1. What is the projected Compound Annual Growth Rate (CAGR) of the Pharmaceutical Quality Control Testing?
The projected CAGR is approximately 12.98%.
 Pharmaceutical Quality Control Testing
Pharmaceutical Quality Control TestingPharmaceutical Quality Control Testing by Type (Sterility Testing, Bioburden Testing, Endotoxin Testing, Stability Testing, Extractable & Leachable Testing, Raw Material Testing, Others), by Application (Vaccines, Plasma Product, Drugs), by North America (United States, Canada, Mexico), by South America (Brazil, Argentina, Rest of South America), by Europe (United Kingdom, Germany, France, Italy, Spain, Russia, Benelux, Nordics, Rest of Europe), by Middle East & Africa (Turkey, Israel, GCC, North Africa, South Africa, Rest of Middle East & Africa), by Asia Pacific (China, India, Japan, South Korea, ASEAN, Oceania, Rest of Asia Pacific) Forecast 2026-2034
MR Forecast provides premium market intelligence on deep technologies that can cause a high level of disruption in the market within the next few years. When it comes to doing market viability analyses for technologies at very early phases of development, MR Forecast is second to none. What sets us apart is our set of market estimates based on secondary research data, which in turn gets validated through primary research by key companies in the target market and other stakeholders. It only covers technologies pertaining to Healthcare, IT, big data analysis, block chain technology, Artificial Intelligence (AI), Machine Learning (ML), Internet of Things (IoT), Energy & Power, Automobile, Agriculture, Electronics, Chemical & Materials, Machinery & Equipment's, Consumer Goods, and many others at MR Forecast. Market: The market section introduces the industry to readers, including an overview, business dynamics, competitive benchmarking, and firms' profiles. This enables readers to make decisions on market entry, expansion, and exit in certain nations, regions, or worldwide. Application: We give painstaking attention to the study of every product and technology, along with its use case and user categories, under our research solutions. From here on, the process delivers accurate market estimates and forecasts apart from the best and most meaningful insights.
Products generically come under this phrase and may imply any number of goods, components, materials, technology, or any combination thereof. Any business that wants to push an innovative agenda needs data on product definitions, pricing analysis, benchmarking and roadmaps on technology, demand analysis, and patents. Our research papers contain all that and much more in a depth that makes them incredibly actionable. Products broadly encompass a wide range of goods, components, materials, technologies, or any combination thereof. For businesses aiming to advance an innovative agenda, access to comprehensive data on product definitions, pricing analysis, benchmarking, technological roadmaps, demand analysis, and patents is essential. Our research papers provide in-depth insights into these areas and more, equipping organizations with actionable information that can drive strategic decision-making and enhance competitive positioning in the market.
The pharmaceutical quality control testing market, valued at $9,970.5 million in 2025, is projected to experience robust growth, driven by stringent regulatory requirements for drug safety and efficacy, increasing pharmaceutical production, and the rising prevalence of chronic diseases globally. This necessitates comprehensive testing across various stages, from raw materials to finished products. Key testing segments include sterility, bioburden, endotoxin, stability, extractable & leachable, and raw material testing, catering to applications such as vaccines, plasma products, and drugs. The market's growth is further fueled by technological advancements in analytical techniques, automation, and the adoption of advanced data analytics for improved efficiency and accuracy in quality control processes. Competitive landscape analysis reveals a mix of established players like BioMérieux, SGS, Charles River Laboratories, and Thermo Fisher Scientific, alongside smaller specialized companies. These companies are constantly innovating to meet evolving industry demands for faster, more sensitive, and cost-effective testing solutions.
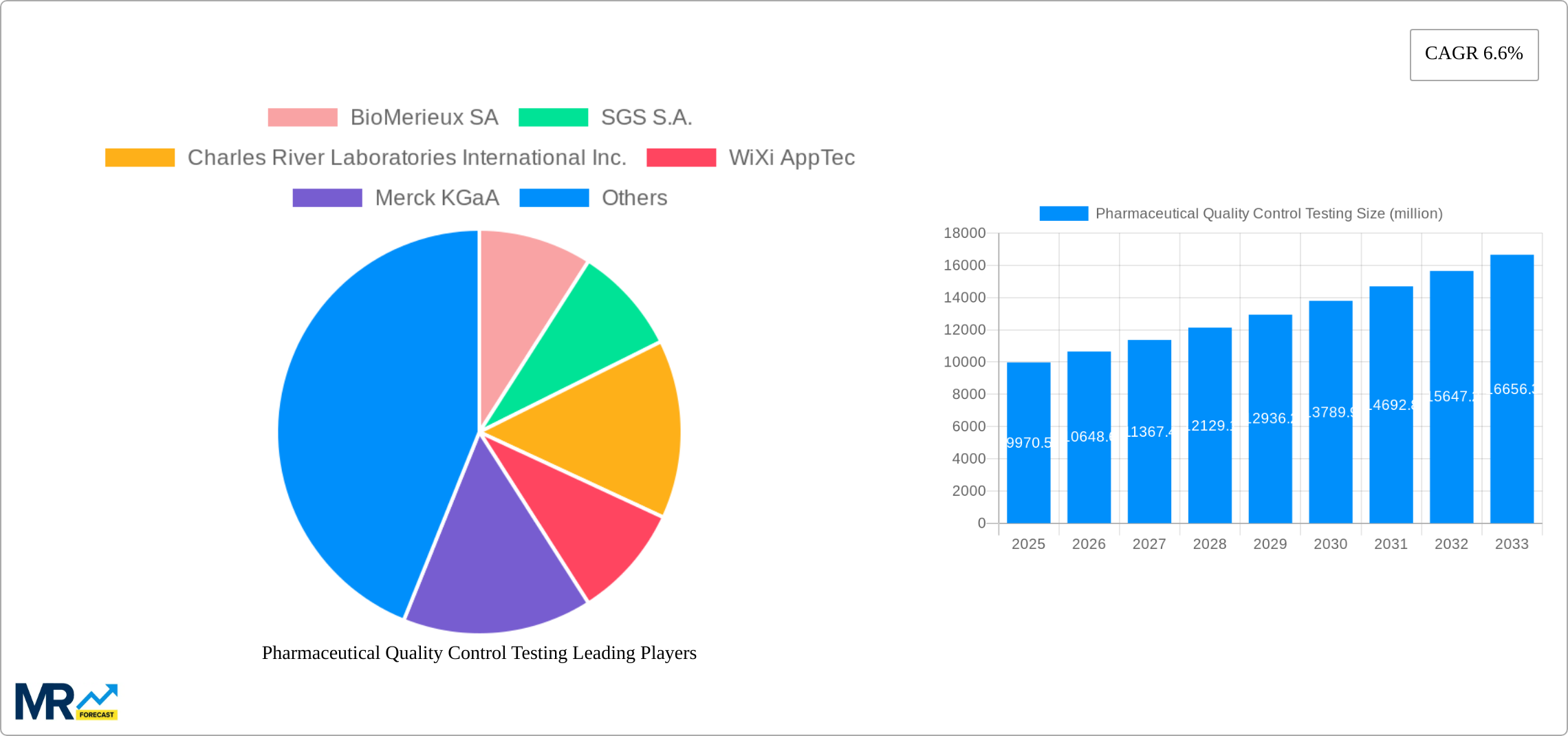

Growth in the forecast period (2025-2033) will be influenced by several factors. Continued investment in research and development within the pharmaceutical sector will stimulate demand for advanced testing services. Expanding global healthcare infrastructure in emerging economies, particularly in Asia-Pacific, will also contribute to market expansion. However, potential restraints include the high cost of advanced testing technologies and the complexities associated with regulatory compliance in different regions. To maintain a competitive edge, companies are focusing on strategic partnerships, mergers and acquisitions, and geographical expansions. The focus on personalized medicine and the development of innovative drug delivery systems are also expected to drive the demand for specialized quality control testing services in the coming years. The market is expected to maintain a healthy CAGR, reflecting the ongoing importance of robust quality control in ensuring patient safety and the success of pharmaceutical products.
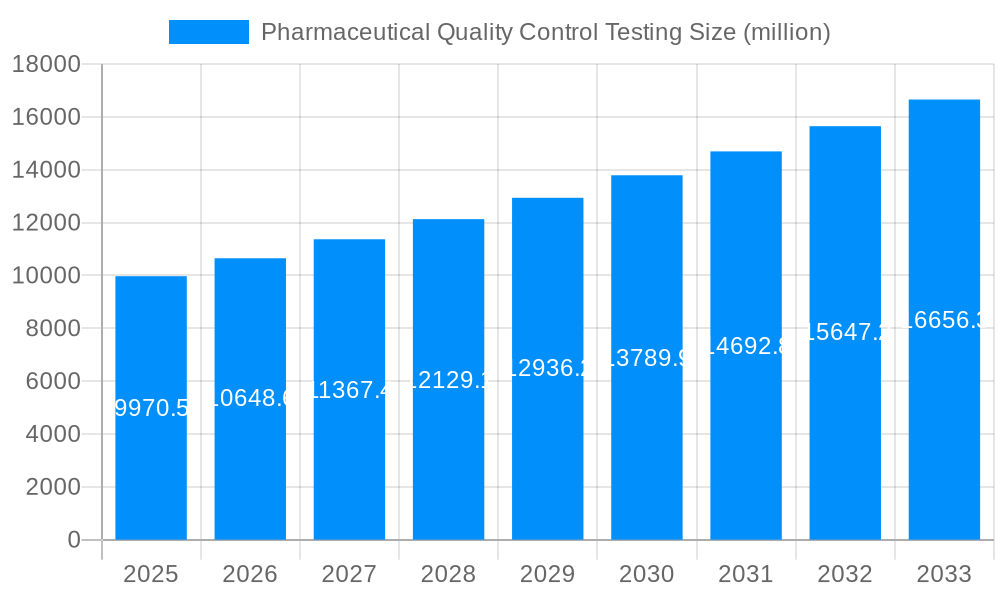

The global pharmaceutical quality control testing market is experiencing robust growth, projected to reach multi-billion dollar valuations by 2033. Driven by stringent regulatory requirements, increasing focus on patient safety, and the growing complexity of pharmaceutical products, the market is witnessing a significant shift towards advanced testing methodologies and automation. The historical period (2019-2024) saw a steady increase in demand, particularly fueled by the rise of biopharmaceuticals and the need for comprehensive quality assurance throughout the drug development lifecycle. The base year (2025) marks a pivotal point, with the market expected to expand significantly during the forecast period (2025-2033). This growth is attributed to several factors, including the rising prevalence of chronic diseases, escalating R&D investments in new drug therapies, and the increasing adoption of outsourcing strategies by pharmaceutical companies. The market is characterized by a diverse range of testing services, encompassing sterility testing, bioburden testing, endotoxin testing, stability testing, and raw material testing, each contributing significantly to the overall market value. The substantial investments in innovative technologies, such as advanced analytical techniques (e.g., mass spectrometry, chromatography) and automation solutions, are further bolstering the market's trajectory. Key players are continuously expanding their service portfolios, forging strategic partnerships, and investing heavily in research and development to maintain their competitive edge. This intense competitive landscape is driving innovation and fostering the development of more efficient and accurate testing methods, resulting in improved patient safety and drug efficacy. The market's dynamism is fueled by a constant demand for improved regulatory compliance and a relentless pursuit of enhanced quality control practices within the pharmaceutical industry. The market value, exceeding several hundred million dollars in 2025, is set to experience exponential growth, surpassing billions of dollars by the end of the forecast period, driven by the factors previously mentioned.
Several key factors are driving the exponential growth of the pharmaceutical quality control testing market. Stringent regulatory frameworks enforced by global health agencies, such as the FDA and EMA, mandate rigorous quality control measures throughout the drug development and manufacturing processes. Non-compliance can result in significant penalties and market withdrawal, compelling pharmaceutical companies to invest heavily in comprehensive testing services. The increasing complexity of pharmaceutical products, including biologics, biosimilars, and advanced drug delivery systems, demands sophisticated and specialized testing procedures. The growing awareness among consumers and healthcare professionals regarding drug safety and efficacy is also placing greater emphasis on robust quality control measures. Furthermore, the rising prevalence of chronic diseases and the consequent surge in pharmaceutical consumption are expanding the market for quality control testing services. The outsourcing trend among pharmaceutical companies, who are increasingly delegating quality control testing to specialized CROs (Contract Research Organizations), further contributes to the market's growth. This allows pharmaceutical companies to focus on their core competencies while ensuring that their products meet the highest quality standards. Finally, continuous advancements in testing technologies, such as automation and advanced analytical techniques, are significantly improving the speed, accuracy, and efficiency of quality control testing, thereby driving market growth.
Despite the significant growth potential, the pharmaceutical quality control testing market faces several challenges. High costs associated with advanced testing equipment, skilled personnel, and regulatory compliance can pose a significant barrier to entry for smaller companies and limit the accessibility of these services in certain regions. The complex regulatory landscape varies across different countries, creating complexities in standardizing procedures and harmonizing testing protocols. This necessitates significant investments in staying updated with ever-changing regulations and adapting testing methodologies accordingly. Furthermore, the development and validation of new testing methods for emerging drug technologies (e.g., gene therapy, cell therapy) can be time-consuming and expensive, slowing down the overall market adoption. Maintaining data integrity and ensuring the traceability of testing results are crucial aspects of quality control, and lapses in these areas can lead to significant reputational damage and regulatory issues. The competition among testing service providers is intense, requiring companies to constantly innovate and enhance their offerings to maintain a competitive edge. Finally, fluctuations in the global pharmaceutical market, influenced by factors such as economic downturns and changes in healthcare spending, can also impact the demand for testing services.
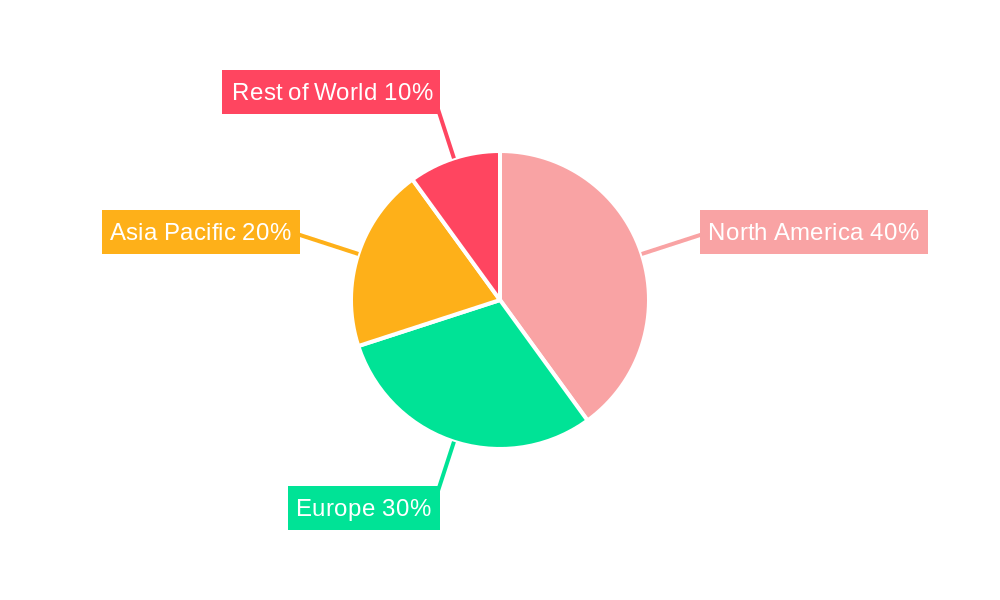
The North American market is expected to maintain its dominance in the pharmaceutical quality control testing market during the forecast period (2025-2033), driven by robust R&D spending in the pharmaceutical and biotechnology sectors and the presence of major pharmaceutical companies and contract research organizations (CROs). Europe is also poised for significant growth, fueled by increasing regulatory scrutiny and a robust pharmaceutical industry.
Dominant Segments:
The demand for these segments is further fueled by the growth of biologics, which necessitate more specialized and stringent testing protocols compared to traditional small-molecule drugs. The complexity and cost associated with testing these products drive the overall market value upwards. The high demand, coupled with the need for precise and reliable results, makes these segments key drivers of market growth in the coming years.
The pharmaceutical quality control testing industry is experiencing remarkable growth, propelled by several catalysts. The increasing complexity of pharmaceutical products, coupled with stringent regulatory requirements, necessitates more sophisticated and rigorous testing. The outsourcing trend, with pharmaceutical companies increasingly leveraging CROs for testing services, fuels market expansion. Advancements in analytical technologies, offering enhanced speed, accuracy, and efficiency, further contribute to growth. Finally, the rise in chronic diseases and the associated surge in pharmaceutical consumption significantly drive the demand for quality control testing services, ensuring the safety and efficacy of life-saving drugs.
This report provides an in-depth analysis of the pharmaceutical quality control testing market, offering valuable insights into current trends, growth drivers, and challenges. It covers a wide range of testing services, including sterility, bioburden, endotoxin, stability, extractable & leachable, and raw material testing, across key applications such as vaccines, plasma products, and drugs. The report also profiles leading players in the industry, highlighting their strategies and market positions. This comprehensive overview is valuable for businesses seeking to understand the dynamics of the pharmaceutical quality control testing market and make informed strategic decisions.

| Aspects | Details |
|---|---|
| Study Period | 2020-2034 |
| Base Year | 2025 |
| Estimated Year | 2026 |
| Forecast Period | 2026-2034 |
| Historical Period | 2020-2025 |
| Growth Rate | CAGR of 12.98% from 2020-2034 |
| Segmentation |
|




Note*: In applicable scenarios
Primary Research
Secondary Research

Involves using different sources of information in order to increase the validity of a study
These sources are likely to be stakeholders in a program - participants, other researchers, program staff, other community members, and so on.
Then we put all data in single framework & apply various statistical tools to find out the dynamic on the market.
During the analysis stage, feedback from the stakeholder groups would be compared to determine areas of agreement as well as areas of divergence
The projected CAGR is approximately 12.98%.
Key companies in the market include BioMerieux SA, SGS S.A., Charles River Laboratories International Inc., WiXi AppTec, Merck KGaA, Thermo Fisher Scientific Inc., Toxikon Corporation, Sartorius AG, REMI Group, Perkin Elmer, Agilent Technologies Inc, Waters Corporation, Charles River Laboratories International Inc, Catalent, Inc, Eurofins Scientific SE, .
The market segments include Type, Application.
The market size is estimated to be USD XXX N/A as of 2022.
N/A
N/A
N/A
N/A
Pricing options include single-user, multi-user, and enterprise licenses priced at USD 3480.00, USD 5220.00, and USD 6960.00 respectively.
The market size is provided in terms of value, measured in N/A.
Yes, the market keyword associated with the report is "Pharmaceutical Quality Control Testing," which aids in identifying and referencing the specific market segment covered.
The pricing options vary based on user requirements and access needs. Individual users may opt for single-user licenses, while businesses requiring broader access may choose multi-user or enterprise licenses for cost-effective access to the report.
While the report offers comprehensive insights, it's advisable to review the specific contents or supplementary materials provided to ascertain if additional resources or data are available.
To stay informed about further developments, trends, and reports in the Pharmaceutical Quality Control Testing, consider subscribing to industry newsletters, following relevant companies and organizations, or regularly checking reputable industry news sources and publications.