1. What is the projected Compound Annual Growth Rate (CAGR) of the Pharmaceutical Quality Control Market?
The projected CAGR is approximately XXX%.
 Pharmaceutical Quality Control Market
Pharmaceutical Quality Control MarketPharmaceutical Quality Control Market by Product Type (Reagents & Consumables, Instruments, Services), by Analysis Type (Physical Testing, Chemical Testing, Microbial Testing), by End-user (Pharmaceutical Companies, Contract Research Organizations, Others), by North America (U.S., Canada, Mexico), by Europe (UK, Germany, France, Italy, Spain, Russia, Netherlands, Switzerland, Poland, Sweden, Belgium), by Asia Pacific (China, India, Japan, South Korea, Australia, Singapore, Malaysia, Indonesia, Thailand, Philippines, New Zealand), by Latin America (Brazil, Mexico, Argentina, Chile, Colombia, Peru), by MEA (UAE, Saudi Arabia, South Africa, Egypt, Turkey, Israel, Nigeria, Kenya) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
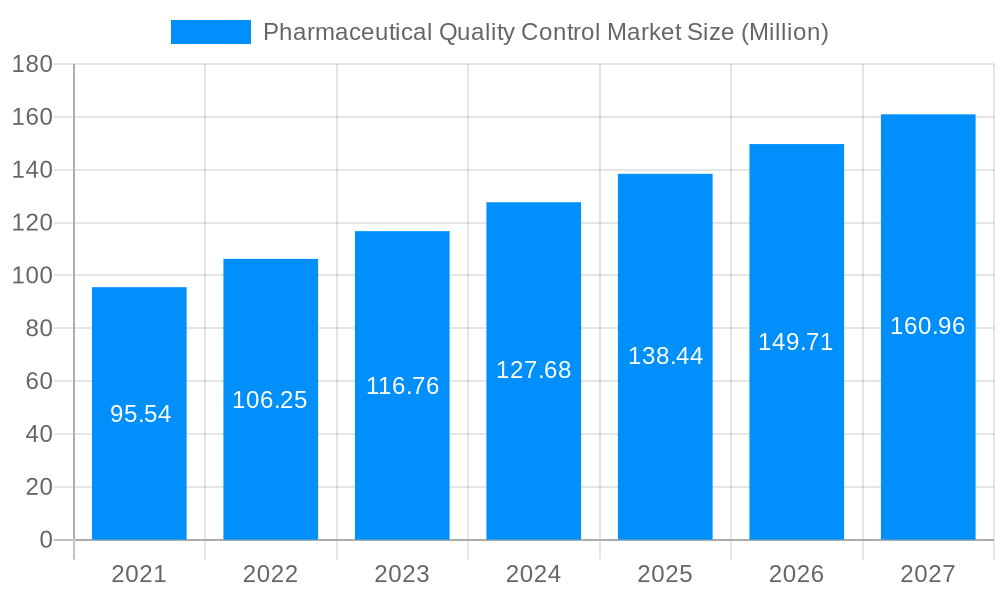
The size of the Pharmaceutical Quality Control Market was valued at USD XX Million in 2023 and is projected to reach USD XXX Million by 2032, with an expected CAGR of XXX% during the forecast period. Pharmaceutical Quality Control (QC) refers to the processes and systems used to ensure that pharmaceutical products meet the required quality standards. These standards cover the purity, strength, consistency, stability, and safety of pharmaceutical products, ensuring that they are effective and safe for patient use. QC is a crucial part of the overall quality assurance system, which also includes practices related to the development, manufacturing, packaging, and distribution of pharmaceuticals.

The Pharmaceutical Quality Control Market is experiencing a dynamic evolution driven by several interconnected trends. These factors are shaping the strategies of manufacturers, contract research organizations (CROs), and regulatory bodies, ultimately impacting the safety and efficacy of pharmaceutical products. Key trends include:

Despite the promising growth, the Pharmaceutical Quality Control Market faces several significant hurdles that can impact its expansion and operational efficiency. Key challenges and restraints include:
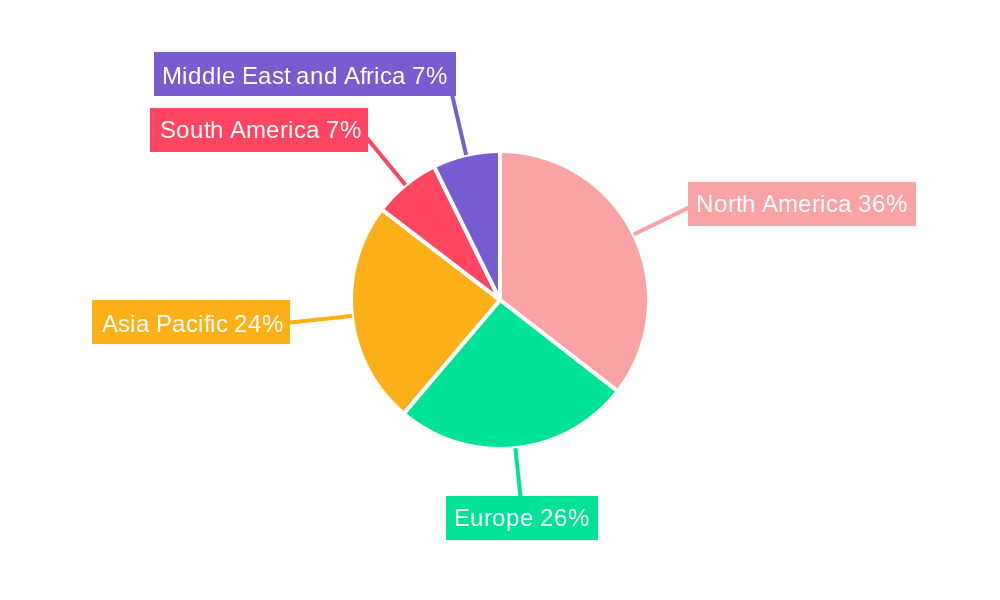
The global Pharmaceutical Quality Control Market is characterized by distinct regional strengths and growth potentials. Understanding these dynamics is crucial for market participants.
Pricing in the Pharmaceutical Quality Control Market can vary depending on factors such as the type of testing, the complexity of the analysis, the volume of samples, and the reputation of the laboratory. The market is characterized by a competitive pricing environment, with a range of pricing models used by different laboratories.
The import and export of pharmaceutical quality control services and equipment can vary significantly depending on the region and country. Some countries may have import restrictions or export controls on pharmaceutical products and related services, impacting the trade of pharmaceutical quality control-related goods.
Patents and trademarks play a role in protecting innovations and intellectual property in the Pharmaceutical Quality Control Market. Patent analysis can provide insights into the technological advancements and competitive landscape, while trademark analysis can help identify the leading brands and market share of different players.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of XXX% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately XXX%.
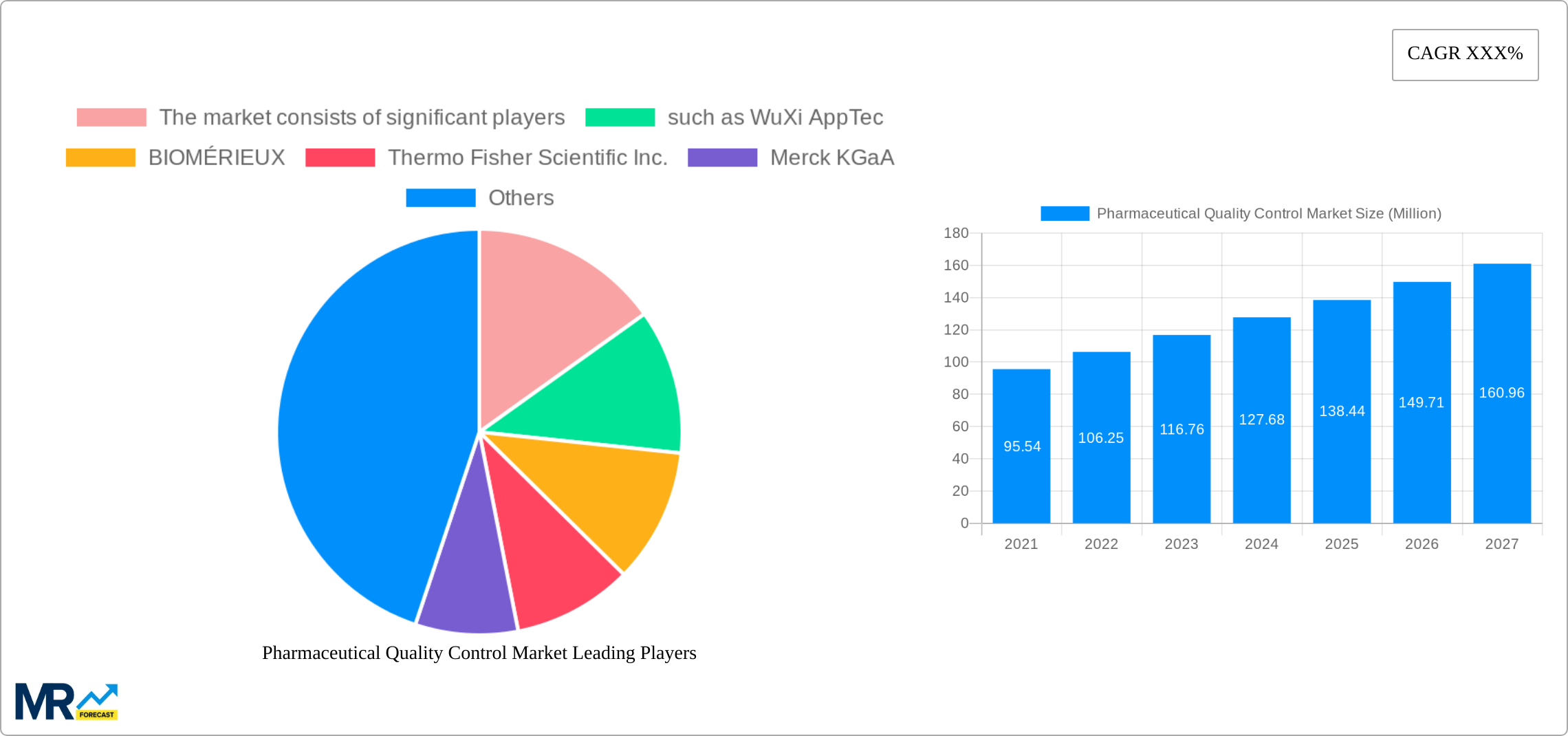
Key companies in the market include The market consists of significant players, such as WuXi AppTec, BIOMÉRIEUX, Thermo Fisher Scientific Inc., Merck KGaA, SGS Société Générale de Surveillance SA., Eurofins Scientific, Lucideon Limited, and Sartorius AG..
The market segments include Product Type, Analysis Type, End-user.
The market size is estimated to be USD XX Million as of 2022.
N/A
N/A
N/A
In March 2024, Element Materials Technology launched a rapid-response pharmaceutical testing service from its Manchester Life Sciences laboratory to support pharmaceutical companies by delivering advanced and complete testing solutions.
Pricing options include single-user, multi-user, and enterprise licenses priced at USD N/A, USD N/A, and USD N/A respectively.
The market size is provided in terms of value, measured in Million.
Yes, the market keyword associated with the report is "Pharmaceutical Quality Control Market," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Pharmaceutical Quality Control Market, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.