, Exthera, GenScript ProBio, EurekaBio, Obio Technology, GeneSail Biotech, VectorBuilder, WuXi ATU, Ubrigene, Porton Biologics, Pharmaron, PackGene Biotechnology, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "AAV Manufacturing Service," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the AAV Manufacturing Service, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.